Table of Contents
ToggleComprehensive Master Guide: HIV Infection & AIDS
I. Introduction: What is HIV/AIDS?
To understand the clinical management of this disease, we must first master the strict definitions and the biological nature of the virus itself.
- HIV (Human Immunodeficiency Virus): A human retrovirus infection that specifically targets, invades, and destroys human T-helper lymphocytes (CD4+ cells). Because CD4+ cells act as the "generals" of the human immune system, their destruction critically compromises the host's entire immune defense network.
- AIDS (Acquired Immunodeficiency Syndrome): The final, life-threatening, symptomatic stage of HIV disease. It occurs when the virus has severely depleted the CD4+ cell count (typically below 200 cells/mm³), leaving the host's immunity completely devastated and highly susceptible to opportunistic infections and infection-related cancers that a normal immune system would easily fight off.
The Nature of the Virus:
- Continuous Replication: Once an individual is infected with HIV, the virus is never completely dormant in their system; it replicates continuously, producing billions of new viral copies every single day.
- Immune Evasion: While the immune system can clear most other viruses out of the body (like the flu or common cold), it cannot get rid of HIV. Scientists attribute this to the virus hiding its DNA directly inside the host's own cellular genome, effectively creating a "reservoir" of hidden virus that the immune system cannot see or destroy.
- Curability vs. Manageability: HIV infection is NOT curable, but it is highly PREVENTABLE and incredibly manageable. Antiretroviral Therapy (ART) can help patients live long, healthy lives by suppressing viral replication to undetectable levels, effectively halting disease progression.
II. Origin & Historical Timeline
Where did it come from?
Through extensive genomic sequencing, scientists identified a specific type of chimpanzee in West Africa as the original source of the HIV infection in humans. The virus found in these chimpanzees is called Simian Immunodeficiency Virus (SIVcpz).
The "Cut Hunter" Hypothesis: The virus most likely jumped to humans (a zoonotic spillover) when humans hunted these chimpanzees for bushmeat. During the butchering process, contact with the chimpanzee's infected blood allowed the virus to enter human cuts or wounds. Over several decades, the virus slowly mutated, adapted to the human host, and spread across Africa and later into other parts of the world via urbanization and global travel.
Crucial Historical Timeline:
| Year | Milestone Event |
|---|---|
| 1981 | First reported case of an unexplained, severe immune collapse occurred among clusters of homosexual men in New York and California, presenting with rare pneumonias and skin cancers. |
| 1982 | First official use of the "AIDS" acronym by the CDC. |
| 1984 | The causative agent (the HIV virus itself) was definitively isolated and identified by scientists. |
| 1985 | The first HIV Antibody Test (ELISA) was developed, allowing the blood supply to be screened. |
| 1986 | First global use of the "HIV" acronym to standardize the nomenclature. |
| 1987 | First medication available for HIV treatment: Zidovudine (AZT), an NRTI originally developed as a failed cancer drug. |
| 1988 | The First World AIDS Day was established (December 1st) to raise global awareness. |
| 1992 | The first combination HIV therapy (HAART - Highly Active Antiretroviral Therapy) was introduced, miraculously turning a universally fatal disease into a manageable chronic condition. |
| 2002 | First rapid diagnostic HIV test kit was approved by the FDA, revolutionizing point-of-care testing. |
III. Epidemiology & Types of HIV
HIV/AIDS is a global pandemic. The statistical data highlights the massive, ongoing burden of the disease across the planet.
Global Statistics (UNAIDS Data Baseline):
- 76.1 million people have become infected with HIV since the start of the epidemic.
- 35.0 million people have died from AIDS-related illnesses since the start of the epidemic.
- 36.7 million people globally are currently living with HIV (Comprising roughly 34.5 million adults, 17.8 million women aged 15+, and 2.1 million children <15 years).
- 1.8 million people become newly infected annually.
- 1.0 million people die from AIDS-related illnesses annually.
Regional Epidemiology Examples:
- United States: Recent diagnoses by transmission category reveal a distinct demographic skew: Male-to-Male Sexual Contact (67%), Heterosexual Contact (24%), Injection Drug Use (6%), and Dual Risk (Male-to-Male + IDU: 3%).
- Egypt (Middle East/North Africa): Egypt is historically a low-HIV-prevalence country. However, between 2006 and 2011, prevalence rates increased tenfold. New cases grew from roughly 400/year (pre-2011) to about 880/year in 2014. Currently, over 11,000 people live with HIV in Egypt. Unsafe behaviors among at-risk populations and limited condom use place it at severe risk for a broader, explosive epidemic.
Types of HIV:
The most common type globally. It is highly virulent, highly infectious, and is the primary agent responsible for the global AIDS epidemic. When we discuss "HIV" clinically, we are almost always referring to HIV-1.
A rare type, restricted mainly to West Africa (traced back to the sooty mangabey monkey reservoir). It is significantly less virulent and progresses much more slowly than HIV-1, but it still ultimately results in AIDS. Both types are transmitted via identical routes.
IV. Viral Classification & Structural Anatomy
Understanding the physical and genetic structure of the HIV virion is critical because every single structural component is a potential target for diagnostic laboratory tests or pharmacological antiretroviral drugs.
Classification:
- Family: Retroviridae (Expansion: "Retro" means backwards. Normal biology dictates DNA makes RNA. Retroviruses violate this central dogma by using an enzyme to convert their RNA backwards into DNA).
- Genus: Lentivirus (Expansion: "Lenti" means slow. This perfectly describes the long clinical latency period—often 10 years—between initial infection and the development of severe AIDS symptoms).
Viral Genome (Genetics):
The virus contains Diploid copies of positive-sense single-stranded RNA. This means the virus carries two identical, ready-to-read strands of RNA.
Structural Components of the Virion:
- Size: Extremely small, approximately 100-120 nm in diameter.
- Envelope (Spiked Lipid Bilayer): The outermost layer. Fascinatingly, the virus does not make this layer itself; it actually steals (derives) this lipid membrane from the human host cell's membrane as it buds out!
- Envelope Glycoproteins: Embedded into the stolen lipid envelope are viral proteins:
- gp120 (Docking Glycoprotein): A surface glycoprotein that acts as the "key," attaching directly to CD4 receptors on the host cell.
- gp41 (Transmembrane Glycoprotein): Acts as the "hinge" or "harpoon" that mediates the actual physical fusion of the viral envelope with the host cell membrane, pulling the virus inside.
- Matrix (p17): A supportive protein shell located just beneath the lipid envelope, providing structural integrity to the virion.
