Table of Contents
ToggleHepatitis B Virus (HBV)
By the end of this exhaustive, step-by-step master guide, you will be deeply conversant with:
- The historical timeline and virological classification of all hepatotropic viruses.
- The unique structure and replication cycle of the Hepatitis B Virus (including cccDNA and reverse transcription).
- The precise mechanisms of pathogenesis, including immune-mediated damage and the development of Hepatocellular Carcinoma (HCC).
- The detailed interpretation of HBV Serological Markers (the key to clinical diagnosis).
- The exact phases of Chronic Hepatitis B and how they dictate treatment protocols.
- The WHO guidelines for antiviral pharmacology, special populations, and post-exposure prophylaxis.
I. History and Introduction to Hepatitis
Hepatitis fundamentally describes the inflammation of the liver. While clinical medicine focuses heavily on infectious viral causes, it is highly vital to remember that hepatitis is a multi-etiological condition (it has many different causes).
Etiologies of Hepatitis:
- Chemicals and Toxins: Chronic alcohol abuse, hepatotoxic drugs (e.g., a massive overdose of Paracetamol/Acetaminophen, or prolonged use of Statins), and industrial poisons.
- Autoimmune Diseases: Autoimmune Hepatitis (Type 1 and Type 2), where the body generates anti-smooth muscle antibodies or anti-liver-kidney microsomal antibodies that attack the liver.
- Metabolic Diseases: Wilson's Disease (toxic copper accumulation) or Hemochromatosis (toxic iron accumulation).
- Infectious Viruses: Viral infections account for more than half the cases of acute hepatitis globally. Viral hepatitis is a systemic infection affecting the liver predominately, caused by any one of a heterogeneous group of hepatotropic (liver-seeking) viruses.
Historical Timeline:
- It is an ancient disease first described in the 5th century B.C. by early physicians.
- The earliest recognized blood-borne outbreak of hepatitis occurred in Germany in 1883. It happened after hundreds of people received a smallpox vaccine that had been accidentally contaminated by infectious human lymph fluid.
- In 1947, medical researchers MacCalum and Bauer officially introduced the specific terms Hepatitis A (originally termed "infectious hepatitis" because it spread through food/water) and Hepatitis B (originally termed "serum hepatitis" because it spread via blood transfusions).
- This clinical terminology was officially adopted by the World Health Organization (WHO) in 1973.
The Hepatotropic Viruses (Discovery Dates):
The alphabet of hepatitis viruses represents completely different viral families that simply share the liver as their primary target.
- Hepatitis A (HAV): Discovered in 1973.
- Hepatitis B (HBV): Discovered in 1970.
- Hepatitis C (HCV): Discovered in 1988 (previously known merely as "Non-A, Non-B Hepatitis").
- Hepatitis D (HDV): Discovered in 1977.
- Hepatitis E (HEV): Discovered in 1983.
- Hepatitis F: Not a separate entity! It was discovered to simply be a mutant strain of the B Virus.
- Hepatitis G (HGV): Discovered in 1995.
RNA vs. DNA Viruses
"All Hepatitis viruses are RNA, except for B!"
Hepatitis A, C, D, E, and G are all RNA viruses (relying on RNA polymerases). Hepatitis B is the ONLY DNA virus in this group, belonging exclusively to the Hepadnaviridae family.
Transmission Rule of Thumb: Vowels for the Bowels! Hepatitis A and E are transmitted via the fecal-oral route (contaminated food and water passing through the bowels). Hepatitis B, C, and D are blood-borne pathogens (transmitted via Blood, Birth, and sexual Copulation).
II. Clinical Classification and Global Burden
Clinical Classification:
- Acute Hepatitis: A sudden, highly symptomatic, but self-limited liver injury lasting less than 6 months. The immune system typically mounts a massive response and clears the virus.
- Chronic Hepatitis: Hepatic inflammation persisting for more than 6 months without viral clearance. The virus has successfully evaded the immune system and settled into the liver permanently.