- Capsid (p24): A bullet or cone-shaped core structure deep inside the virus containing the viral RNA and enzymes. Clinical Note: The p24 antigen is highly abundant and is a primary diagnostic marker used in early-stage HIV laboratory blood tests!
The Three Crucial Viral Enzymes:
- Reverse Transcriptase: Converts viral single-stranded RNA into double-stranded DNA. This conversion allows the viral genetic material to be compatible with the human host cell. (Note: This enzyme is highly error-prone, which is why HIV mutates so rapidly, easily developing drug resistance).
- Integrase: Integrates (splices) the newly formed viral DNA directly into the host cell's genome (the CD4 cell's own DNA), making the virus a permanent, lifelong part of the host cell.
- Protease: Cleaves (cuts) long, non-functional precursor polyproteins into mature, infectious, functional proteins. Without protease, new HIV particles that bud off remain immature, defective, and completely non-infectious!
To remember the glycoproteins and capsid numbers, think of a lock, a door, and a vault:
- gp120: Is the "1-2-0" combination that unlocks the outside (attaches to CD4).
- gp41: "For-ty one" forces the door open (fusion).
- p24: Is the precious cargo protected "24/7" inside the capsule (Capsid protein / Early Diagnostic marker).
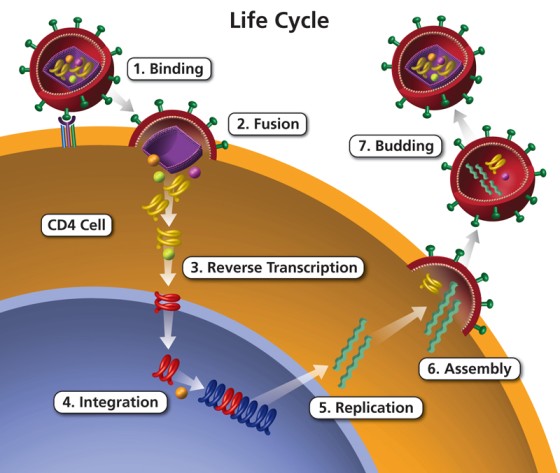
V. Pathogenesis: The 7 Steps of the HIV Life Cycle
The HIV Life Cycle involves seven distinct, sequential steps where the virus hijacks the machinery of the CD4 cells to multiply and spread throughout the body. Every step is a specific pharmacological target for Antiretroviral Therapy (ART).
- Binding (Attachment):
- Mechanism: The viral gp120 binds to CD4 receptors on the surface of T-helper cells, macrophages, and dendritic cells. It MUST also bind to a co-receptor (either CCR5 or CXCR4) to enable entry.
- Pharmacological Target: Blocked by CCR5 Antagonists (e.g., Maraviroc) and Post-attachment inhibitors.
- Fusion:
- Mechanism: Mediated by gp41. The HIV envelope fuses with the CD4 cell membrane, allowing the viral capsid to plunge into the cytoplasm of the CD4 cell.
- Pharmacological Target: Blocked by Fusion Inhibitors (e.g., Enfuvirtide).
- Reverse Transcription:
- Mechanism: Inside the cytoplasm, HIV releases Reverse Transcriptase to convert its viral RNA into HIV DNA. This conversion is mandatory for the virus to enter the human nucleus.
- Pharmacological Target: Blocked by Nucleoside Reverse Transcriptase Inhibitors (NRTIs) and Non-Nucleoside Reverse Transcriptase Inhibitors (NNRTIs).
- Integration:
- Mechanism: The newly formed viral DNA enters the CD4 cell nucleus. HIV releases the enzyme Integrase to splice and physically insert its viral DNA directly into the host's cellular DNA.
- Pharmacological Target: Blocked by Integrase Strand Transfer Inhibitors (INSTIs) (e.g., Dolutegravir).
- Replication:
- Mechanism: Once integrated, HIV hijacks the CD4 cell's machinery to transcribe and translate its DNA, creating long, continuous chains of HIV proteins and thousands of copies of viral RNA.
- Assembly:
- Mechanism: New HIV proteins and HIV RNA migrate to the inner surface of the cell membrane and assemble into an immature (non-infectious) HIV particle pushing against the cell wall.
- Budding & Maturation:
- Mechanism: The newly formed immature HIV pushes itself out of the host cell (stealing some of the host's lipid membrane to form its envelope). Finally, the new HIV virus releases Protease, which acts as molecular scissors to break the long protein chains into smaller, mature, functional proteins. This creates a mature, highly infectious virus ready to attack other CD4 cells.
- Pharmacological Target: Blocked by Protease Inhibitors (PIs) (e.g., Darunavir).
❓ Applied Pharmacology Question
Case: A patient with HIV is started on an antiretroviral regimen that includes a drug called Darunavir, which is a Protease Inhibitor. If this drug works perfectly, what will the patient's blood look like under an electron microscope?
Answer: The blood will contain numerous HIV particles, but they will all be immature, non-functional virions. Because the protease enzyme is blocked, the long polyproteins cannot be cleaved. This means the virus can successfully bud out of the cell, but it cannot mature into an infectious state to infect new CD4 cells! The infection is halted in its tracks.
VI. HIV Transmission Dynamics: Body Fluids & Viability
For HIV to be transmitted, a specific body fluid containing a sufficient, concentrated viral load must come into direct contact with a mucous membrane, damaged tissue, or be injected directly into the bloodstream.
These fluids carry massive viral loads and have high chances of transmitting HIV infection:
- Blood
- Semen (cum)
- Pre-seminal fluid (pre-cum)
- Breast milk
- Vaginal fluids
- Rectal (anal) mucous
Transmission of the infection through these fluids is biologically highly unlikely/impossible:
- Saliva, Tears, Sweat, Urine, Feces, Nasal fluid, Vomit.
Crucial Exception: If blood or open wounds are present and mixed with these fluids (e.g., visible blood in saliva from severely bleeding gums), the fluid immediately becomes an infectious hazard.
Occupational Exposure Fluids (Healthcare Workers):
Doctors, nurses, and lab technicians may be exposed to other specific body fluids that carry high concentrations of HIV. Standard precautions (gloves, goggles) must be strictly used when handling:
- Amniotic fluid (surrounding the fetus during childbirth)
- Cerebrospinal fluid (CSF) (surrounding the brain/spinal cord during lumbar punctures)
- Synovial fluid (surrounding bone joints during aspirations)
Viability of the Virus Outside the Body:
- HIV is an extremely fragile virus outside the human body.
- Hot water, soap, bleach (1:10 concentration), and standard alcohol easily destroy the viral lipid envelope, instantly killing the virus.
- The length of time the virus can survive outside the body depends directly on the amount of HIV present in the fluid and the environmental conditions (temperature, moisture).
- The CDC reports that drying HIV fluids reduces the viral load by 90-99% within several hours, rendering it virtually non-infectious on surfaces like tables or clothing.
VII. Points of Entry & Transmission Routes
A. Points of Entry into the Host:
- Percutaneously (Through the skin): Puncture/needle stick, or any break, cut, or abrasion in the skin.
- Mucous Membranes: Eyes, Nose, Mouth, Genitals, and Anus.
The most efficient way to transmit HIV sexually is through anal sex, followed by vaginal, and then oral. Why is anal sex so overwhelmingly risky? It comes down to histology.
The vagina is lined with tough, multi-layered stratified squamous epithelium designed by nature to withstand intense friction and trauma during childbirth and intercourse. Conversely, the rectum is lined with a delicate, single layer of simple columnar epithelium designed purely for gentle water absorption. It is highly vascular and tears incredibly easily during friction (micro-tears), creating immediate, wide-open direct pathways for the virus in the semen to enter the recipient's bloodstream.
B. Most Likely Routes of Transmission:
- Unprotected Sex: With an HIV infected person (Anal > Vaginal > Oral).
- Blood-to-Blood Contact: Mostly via sharing contaminated injection needles (IV drug abuse), tattoo needles, piercing needles, syringes, blades, or sharp equipment. (Note: HIV can live in a used needle/syringe for up to 42 days depending on temperature, because the blood trapped inside the vacuum of the barrel remains moist and protected from the air!).
- Mother-to-Child Transmission (MTCT): Also known as vertical transmission. Can occur during Pregnancy (in-utero crossing the placenta), during Delivery (exposure to maternal blood in the birth canal), or via Breastfeeding.
- Blood Transfusions / Organ Transplants: Receiving contaminated blood products or organs. However, there is now a very low risk of this route in developed nations because donors undergo extensive, mandatory nucleic acid testing (NAT).
- Sexual Abuse: Such as rape, which often involves violent tissue trauma, tearing, and bleeding, massively increasing transmission risk.
C. Less Likely Routes of Transmission:
- Eating food pre-chewed by an HIV infected person (only possible if severe dental/gum wounds and blood are present in both the caregiver and the infant).
- Being bitten by an HIV infected person (only if it results in a severe skin break with extensive blood exchange).
- Contact of an open wound (broken skin) with HIV infected blood or fluids.
- Open-mouth (French) kissing (only if sores or bleeding gums are actively present in both parties).
D. How HIV is NOT Transmitted (Debunking Myths):
- Air or drinking water: Cannot be contracted from the same cooking pot or breathing the same air as an infected person.
- Insects: Including mosquitoes, ticks, or bedbugs. (The virus cannot replicate inside a mosquito's gut and is instantly digested as food by the insect).
- Saliva, tears, or sweat: There is NO documented case of HIV being transmitted by spitting or crying.
- Casual Contact: Shaking hands, hugging, sharing dishes/food utensils, sharing swimming pools, or sharing toilet seats.
- Social Kissing: Closed-mouth (cheek) kissing.
VIII. High Risk Populations
According to CDC data tracking new diagnoses, the highest risk groups are clearly defined by behavioral and demographic factors:
- Homosexual (Gay) and Bisexual Men: Accounting for 67% of new transmissions.
- Heterosexual Contact: Accounting for 24%.
- IV Drug Abusers: Accounting for 6%.
- Male-to-Male + IV Drug Use: Accounting for 3%.
Other Vulnerable / High-Risk Groups Include:
- Young people (20–29 years old) engaging in exploratory behaviors.
- HIV-negative spouses of HIV-positive persons (Serodiscordant couples).
- Newborn babies of HIV-positive mothers lacking access to PMTCT care.
- Women working in commercial sex (high partner turnover, trauma risk).
- Healthcare workers (occupational risk of being accidentally wounded by contaminated needles/sharps).
- Researchers working with concentrated HIV biosamples in laboratories.
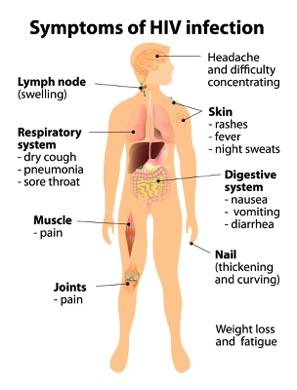
IX. Stages of HIV Infection & Pathogenesis
Upon contracting HIV, the infected person often doesn't have obvious, immediate signs indicating a deadly infection. However, while signs may be absent, the virus continuously damages CD4 cells and the immune system. The disease progresses through 4 recognized clinical stages.
The Window Period is the amount of time it takes for an infected individual to seroconvert (produce detectable levels of HIV antibodies in response to the infection). This period usually lasts 2 to 6 weeks.
Clinical Significance: During this time, the patient's viral load is astronomically high, and they are highly contagious. However, standard HIV Antibody tests (like the rapid ELISA prick test) will come back NEGATIVE because the body hasn't built the antibodies yet! This leads to severe false-negative diagnoses.
STAGE 1: Primary / Acute HIV Infection
- Timeline: Occurs 2 to 6 weeks after initial exposure.
- Pathology (Acute Retroviral Syndrome): Characterized by wide dissemination of the virus, seeding of lymphoid organs, rapid viral replication, incredibly high viremia (millions of viral copies per mL in the blood), and a sudden, sharp drop in CD4 cells as the host immune system attempts to mount a massive antibody response.
- Clinical Presentation: Highly non-specific "Flu-like" or "Mononucleosis-like" symptoms that last for 2 to 4 weeks. Because they are non-specific, it is frequently misdiagnosed as malaria, typhoid, or the common flu.
- High fever
- Sore throat (pharyngitis, mouth sores, thrush)
- Headache
- Persistent Generalized Lymphadenopathy (PGL) (Significantly swollen glands in the neck/armpits)
- Stomach upset, Nausea, Vomiting & Diarrhea
- Skin rashes (maculopapular rash on the trunk)
- Joint and Muscle pain (Myalgia/Arthralgia)
- Tiredness, Malaise, Fatigue, and mild Weight loss
- Liver & Spleen enlargement (Hepatosplenomegaly)
❓ Applied Clinical Case
Case: A 24-year-old male presents to the clinic with a fever, severe sore throat, a rash on his chest, and swollen lymph nodes in his neck. He mentions having unprotected sex with a new partner 3 weeks ago. The doctor orders an HIV ELISA (Antibody) test, which comes back negative.
Does a negative ELISA test mean he does not have HIV? What stage is he likely in?