Epidemiology & Prevalence:
Hepatitis B is a serious, highly infectious disease affecting millions worldwide, posing a massive public health crisis.
- Global Stats: More than 2,000 million (2 billion) people alive today have serological evidence of past or present infection with HBV.
- Carriers: Of these, about 240 to 350 million individuals fail to clear the virus, remaining infected chronically. They become permanent "carriers" of the virus, capable of spreading it to others.
- Mortality: Every year, there are over 4 million acute clinical cases of HBV. Roughly 25% of all chronic carriers (which translates to over 1 million people a year) will eventually die from the severe pathological consequences of chronic active hepatitis, cirrhosis (scarring), or primary liver cancer (Hepatocellular Carcinoma - HCC).
Geographic Distribution of Chronic HBV:
The world is divided into three distinct epidemiological areas based on the prevalence of chronic hepatitis infection:
| Endemicity Level | Prevalence | Regions & Characteristics |
|---|---|---|
| High | >8% | South-East Asia, Pacific Basin, sub-Saharan Africa, parts of the Middle East, some Eastern European countries. In these areas, 70-90% of the population becomes infected before age 40, usually due to high rates of mother-to-child transmission at birth. 8-20% become lifelong chronic carriers. |
| Intermediate | 2-8% | The rest of the world falls into this range. Transmission is a mix of vertical (birth) and horizontal (childhood play, unsterile medical practices). |
| Low | <2% | North America, Western/Northern Europe, Australia. Less than 20% of the population is ever infected. The carrier rate is very low (about 2%). Infections here usually occur in high-risk adult populations (IV drug users, unprotected sex). |
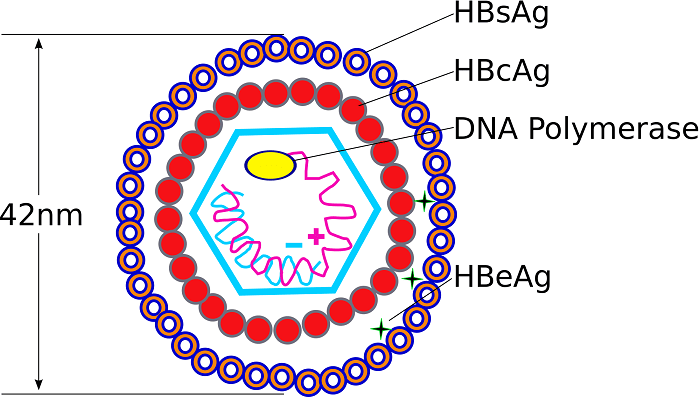
III. Structure of the Hepatitis B Virus (HBV)
HBV is a remarkably compact, highly complex virus classified within the family Hepadnaviridae (Hepa = liver, dna = DNA virus).
The Genome:
- It possesses a small, circular, partially double-stranded DNA genome.
- It is incredibly efficient, measuring only 3,200 base pairs in size (one of the smallest known viral genomes).
- It utilizes overlapping reading frames to code for exactly four sets of viral products: Surface proteins (S), Core proteins (C), Polymerase (P), and the X protein (X).
The Virion (The Dane Particle):
The complete, fully mature, infectious HBV virion is 42 nm in diameter. Under an electron microscope, it looks like a sphere. It is officially referred to as the Dane particle (named after the scientist who first identified it).
- Nucleocapsid Core (27 nm): The inner protective shell of the virus. It securely contains the viral DNA, the highly vital viral DNA polymerase (reverse transcriptase), protein kinase, and the Core antigen (HBcAg).
- Envelope (Outer Coat): An outer lipo-protein coat that surrounds the core, derived from the host cell's endoplasmic reticulum. It contains the highly immunogenic Surface antigen (HBsAg), which the virus uses to dock onto healthy liver cells.
The Three Key Antigens (Crucial for Diagnosis):
1. HBsAg (Surface Antigen): The surface (coat) protein. This is the primary serological marker of infection. If it is in the blood, the virus is currently in the body (whether acute or chronic).
2. HBcAg (Core Antigen): The inner core protein. It remains trapped inside the hepatocyte (liver cell) and the intact virion. It is not typically found free-floating in the blood serum.
3. HBeAg (e-Antigen): A secreted protein. Virology Expansion: This is a truncated (cut-short) version of the core protein that is actively pushed out of the cell. Its presence in the blood indicates incredibly high active viral replication and extreme infectivity.
Antigenic Decoy Particles
During the intense phase of HBV replication, the virus produces massive, overwhelming excess amounts of the surface protein (HBsAg). These excess proteins clump together to form spherical and elongated (tubular) particles measuring about 22nm in the blood.
The Catch: These particles are completely empty! They contain no viral core and absolutely no DNA. Therefore, they cannot cause infection.
Why does the virus do this? They act as immunological Decoys! By flooding the bloodstream with billions of empty ghost shells, the virus effectively soaks up and distracts all of the host body's neutralizing antibodies. The immune system attacks the empty shells, allowing the real, fully infectious Dane particles to sneak by unnoticed and infect other healthy liver cells.
IV. Replication Cycle of Hepatitis B Virus
Although HBV is classified as a DNA virus, it behaves completely uniquely because its replication strictly occurs via an RNA intermediate, utilizing the enzyme Reverse Transcriptase (very similar to the replication cycle of HIV).
- Attachment & Entry: The complete virion (Dane particle) utilizes its HBsAg envelope to attach to a highly specific receptor (NTCP) on the surface of the human hepatocyte (liver cell) membrane. The virus is then pulled inside via endocytosis.
- Nuclear Transport & Repair (cccDNA formation):
- The viral envelope is stripped away, and the bare nucleocapsid delivers its partially double-stranded DNA deep into the host cell's nucleus.
- Inside the nucleus, the host cell's own repair enzymes mistakenly "fix" the partial viral DNA strand, converting it into a perfectly seamless, covalently closed circular DNA known as cccDNA.
- Deep Physiology Expansion: This cccDNA acts as a highly stable "mini-chromosome." It physically anchors itself inside the nucleus and hides there permanently. This is exactly why chronic Hepatitis B is so incredibly difficult to completely "cure" with current antiviral drugs—drugs can stop the virus from multiplying, but they cannot reach inside the nucleus to destroy the hidden cccDNA templates!
- Transcription: The host cell's RNA polymerase binds to the cccDNA and reads it, producing several viral messenger RNAs (mRNAs). The most important of these is a massive RNA strand called the pre-genomic RNA (pgRNA).
- Translation: The mRNAs exit the nucleus and enter the cytoplasm, where the host's ribosomes are hijacked to translate them into viral proteins: Surface proteins (HBsAg), Core proteins (HBcAg), e-antigen (HBeAg), and the viral DNA Polymerase enzyme.
- Reverse Transcription (The Unique Step!):
- The massive pre-genomic RNA (pgRNA) is packaged inside a newly forming shell made of core proteins.
- Inside this secure core, the newly manufactured viral Reverse Transcriptase reads the RNA strand and synthesizes a brand new, partially double-stranded viral DNA genome! (Translating RNA backwards into DNA).
- Assembly & Release:
- The newly minted DNA core travels to the endoplasmic reticulum, where it steals a piece of membrane packed with HBsAg to serve as its new envelope.
- The cell then assembles these "live" copies of the virus and releases them via exocytosis into the bloodstream to infect other liver cells.
- Simultaneously, the massive excess of empty surface proteins (HBsAg) is released as the spherical and elongated antigenic decoy particles.
- Viral Integration (Carrier State & Cancer Risk): During this chaotic replication, some of the HBV DNA accidentally integrates directly into the host hepatocyte's native human genome. While not necessary for viral survival, this accidental integration causes the chronic carrier state and is the primary molecular trigger that causes cellular mutations, eventually leading to Hepatocellular Carcinoma (HCC).