Answer: NO! He is exhibiting classic signs of Stage 1 (Acute Retroviral Syndrome). He is currently deep in the Window Period. His body has not yet produced enough antibodies to trigger a positive ELISA test. To secure a diagnosis, a Viral Load (PCR) test or a p24 Antigen test must be ordered immediately, which would likely show massive, active viremia.
STAGE 2: Chronic HIV Infection / Asymptomatic Stage
- Timeline: Also known as Clinical Latency. Can last for a decade (approx. or >10 years) without treatment. Length depends heavily on the patient's baseline immunity and ART adherence.
- Pathology: The virus retreats into the lymph nodes and is growing very slowly, reproducing at much lower levels. The CD4 count rebounds slightly but steadily, relentlessly declines over the years. HIV Antibodies are now fully detectable (Positive ELISA).
- Clinical Presentation: The patient is largely free from symptoms (though there may be persistently swollen glands). Minor mucocutaneous infections may appear (e.g., Herpes Zoster / Shingles, and recurrent Upper Respiratory Tract Infections - URTI).
- Weight Loss: Less than 10% of total body weight.
- Transmission Risk: Although the viral level in the blood drops to low levels, the patient CAN STILL SPREAD HIV TO OTHERS! They feel perfectly fine, making this the stage where the disease is spread most widely in populations.
STAGE 3: Symptomatic HIV Disease
- Pathology: The immune system heavily deteriorates. The CD4 count drops significantly (typically between 200–499 cells/mm³). The viral load begins to climb again.
- Clinical Presentation: The patient begins showing moderate, persistent symptoms indicating systemic immune failure:
- Loss of weight: Greater than 10% of total body weight.
- Chronic diarrhea: Unexplained, lasting greater than 1 month.
- Prolonged fever: Relentless, lasting greater than 1 month.
- Oral Candidiasis (Thrush): Thick white plaques on the tongue, and Oral Hairy Leukoplakia (white, corrugated patches on the sides of the tongue caused by EBV).
- Severe bacterial infections and Pulmonary Tuberculosis (TB).
- Persistent Vulvovaginal candidiasis in women.
STAGE 4: AIDS (Acquired Immunodeficiency Syndrome)
- Pathology: The final and most severe stage. Host immunity is badly damaged, making the patient highly susceptible to lethal Opportunistic Infections and cancers.
- Diagnostic Criteria: Defined strictly by a CD4 Cell Count below 200 cells/mm³ OR the emergence of specific AIDS-defining illnesses (regardless of CD4 count).
- Clinical Presentation: A life-threatening condition presenting with:
- HIV Wasting Syndrome: Extreme, rapid weight loss with severe muscle atrophy.
- Severe respiratory signs: Shortness of breath (SOB) and chronic cough.
- Skin lesions, poor wound healing, and extreme night sweats soaking the bedsheets.
- Central Nervous System (CNS) Complications: HIV actively crosses the blood-brain barrier causing HIV Encephalopathy, AIDS Dementia Complex, Confusion, Personality changes, Visual changes, Seizures, and HIV-associated Progressive Encephalopathy (HPE). (Occurs in ~70% of end-stage AIDS clients).
X. Opportunistic Infections (OIs)
Opportunistic infections are caused by normal, everyday pathogens that take advantage of a severely weakened host immunity. They Do Not usually cause disease in a healthy immune system. However, they occur far more often and are far more severe in HIV/AIDS patients. Without ART, this phase is generally fatal.
1. Fungal Infections:
- Candidiasis: Can severely affect the stomatitis (mouth/oral thrush), esophagus (causing severe pain when swallowing), trachea, bronchi, lungs, or vagina.
- Cryptococcosis: Causes deadly fungal meningitis in the brain.
- Pneumocystis carinii pneumoniae (PCP): A classic, deadly AIDS-defining fungal pneumonia (recently renamed Pneumocystis jirovecii). Causes severe "ground-glass" opacities in the lungs on X-ray.
2. Bacterial Infections:
- Tuberculosis (TB): Both Pulmonary (usually Stage 3) and Extrapulmonary (spreading to bones/organs in Stage 4). TB is the leading cause of death among people living with HIV globally.
- Mycobacterium Avium Complex (MAC): A severe systemic bacterial infection resembling TB but disseminated throughout the blood and bone marrow.
- Severe recurrent bacterial pneumonias.
3. Viral Infections:
- Cytomegalovirus (CMV): Can cause permanent blindness (CMV retinitis) and severe painful GI ulcers.
- Herpes Simplex Virus (HSV): Causes chronic, non-healing ulcers lasting more than a month.
- Varicella-Zoster Virus (VZV): Presents as severe, multi-dermatomal Shingles.
4. Parasitic (Protozoal) Infections:
- Toxoplasmosis: A parasitic infection of the brain (often acquired from cat feces or undercooked meat) causing severe ring-enhancing lesions, seizures, and encephalitis.
- Cryptosporidiosis: Causes massive, relentless, watery gastrointestinal diarrhea leading to extreme dehydration.
5. Malignancies (AIDS-Defining Cancers):
- Kaposi's Sarcoma (KS): A cancer caused by Human Herpesvirus 8 (HHV-8) that causes dark purple/red patches of abnormal tissue to grow under the skin, in the lining of the mouth, nose, throat, and internal organs.
- Non-Hodgkin's Lymphoma and Hodgkin's Lymphoma.
- Invasive Cervical Carcinoma: Driven by Human Papillomavirus (HPV) taking advantage of the depressed immune system to rapidly cause cervical cancer in women.
Remember that when CD4 drops below 200, the patient is at risk for "The Big C's":
- Candidiasis (Esophageal/Lung)
- Cryptococcus (Meningitis)
- Cryptosporidium (Diarrhea)
- CMV (Cytomegalovirus Retinitis)
- Carcinomas (Kaposi's & Cervical)
XI. HIV Diagnosis: Laboratory Tests
Because the acute symptoms of HIV are so non-specific, laboratory diagnosis is the only definitive way to confirm infection. Tests are divided into those that look for the antibodies and those that look for the virus itself.
A. Antibody Tests (Detect Host Response):
- ELISA / EIA (Enzyme-Linked Immunosorbent Assay):
- Tests for HIV Antibodies, NOT the virus itself!
- Samples used: Blood, Serum, Plasma, Saliva, Urine.
- Usually tests positive 2 to 8 weeks after infection.
- Clinical Pitfall: Because it relies on antibodies, an infected person can test Negative during the "Window Period" (few weeks to few months after infection).