V. Pathogenesis & "Ground Glass" Hepatocytes
1. Immune-Mediated Damage:
A crucial fact in hepatology is that HBV is generally not directly cytopathic. This means the virus does not kill the liver cell simply by replicating inside it. It merely turns the cell into a quiet virus factory.
However, as the cell produces viral proteins, it displays fragments of these proteins on its surface (via MHC Class I molecules). The host's own immune system (specifically CD8+ Cytotoxic T-cells) recognizes these foreign proteins and mounts a violent attack. The severe pathological damage, the inflammation (hepatitis), the cell necrosis, and the resulting clinical pain are actually caused entirely by your own immune system indiscriminately destroying your liver to eradicate the virus hiding inside!
2. Ground Glass Appearance:
Because of the massive, unbridled production of excess surface proteins (HBsAg), these lipo-proteins accumulate and stick together inside the cytoplasm (specifically swelling the Endoplasmic Reticulum). If a pathologist looks at an infected liver biopsy under a microscope using an H&E stain, this protein accumulation gives the cytoplasm of the infected liver cell a characteristic, finely granular, hazy "ground glass" appearance.
3. Cancer Risk (Oncogenesis):
Because the liver is subjected to constant chronic inflammation, continuous cell death, and rapid, desperate cellular regeneration, mistakes happen in the DNA. Combine this rapid cell turnover with the fact that viral DNA integrates directly into the host genome (often disrupting tumor suppressor genes like p53), HBV becomes a highly oncogenic virus. It is the direct cause of up to 80% of all cases of Hepatocellular Carcinoma (HCC) worldwide.
Antiviral Targets
Case: A patient with highly active chronic Hepatitis B is started on a daily oral medication called Tenofovir. Given the unique replication cycle of the Hepatitis B virus, what specific viral enzyme is this drug most likely targeting to stop the virus from multiplying?
Answer: Reverse Transcriptase (Viral DNA Polymerase). Since HBV must convert its pre-genomic RNA intermediate back into DNA to create new infectious virions, drugs like Tenofovir (which are Nucleotide Reverse Transcriptase Inhibitors) perfectly block this exact step. By causing chain termination, they halt viral replication instantly, protecting the remaining healthy liver cells.
VI. Modes of Transmission
HBV is notoriously highly infectious—it is estimated to be 100 times more infectious than the HIV virus. It can survive outside the body for at least 7 days and remain fully capable of causing infection. It is transmitted strictly via exposure to blood or mucous membranes containing infectious bodily fluids.
Concentration of Virus in Body Fluids:
- High Concentration: Blood, blood serum, and weeping wound exudates.
- Moderate Concentration: Semen, vaginal fluid, and saliva.
- Low / Not Detectable: Urine, feces, sweat, tears, and breast milk. (HBV cannot be spread by hugging, sneezing, coughing, or sharing food).
Transmission Routes & Risk Groups:
- Perinatal (Vertical) Transmission:
- Transmission directly from an infected mother to her infant during the trauma of childbirth (exposure to maternal blood in the birth canal).
- Mothers who are highly infectious (HBeAg positive) are up to 90% more likely to transmit the virus to their offspring compared to those who are HBeAg negative.
- This route represents the primary means of transmission in high-prevalence populations (e.g., Sub-Saharan Africa and rural Asia), heavily contributing to the massive global burden of chronic carriers.
- Parenteral / Percutaneous Transmission:
- Direct exposure of the bloodstream to infectious blood via contaminated sharps.
- Extremely high risk for Intravenous Drug Abusers (IVDA) sharing needles, healthcare workers (accidental needle-stick injuries in the ward), patients undergoing prolonged hemodialysis, individuals receiving unsanitary tattoos or piercings, and recipients of unscreened blood transfusions or organ transplants.
- Sexual Transmission:
- Exposure to semen or vaginal fluids during unprotected intercourse.
- High risk for commercial sex workers, men who have sex with men (MSM), and individuals with multiple, concurrent sexual partners.
- Horizontal Transmission (Person-to-Person):
- Can occur in settings involving close, continuous interpersonal contact over an extended period.
- High risk for household contacts of chronically infected individuals, and developmentally disabled persons living in crowded long-term care facilities.
- In children, this can easily occur through minor scratches during play, iatrogenic events, folk remedies (e.g., unsterile tribal scarification or acupuncture), or simply sharing blood-contaminated items like toothbrushes or razors.
VII. Clinical Presentation of Acute Hepatitis B
The incubation period for HBV is exceptionally long, ranging widely from 45 to 180 days (Average: 60-90 days before any symptoms appear). Once symptomatic, the clinical illness progresses sequentially through three distinct phases:
1. Preicteric Phase (Before Jaundice):
- Begins an average of 3 months after the initial infectious exposure.
- Characterized by generalized, non-specific, flu-like viral symptoms: overwhelming tiredness, severe fatigue, anorexia (total loss of appetite), nausea, vomiting, vague right upper quadrant abdominal pain (due to liver swelling stretching the liver capsule), and low-grade fever.
- Serum sickness-like illness: In 10-20% of patients, the massive amounts of HBsAg bind to the body's antibodies, forming circulating "immune complexes." These heavy complexes crash out of the bloodstream and deposit in joints and skin, causing severe arthralgias (joint pain) and hives/rash weeks before liver symptoms even appear.
2. Icteric Phase (Jaundice Phase):
- Typically begins within 10 days of the initial vague symptoms.
- Characterized by a striking yellowish discoloration of the mucous membranes, the sclera (white of the eyes), and eventually the skin, known clinically as Jaundice (or Icterus).
- Dark urine and Pale/Clay-colored stool: As the liver fails, conjugated bilirubin is blocked from entering the intestines (hence the stool loses its brown pigment and turns clay-colored). The excess, water-soluble conjugated bilirubin spills back into the blood and is filtered by the kidneys, turning the urine as dark as Coca-Cola.
- Laboratory tests show total serum bilirubin massively exceeding 20 to 40 mg/L.
- Physical examination by a physician frequently reveals Hepatosplenomegaly (a physically enlarged, highly tender liver and a swollen spleen).
3. Convalescent Phase (Recovery):
- Begins slowly after the disappearance of jaundice and the return of normal stool color.
- Symptoms like fatigue typically last for several weeks but can persist stubbornly for up to 6 months.
- Biochemically, it is characterized by the transient presence of HBsAg, HBeAg, and viral DNA, followed by the highly desired, successful seroconversion (the production of protective anti-HBs and anti-HBe antibodies).
The Age Factor
The clinical outcome of an HBV infection (whether you successfully clear it or become a chronically infected lifelong carrier) depends almost entirely on the age at which you are infected!
- Adults: Have a fully mature, aggressive immune system. Upon sensing the virus, the adult immune system attacks the liver violently. This causes severe, highly symptomatic acute hepatitis (severe jaundice, intense pain), but because the attack is so thorough, <5% of adults become chronic. Over 95% of adults clear the virus completely and gain lifelong immunity!
- Infants (Perinatal Infection): Have a highly immature immune system. The infant immune system completely fails to recognize the HBV virus as foreign (a phenomenon known as Immune Tolerance). Therefore, infants show absolutely zero acute symptoms when infected, but tragically, >90% of infected infants become chronically infected carriers for life, leading to cirrhosis and cancer decades later.
- Children (1-5 years): Fall in the middle; 25% to 50% will fail to clear the virus and develop chronic infection.
VIII. Markers of Infection & Diagnosis
Because HBV can hide silently in the body for decades without causing pain, accurate diagnosis relies on a comprehensive panel of four specific investigative domains:
1. Virological Markers (Measuring The Virus Itself)
- Viral Load (HBV DNA): Directly correlates with active viral replication. Quantification of serum HBV DNA (measured in IU/mL via PCR) is the absolute pivotal tool used by hepatologists to select candidates for antiviral therapy and to monitor whether the drug treatment is successfully working.