- Western Blot: If an ELISA test shows HIV-Positive results, a Western Blot cross-check MUST be done to confirm the results (it is highly specific and checks for specific viral protein antibodies). If the Western Blot is positive, HIV infection is definitively confirmed.
B. Quick / Rapid HIV Diagnosis Tests:
- Rapid HIV Antibodies Test: Rapid & easy to perform. Uses a finger-prick blood sample. Detection occurs within 30 minutes. (Common in resource-limited settings).
- OraQuick: An FDA-approved oral swab in-home test for HIV-1 and HIV-2. Results in 20 minutes.
- Crucial Rule: If ANY of these rapid/home tests show a positive result, it must be confirmed by a standard blood test (Western Blot or Geenius assay).
C. Tests for the Virus (Detect Viral Particles):
- Qualitative PCR (Polymerase Chain Reaction): Tests if the virus DNA/RNA is present. Highly useful in newborns where maternal antibodies crossing the placenta would cause a false positive on an ELISA test.
- Antigen p24 Test: Detects the p24 capsid protein of the virus itself. Can detect HIV much earlier than antibody tests (typically 18 – 45 days post-exposure).
XII. Monitoring Therapy: Viral Load & CD4 Counts
1. Viral Load Test (Quantitative PCR):
- Measures the exact amount of HIV-RNA in the blood (Number of copies of HIV-RNA in a milliliter of blood).
- Can be tested very early: 1 to 4 weeks from HIV exposure.
- Clinical Use: It is the primary test used in the evaluation of the efficacy of Antiretroviral Therapy (ART). A successful ART regimen should cause the Viral Load to fall dramatically within 3 to 6 months of treatment.
- What does a HIGH (↑↑) Viral Load indicate?
- High HIV amount in the blood.
- Fast damage to CD4 cells (CD4 count will fall rapidly).
- High risk of developing AIDS and death.
- Treatment failure: The patient may not be taking their ART medications (non-compliance), or the virus has mutated and is not responding well to the ART.
2. CD4 Cell Counts:
- Normal blood values for CD4 cells are 500 – 1500 cells/mm³.
- Since HIV specifically targets and destroys CD4 cells, this count is a vital biomarker to assess:
- The state of the patient's immune system.
- HIV disease progression.
- Efficacy of HIV Therapy.
- Should be measured every 3 to 6 months in the first 2 years of HIV infection.
- What does a LOW (↓↓) CD4 count indicate? Severe damage to CD4 cells, imminent risk of developing AIDS (count < 200) and opportunistic infections, or ART failure/non-compliance.
XIII. Management of HIV: Antiretroviral Therapy (ART)
HIV is NOT curable. However, significant advances have been made since the introduction of the first drug, Zidovudine (AZT), in 1987. With the advent of HAART (Highly Active Antiretroviral Therapy), HIV is now manageable as a chronic disease for patients who have access to medication and achieve durable virologic suppression.
Goals of HIV Therapy:
- Improving the patient's quality of life and reducing morbidity/mortality.
- Restoring the patient's CD4 cell counts.
- Preventing HIV replication and reducing Viral Load to an undetectable level (< 50 copies/mL).
- Preventing and treating opportunistic infections.
- Preventing HIV transmission to others (U=U: Undetectable = Untransmittable).
ART is evolving quickly with over 30 drugs available. Modern ART consists of a combination of three drugs from two different pharmacological classes to create a synergistic effect and prevent the virus from mutating into drug-resistant strains. Each class blocks a specific step of the life cycle:
- Entry / Fusion Inhibitors (Enfuvirtide): Blocks Step 2. Stops the viral envelope from fusing with the CD4 membrane.
- CCR5 Co-receptor Antagonists (Maraviroc): Blocks Step 1. Plugs the host's CCR5 receptor so gp120 cannot dock.
- NRTIs & NNRTIs: Blocks Step 3. Inhibits Reverse Transcriptase so viral RNA cannot become DNA. (NRTIs act as broken building blocks causing DNA chain termination).
- Examples (NRTIs): Tenofovir, Emtricitabine, Zidovudine, Lamivudine, Abacavir. (Truvada® = Tenofovir + Emtricitabine).
- Examples (NNRTIs): Efavirenz, Nevirapine, Etravirine.
- INSTIs (Integrase Strand Transfer Inhibitors): Blocks Step 4. Prevents viral DNA from splicing into host DNA.
- Examples: Raltegravir, Dolutegravir.
- Protease Inhibitors (PIs): Blocks Step 7. Prevents the new virus from maturing, leaving it non-infectious.
- Examples: Darunavir, Ritonavir, Atazanavir, Saquinavir.
Examples of ART Combinations:
- Preferred Combinations:
- HIV-PI Based: Darunavir + Ritonavir + Tenofovir + Emtricitabine.
- INSTI Based: Raltegravir + Tenofovir + Emtricitabine.
- Alternative Combinations:
- NNRTI Based: Efavirenz + Tenofovir + Emtricitabine.
- Alternative PI Based: Atazanavir + Ritonavir + Tenofovir + Emtricitabine.
When to Initiate ART:
- DHHS 2017 Guidelines (And Current Standard): ART is recommended for ALL individuals with HIV, regardless of CD4 cell counts, to reduce morbidity, mortality, and comprehensively prevent transmission to partners!
- Older / Specific Priorities (WHO Guidelines): Historically, initiated as a priority if CD4 ≤ 500 cells/mm³. High priority for: Severe/advanced HIV (WHO stage 3 or 4), CD4 ≤ 350, Active TB disease, HBV coinfection with severe liver disease, Pregnant/breastfeeding women, HIV-positive individuals in serodiscordant partnerships, and ALL Infants < 1 year old (regardless of stage/CD4).
Common Side Effects of ART (Drug Toxicity):
- NRTIs: Bone-marrow suppression (anemia), myopathy, peripheral neuropathy, diarrhea, life-threatening lactic acidosis, pancreatitis.
- NNRTIs: Severe skin rash, central nervous system effects (dizziness, vivid nightmares with Efavirenz), teratogenicity (Efavirenz causes birth defects in the first trimester), hepatic enzyme induction.
- Protease Inhibitors (PIs): Nephrolithiasis (kidney stones), massive diarrhea, elevated triglycerides, insulin resistance, and fat redistribution (lipodystrophy/buffalo hump).
- Integrase Inhibitors: Mild hepatotoxicity, insomnia, weight gain.
- Fusion Inhibitors: Injection site reactions, headache, increased risk of bacterial pneumonia.
XIV. Management of HIV in Pregnancy & PMTCT
A pregnant woman living with HIV can pass the virus to her baby in-utero, during childbirth, and through breastfeeding (Mother-To-Child-Transmission / MTCT). However, taking treatment correctly can virtually eliminate this risk down to less than 1%.
- Pregnancy Rules: HIV-infected women should be treated regardless of pregnancy status. ART is NOT contraindicated in pregnancy. Safety, efficacy, and pharmacokinetic data must be carefully considered (e.g., avoiding Efavirenz in the first trimester due to teratogenicity risk).
- Preferred Pregnancy ART Combination: Two NRTIs (Tenofovir + Emtricitabine) + Two PIs (Darunavir + Ritonavir).
Breastfeeding Guidelines:
Breast milk contains a high viral load of HIV. Guidelines vary globally purely based on available resources and clean water supply.
- If formula is always accessible and safe (Developed Nations): You should NOT breastfeed. Give formula exclusively instead to completely eliminate transmission risk.
- If formula is NOT accessible (Developing Nations): You are advised to breastfeed while BOTH mother and baby take ART. You must exclusively breastfeed for at least 6 months. Mixing breast milk and other foods (mixed feeding) before 6 months damages the infant's delicate gut lining, heavily increasing the baby's risk of contracting HIV!
Testing the Baby:
- The baby should be tested for HIV (using early PCR, not antibody ELISA) at birth and again 4 to 6 weeks later.
- If negative, test again at 18 months and/or when breastfeeding is finished to determine final status.
- If ANY of these tests come back positive, the baby must start pediatric ART treatment straight away.
Study Evidence (Malawi & Mozambique):
A retrospective cohort of 3,273 HIV+ women received triple antiviral therapy during pregnancy until 6 months postpartum. Result: Regardless of maternal CD4 count, ART provided a massive protective effect against mortality, fetal demise, and premature birth compared to historical controls.
XV. Broad HIV Prevention Strategies
There is NO vaccination for preventing HIV. (Initial hopes were dashed, though a Thai study using 4 priming injections of canarypox vector vaccine [ALVAC-HIV] plus 2 booster shots [AIDSVAX B/E] in 16,402 participants showed a modest 31.2% efficacy, especially in those who maintained lower-risk sexual behavior. However, this is not clinically viable for global rollout).
- Prevent Sexual Transmission:
- Abstinence and marital fidelity are the absolute best ways to prevent transmission.
- Ensure pre-marital HIV testing (ensure mutual non-infected marriage).
- Condoms (Male & Female): A critical element. Without a prescription, if used consistently and correctly, they reduce transmission risk by up to 96% (approx 80% on average).
- Voluntary Medical Male Circumcision (VMMC):
- One of the most powerful, cost-effective prevention tools. Studies (2006) proved it reduces a man's risk of acquiring HIV from a female partner by up to 60% in high-risk areas like Sub-Saharan Africa. The inner foreskin is highly susceptible to HIV due to dense target cells.
- PrePex Device: A newly available non-surgical circumcision kit. Prequalified by WHO/UNAIDS in 2007. It claims: No injected anesthesia, No surgery, No sutures, and No sterile settings required. (Laser circumcision is also a modern new method).
- Blood-Borne & Healthcare Prevention:
- Avoid IV drug abuse and never share needles/syringes. Ensure proper NAT testing of blood products and organs.
- Standard Precautions (Healthcare Workers): Wash hands, wear protective barriers (gloves, mask, eye shield, gown). Consider ALL body fluids contaminated. DO NOT recap needles. Clean blood spills immediately using germicidal solution (1:10 concentration of household bleach is highly effective).
- Chemical Prophylaxis:
- Postexposure Prophylaxis (PEP): Taken after an accidental exposure (e.g., needlestick, rape).
- Basic 2-drug regimen: Zidovudine + Lamivudine, OR Tenofovir + Emtricitabine.
- Expanded regimen: Basic PEP + Lopinavir-ritonavir (for high-risk exposures).
- Pre-exposure Prophylaxis (PrEP): Taken before exposure.
- Indicated for: Non-HIV-infected people at high risk: those in serodiscordant relationships, those who don't consistently use condoms with high-risk partners, or those who have injected illicit drugs/shared equipment in the last 6 months.
- Regimen: Daily oral fixed-dose combination of Tenofovir (300mg) and Emtricitabine (200mg) known as Truvada.
- Postexposure Prophylaxis (PEP): Taken after an accidental exposure (e.g., needlestick, rape).
- Non-Pharmacological Treatment (For the Patient):
- Patient education, providing social support, regular exercising, adequate rest, avoiding alcohol, smoking cessation, and eating a healthy nutritious diet to boost baseline immunity.
❓ Applied Clinical Question: Prevention Protocols
Case: A nurse accidentally sticks herself with a hollow-bore needle after drawing blood from a known HIV-positive patient with a high viral load. What is the immediate pharmacological protocol?
Answer: She requires immediate Expanded Postexposure Prophylaxis (PEP). Because it is a high-risk exposure (deep wound with a hollow needle containing fresh blood, high patient viral load), she should be started on a robust 3-drug regimen (e.g., Tenofovir + Emtricitabine + Lopinavir/ritonavir) as soon as possible, ideally within 1-2 hours, to prevent the virus from establishing integration into her CD4 cells.
Part II: Uganda Consolidated HIV Guidelines (2022) - Clinical Application
6.1 The Goal of Antiretroviral Therapy (ART)
The primary aim of antiretroviral therapy in Uganda is not to cure, but to achieve complete virological control. The specific goals are to:
- Suppress viral load levels amongst People Living with HIV (PLHIV) to strictly undetectable levels.
- Reduce the risk of morbidity and mortality associated with HIV (preventing opportunistic infections).
- Reduce the transmission of HIV (Undetectable = Untransmittable).
6.2 Composition of ART
Standard ART requires a synergistic pharmacological approach to prevent the virus from mutating and developing resistance.
- The Triple Therapy Rule: Standard ART consists of a combination of at least 3 antiretroviral (ARV) drugs to maximally suppress the HIV and stop the spread of HIV/AIDS disease.
- The "Backbone": Usually comprises 2 Nucleoside Reverse Transcriptase Inhibitors (NRTIs).
- Pharmacology Expansion: NRTIs act as "faulty building blocks." When the viral Reverse Transcriptase enzyme tries to build viral DNA, it grabs the NRTI, which lacks a 3'-OH group on its chemical structure, causing immediate, irreversible DNA chain termination.
- The "Anchor": A 3rd ARV drug from an entirely different pharmacological class to attack the virus from a second angle. This includes:
- Integrase Strand Transfer Inhibitors (INSTIs) - The preferred anchor in modern Uganda guidelines (e.g., Dolutegravir).
- Protease Inhibitors (PIs).