- HBV Genotypes: The virus is not entirely uniform. There are at least nine different genotypes globally (A through I), classified based on >8% genomic differences. Genotypes C and F are known to have a significantly higher rate of progression to Hepatocellular Carcinoma (HCC). Note: Antiviral therapy and standard vaccines remain equally effective against all genotypes.
- Mutant Viruses (High-Yield Clinical Challenge):
- Precore Mutants: A terrifying genetic mutation that completely abolishes the virus's ability to produce HBeAg. The virus still replicates wildly and destroys the liver, but standard blood tests for HBeAg come back negative. This is extremely dangerous because it fools inexperienced doctors into thinking the virus is inactive. Clinically known as "HBeAg-negative Chronic Hepatitis B."
- Core Mutants: Down-regulates, but does not abolish, HBeAg production.
- YMDD Mutant: A specific, highly targeted mutation in the active site of the viral DNA polymerase. This mutation develops specifically during long-term treatment, causing absolute drug resistance to the older antiviral drug Lamivudine.
2. Biochemical Markers (Liver Function Tests - LFTs)
- Liver Enzymes (ALT - Alanine Aminotransferase): The most critical cellular enzyme to look for. When liver cells die, they burst and spill ALT into the blood. Normal physiological limits are <30 U/L (men) and <19 U/L (women).
- Acute HBV: ALT is temporarily, but massively, elevated (often in the thousands).
- Chronic HBV: ALT fluctuates wildly. Consistently increased ALT indicates a severe, ongoing immune war, carrying a much higher risk of long-term permanent liver damage. Requires strict longitudinal monitoring.
- Other secondary markers include Serum Albumin (drops if the liver stops producing proteins), Prothrombin Time (PT - prolongs if the liver stops making clotting factors), and Serum Bilirubin.
3. Histological & Non-Invasive Markers (Assessing Liver Damage)
- Liver Biopsy: Invasive and painful. Involves driving a needle into the liver to extract tissue. Shows chronic hepatitis with necroinflammation (graded clinically by a Knodell score ≥ 4). Indicated today only when non-invasive tests are confusing or inconclusive.
- Transient Elastography (FibroScan®): A rapid, painless, non-invasive specialized ultrasound technique that sends a mechanical wave into the liver to evaluate "liver stiffness." A stiff liver heavily predicts advanced fibrosis or cirrhosis.
- Serum Marker Panels: Advanced algorithms like FibroTest® or the APRI (AST-to-Platelet Ratio Index) can accurately predict the exact stage of liver fibrosis based on simple blood draws, avoiding needles entirely.
4. Serological Markers (Antigens & Antibodies)
- HBsAg (Surface Antigen): The general marker of active infection. If it remains present in the blood for >6 months, the patient officially has Chronic Hepatitis B.
- Anti-HBs (Surface Antibody): Indicates clinical recovery and protective immunity. Crucial Note: It is the ONLY marker that will be positive in a person who is immune because of a VACCINE!
- HBcAg (Core Antigen): Found strictly locked inside the nuclei of infected liver cells; no free HBcAg is ever found floating in blood serum.
- Anti-HBc (Core Antibody):
- IgM type: The massive, fast-acting antibody. It is the definitive marker of a recent, Acute infection.
- IgG type: The long-term memory antibody. It is the marker of past recovery OR ongoing chronic infection.
- HBeAg (e-Antigen): Soluble antigen indicating highly active viral replication and extreme infectivity (the patient is highly contagious).
- Anti-HBe (e-Antibody): Indicates the host immune system has forced the virus to stop heavily replicating (except in precore mutants). It is a highly favorable marker of reduced infectivity.
IX. Interpretation of Serologic Test Results
This is arguably the most highly tested concept in all of hepatology. You must be able to read an HBV serology panel and diagnose the patient's exact clinical status flawlessly.
| HBsAg (Surface Ag) | Total Anti-HBc (Core Ab) | IgM Anti-HBc (Acute Core) | Anti-HBs (Surface Ab) | Clinical Interpretation / Diagnosis |
|---|---|---|---|---|
| - (Negative) | - (Negative) | - (Negative) | - (Negative) | Never Infected / Susceptible. This patient has zero markers and needs the vaccine immediately. |
| + (Positive) | - (Negative) | - (Negative) | - (Negative) | Early Acute Infection. Transient, occurs up to 18 days after initial exposure before the immune system has even reacted. |
| + (Positive) | + (Positive) | + (Positive) | - (Negative) | Acute Infection. The HBsAg proves the virus is there. The presence of the fast-acting IgM Core Ab absolutely proves the infection is recent and acute! |
| - (Negative) | + (Positive) | + (Positive) | + or - | Acute Resolving Infection (The Window Period). See detailed explanation below. |
| - (Negative) | + (Positive) | - (Negative) | + (Positive) | Recovered from Past Infection (Natural Immunity). The Surface Ab provides immunity. The presence of the Core Ab proves they actually fought off the real, wild virus in the past. |
| + (Positive) | + (Positive) | - (Negative) | - (Negative) | Chronic Infection. HBsAg is heavily positive, but the acute IgM is totally gone (replaced by IgG), meaning >6 months have passed and the patient failed to clear it. |
| - (Negative) | - (Negative) | - (Negative) | + (Positive) | Immune due to VACCINATION. The vaccine only contains isolated surface proteins. Therefore, only Surface Ab is produced. No Core Ab is present because the core of the virus was never injected! (Requires a titer of > 10 mIU/mL for protection). |
The "Window Period"
Look carefully at the "Acute Resolving Infection" row in the table above. Notice that HBsAg is officially NEGATIVE, and Anti-HBs is also NEGATIVE. How is this physiologically possible if the patient is currently in the middle of fighting off the virus?
The Answer: During the crucial acute recovery phase, the body begins massively producing protective Anti-HBs antibodies. However, as soon as they are produced, they immediately collide and bind tightly to the massive amounts of HBsAg antigens still floating in the blood, physically pulling them out of circulation. Because they are locked together in giant immune complexes, standard laboratory machines cannot detect either the free antigen OR the free antibody! During this confusing "Window Period," the ONLY positive marker in the blood that can prove the patient currently has Hepatitis B is the IgM Anti-HBc (Core Antibody)!
X. Natural History & Phases of Chronic Hepatitis B (CHB)
Chronic Hepatitis B is a highly dynamic and extraordinarily complex disease. It progresses non-linearly through several recognizable clinical phases. These phases are of highly variable duration, do not always happen in a neat sequence, and strictly dictate whether aggressive antiviral treatment is necessary.
- Serology: HBeAg Positive.
- Pattern: Insanely high levels of HBV DNA replication (often >200,000 IU/mL or even in the billions). However, ALT is persistently NORMAL, and biopsies show minimal histological liver disease.
- Mechanism: Seen heavily in children infected at birth. The immature immune system "tolerates" the virus, ignoring it completely. Because the T-cells do not attack the infected liver cells, there is no inflammation or cell death (hence the perfectly normal ALT).
- Treatment: Not generally indicated (but extremely close monitoring is required). Giving drugs here is useless because the immune system won't assist the drugs.
(HBeAg-Positive CHB)
- Serology: HBeAg Positive (but the body may slowly start developing anti-HBe).
- Pattern: High or wildly fluctuating HBV DNA (>2000 IU/mL). ALT is severely abnormal or intermittently elevated. Liver biopsy shows highly active necroinflammation.
- Mechanism: As the patient ages, the immune system finally "wakes up" and recognizes the massive viral threat. It starts actively slaughtering infected liver cells to kill the virus, causing intense inflammation.