- Non-Nucleoside Reverse Transcriptase Inhibitors (NNRTIs).
6.3 When to Start ART
- The "Treat All" Policy: ART should be initiated at the earliest opportunity in all people with confirmed HIV infection, regardless of their clinical stage or CD4 cell count.
- Rationale: Since 2013, massive global evidence and programmatic experience have continued to favor early initiation of ART because it directly results in drastically reduced mortality, morbidity, and significantly limits HIV transmission outcomes in the community by slashing the viral load of the population.
💡 Clinical Exception: When do we DEFER starting ART?
While the goal is to "Treat All" immediately, starting ART the same day is exceptionally dangerous if the patient has a severe, active central nervous system or systemic infection (especially TB or fungal meningitis). Why?
Because rapidly restoring the immune system with ART will cause the newly awakened white blood cells to aggressively attack the infection all at once, causing massive, fatal inflammation known as Immune Reconstitution Inflammatory Syndrome (IRIS). In the brain, this sudden inflammation causes deadly swelling.
6.4 The Process of Starting ART
Although the Uganda program recommends starting all PLHIV on ART, health workers must systematically follow these specific steps to ensure safety:
- Assess for Advanced Disease & Opportunistic Infections (OIs):
- Use the Symptom Screen for Advanced Disease Pathway.
- Look for any evidence of OIs, especially Tuberculosis (TB) and Cryptococcal Meningitis.
- If the patient has TB or cryptococcal meningitis, ART MUST be deferred. It should be initiated only after starting targeted treatment for these specific OIs.
- Note: Treatment for other minor OIs (like oral thrush) and ART can be initiated concurrently without delay.
- The Opt-Out Approach (Same-Day Initiation):
- For patients without TB or cryptococcal meningitis, offer ART on the same day through an opt-out approach.
- Patients should be comprehensively prepared for ART on the same day and assessed for psychological and social readiness using the readiness checklist.
- If a client is ready, ART should be initiated immediately on the same day.
- Delayed Initiation Protocol:
- If a client is not ready or opts out of same-day initiation, a timely ART preparation plan must be agreed upon.
- Target timelines for delayed initiation: Within 7 days for children and pregnant women, and within 1 month for adults.
- Post-Trial Access (For Research Institutions):
- For institutions starting patients on experimental/new ARTs for research purposes, there must be a clear post-ART access plan for drugs that are approved but not yet accessible in standard public facilities.
- Research institutions must collaboratively work with partners to ensure continuous post-trial access to efficacious/safe study regimens for enrolled clients until the drugs become available through the national supply chain.
- If patients can afford prescribed ART regimens that are not on the essential drugs list or accessible via the national system, the institution must facilitate access to options of these new generation drugs.
Remember "T.C." takes Time and Care before starting ART!
- T - Tuberculosis (Treat TB first, wait 2-8 weeks before ART to prevent IRIS)
- C - Cryptococcal Meningitis (Treat with antifungals first, wait 4-6 weeks before ART to prevent fatal brain swelling)
6.5 First-Line ART Regimens (Treatment Optimization)
The first-line ART regimens for treating HIV infection in Uganda were selected based on universal optimization principles:
- Toxicity: Regimens with fewer side effects are preferred.
- Palatability and Pill Burden: Better taste (for children) and lower pill burden (e.g., fixed-dose combination, one pill a day) preferred to heavily boost patient adherence.
- Increased Durability and Efficacy: The drugs must work powerfully for a long time.
- Sequencing: The first-line choice must safely spare other available drug formulations so they can be used later as a 2nd-line regimen if the patient fails 1st-line therapy.
- Harmonization: Keeping regimens standard across different ages and populations to avoid supply chain confusion.
- Lower Cost: Essential for maintaining sustainable public health supply chains in Uganda.
Dolutegravir (DTG) - The Anchor of Choice:
DTG is an Integrase Inhibitor (INSTI). It is currently recommended for use as the anchor ARV in the preferred first, second, and third-line treatment regimens for all recipients of care (children, adolescents, men, women, including pregnant women, breastfeeding women, adolescent girls, and women of childbearing potential).
Rationale for Using Dolutegravir (DTG) in Uganda:
- High Circulating Levels of NNRTI Resistance: NNRTI-containing combinations (like Efavirenz or Nevirapine) have been used as first-line regimens in Uganda since 2005. There are massive growing concerns about transmitted drug resistance. A 2016/2017 study by the Uganda Virus Research Institute (UVRI) revealed extremely high levels of pre-treatment drug resistance (PDR) estimated at 15.9% to NNRTIs, far exceeding the 10.0% safety threshold set by the WHO!
- Superior Efficacy over Standard of Care: DTG is vastly superior to alternative options. Patients experience rapid viral suppression, heavily reducing the risk of transmitting HIV while prolonging the time they can safely stay on first-line treatment. Patients on DTG achieve viral suppression much faster compared to those on Efavirenz (EFV).
- Better Tolerability: DTG shows improved tolerability with substantial reductions in treatment-limiting adverse drug reactions. Specifically, patients avoid the severe psychiatric adverse events associated with EFV (e.g., severe depression, vivid nightmares, and suicidal tendencies). Less toxicity equals fewer patients stopping their meds.
- Higher Genetic Barrier to Resistance: The virus has a very hard time mutating against DTG. This high genetic barrier means patients are far less likely to develop resistance even with minor adherence lapses, postponing the need for expensive and complex second-line treatments.
❓ Applied Pharmacology Question
Case: A newly diagnosed HIV patient in Kampala asks why they are being given Dolutegravir (an Integrase Inhibitor) instead of Efavirenz (an NNRTI), which their friend was given 10 years ago. How do you explain the epidemiological reason to them?
Answer: You explain that because Efavirenz has been used in Uganda for so long, the virus has mutated in the general population. The UVRI found that nearly 16% of new patients have a virus that is already immune to Efavirenz before they even take their first pill! Dolutegravir is stronger, safer, and the virus in the community is not currently resistant to it, ensuring their treatment will work successfully.
6.6 Screening for Risk Factors Prior to Initiating DTG
While DTG is very well-tolerated, it has one major metabolic side effect: Hyperglycemia (High Blood Sugar) and weight gain. This has been reported among previously non-diabetic adults, and it heavily worsens hyperglycemia and insulin resistance among existing diabetics. (This occurs more commonly in clients transitioning to DTG from other regimens than in newly initiated clients).
Adults being initiated on DTG MUST be screened for these 3 Hyperglycemia Risk Factors:
- Age ≥ 40 years
- BMI ≥ 24 kg/m² (Overweight/Obese)
- History of hypertension
The DTG Initiation Algorithm based on Risk Factors:
- 1. Known Diabetics: Should NOT be initiated or transitioned to DTG. Give them an EFV400 or an ATV/r-based regimen instead.
- 2. Patients with 2 or more risk factors AND a High Baseline RBS/FBS: Should NOT be initiated/transitioned to DTG. Give an EFV400 or ATZ/r-based regimen.
- 3. Patients with 2 or more risk factors BUT a Normal Baseline RBS/FBS: Initiate or transition to DTG, but closely monitor their Random Blood Sugar (RBS) or Fasting Blood Sugar (FBS) every 3 months for the first 6 months to ensure they are not developing diabetes.
6.6.1 Rationale for Using EFV400 (Efavirenz 400mg):
- When DTG is contraindicated (like in severe diabetics), EFV400 is the highly preferred alternative anchor.
- Studies have shown that a 400mg dose of Efavirenz is virologically non-inferior (works just as well) to the old 600mg dose, but has significantly fewer psychiatric adverse events (which was the major limiting factor of EFV use).
- Furthermore, EFV 400mg can be safely co-administered with Rifampicin-containing anti-TB treatment, maintaining effective plasma concentrations without dangerous drug-drug interactions.
6.7 - 6.11 First-Line Regimen Guidelines & Alternatives
A. Adults & Adolescents (Weighing ≥ 30kg)
- Preferred Regimen: Tenofovir (TDF) or Tenofovir Alafenamide (TAF) + Lamivudine (3TC) + Dolutegravir (DTG).
(Commonly known as TLD: TDF + 3TC + DTG in a single pill). - When to use EFV400: If ineligible for DTG (e.g., diabetics, weight under 50mg formulation limits, or needing concurrent TB treatment where doubling the DTG dose is not a viable option).
- When to use ATV/r (Atazanavir/ritonavir): Only if they are ineligible for BOTH DTG and EFV.
- When to use Abacavir (ABC) backbone: Used only if TDF is contraindicated. Contraindications for TDF include: Severe kidney disease (GFR below 60 ml/min) or adolescents below 30kg where TDF harms bone density.
B. Pregnant & Breastfeeding Women
- Newly Diagnosed Preferred: TDF (or TAF) + 3TC + DTG.
- Alternative: Use EFV400 only if DTG is contraindicated. Use ATV/r if both EFV and DTG are contraindicated.
- Managing women ALREADY on ART at 1st ANC/PNC Visit:
- If on TDF/TAF + 3TC + EFV (and VL is suppressed): Maintain the EFV until 6-9 months postpartum, then transition safely to DTG.
- If already on TDF/TAF + 3TC + DTG (and VL is suppressed): Maintain this regimen throughout.
- If on NVP, ABC, or AZT (and VL is suppressed): Maintain the same regimen during pregnancy to avoid disrupting stability, then switch to TLD at 6-9 months postpartum. Note: Carefully screen women on Abacavir (ABC) to see if they were originally put on it because of a kidney contraindication to Tenofovir before attempting to switch them to TLD!
C. Children (≥20Kg to <30Kg)
- Preferred Regimen: Abacavir (ABC) or TAF + 3TC + DTG.
- Rationale for ABC: Using ABC in first-line pediatric regimens safely spares AZT (Zidovudine) for use in 2nd-line therapy. Additionally, ABC+3TC+DTG can be given as a once-a-day dose, highly improving adherence in children compared to twice-daily syrups.
- Alternatives: If DTG is contraindicated, use LPV/r (Lopinavir/ritonavir tablets). If ABC is contraindicated, use AZT or TAF (TAF only if >6 years and ≥25kg).
D. Infants & Small Children (< 20Kg)
- Preferred Regimen: ABC + 3TC + DTG.
- Alternatives: If DTG is intolerant or unavailable in correct pediatric formulations, initiate on ABC + 3TC + LPV/r (Ritonavir-boosted Lopinavir).
- Formulation Note: LPV/r syrup, pellets, or tablets must be prescribed strictly on the individual child's ABILITY to correctly take them. As soon as a child can take pellets, stop the awful-tasting syrup. As soon as they can swallow tablets without chewing/crushing, stop the pellets.
- Transitioning: ALL PLHIV on Raltegravir should be immediately transitioned to DTG irrespective of their Viral Load status. Use AZT only if the child experiences a severe hypersensitivity reaction to Abacavir.
Table Summary: Recommended First-Line ARV Regimens (Uganda 2022)
| Patient Category | Preferred Regimens | Alternative Regimens |
|---|---|---|
| Adults & Adolescents (≥30Kg) | TAF+FTC+DTG or TDF+3TC+DTG |
If DTG Contraindicated: TDF+3TC+EFV400 If TDF Contraindicated: ABC+3TC+DTG If Both Contraindicated: ABC+3TC+EFV400 |
| Pregnant & Breastfeeding Women | TAF+FTC+DTG or TDF+3TC+DTG |
TDF+3TC+EFV400 If EFV/DTG Contraindicated: TDF+3TC+ATV/r |
| Children (≥20Kg to <30Kg) | ABC+3TC+DTG |
If DTG Contraindicated: ABC+3TC+LPV/r (tablets) If ABC Contraindicated: AZT+3TC+DTG |
| Children (<20Kg) | ABC+3TC+DTG |
If DTG unavailable: ABC+3TC+LPV/r (granules/syrup) If LPV/r intolerant: ABC+3TC+EFV (if >3 yrs and >10kg) |
XVII. References
- UNAIDS. (2017). Global HIV & AIDS statistics — Fact sheet. Joint United Nations Programme on HIV/AIDS.
- Centers for Disease Control and Prevention (CDC). (2016-2020). HIV Surveillance Report: Diagnoses of HIV Infection in the United States and Dependent Areas.
- Ministry of Health Uganda. (2022). Consolidated Guidelines for the Prevention and Treatment of HIV and AIDS in Uganda. Kampala, Uganda.
- World Health Organization (WHO). (2016). Consolidated guidelines on the use of antiretroviral drugs for treating and preventing HIV infection. Geneva: WHO.
- Uganda Virus Research Institute (UVRI). (2017). National HIV Drug Resistance Surveillance Report.
- Department of Health and Human Services (DHHS). (2017). Guidelines for the Use of Antiretroviral Agents in Adults and Adolescents with HIV.
Quick Quiz
HIV Quiz
Microbiology - mobile-friendly and focused practice.
Privacy: Your details are used only for quiz tracking and certificates.
HIV Quiz
Microbiology
Preparing questions...
Choose your answer and keep your streak alive.
Great effort.
Here is your quick performance summary.