- Treatment: May be heavily indicated here to assist the immune system and prevent permanent cirrhosis from the collateral damage.
("Immune Control")
- Serology: HBeAg turns Negative, Anti-HBe turns Positive.
- Pattern: Low or totally undetectable HBV DNA (<2000 IU/mL). Persistently normal ALT.
- Mechanism: The immune system definitively won the active battle! It forced the virus into deep hiding and stopped replication. The ongoing risk of developing cirrhosis and HCC is greatly reduced.
- Treatment: Not generally indicated (but continuous monitoring for sudden reactivation is absolutely required).
(HBeAg-Negative CHB)
- Serology: HBeAg Negative, Anti-HBe Positive.
- Pattern: Moderate to high HBV DNA (>20,000 IU/mL) returning, accompanied by heavily Abnormal ALT.
- Mechanism: The virus mutated under pressure! (This is the terrifying Precore Mutant). The mutated virus successfully evades immune control and begins aggressively destroying the liver again, but successfully hides its HBeAg marker from doctors. Older persons are especially at risk for rapid, progressive fibrosis and cirrhosis.
- Treatment: Highly indicated to prevent end-stage liver disease.
(Acute-on-Chronic)
- Serology: HBeAg Positive OR Negative.
- Pattern: Skyrocketing HBV DNA, massively Abnormal ALT. Seroreversion (HBeAg turns positive again) can unexpectedly occur.
- Mechanism: The sleeping virus violently wakes up. This occurs spontaneously or is precipitated directly by profound immunosuppression (e.g., undergoing Chemotherapy, acquiring HIV, receiving an organ transplant), severe antiviral drug resistance, or suddenly stopping antiviral therapy without a doctor's order. It carries an insanely high risk of deadly liver decompensation and outright failure.
- Treatment: Indicated immediately as a life-saving measure.
XI. Disease Progression & Complications
- Acute Outcomes: Approximately 90% of adults resolve completely, roughly 9% become chronic (HBsAg+ > 6 months), and roughly 1% develop Fulminant Hepatitis (a devastating, massive, lightning-fast necrosis of the entire liver substance, leading to hepatic encephalopathy, coma, and usually death within days without an emergency liver transplant).
- Chronic Outcomes: 15-40% of all chronic patients will eventually progress relentlessly to End-Stage Liver Failure, Cirrhosis, or Liver Cancer over several decades.
Hepatocellular Carcinoma (HCC):
- HBV is a proven, highly dangerous oncogenic (cancer-causing) virus! It integrates its DNA directly into the host genome via insertional mutagenesis, often breaking the cell's natural tumor suppressors.
- While only 5% of patients with generalized alcoholic cirrhosis develop HCC, HBV is directly responsible for a staggering 90% of all primary malignant tumors of the liver globally.
- It represents the 7th most common cancer in males globally and 9th in females, causing >500,000 deaths annually. It typically appears after a mean duration of about 35 years of chronic, silent infection.
Extra-Hepatic Manifestations (Immune-Complex Mediated):
Because of the massive amounts of virus and antibodies floating in the blood, they form clumps that get stuck in tiny blood vessels, causing damage far outside the liver:
- Polyarteritis Nodosum: Severe, necrotizing inflammation of medium-sized blood vessels leading to ischemia.
- Glomerulonephritis: The immune complexes get stuck in the kidney filters, causing severe kidney damage and protein in the urine.
- Papular acrodermatitis: Also known as Gianotti-Crosti syndrome, a distinct, blistering rash frequently seen on the limbs and face of infected children.
Management of Hepatitis B: Diagnosis, Pharmacology, Guidelines & Prevention
XII. Diagnostic Criteria & Initial Evaluation
Before initiating any potentially lifelong treatment, a patient with Chronic Hepatitis B (CHB) must be meticulously and thoroughly evaluated to determine their exact disease phase and the physical extent of their liver damage.
Diagnostic Criteria for CHB:
- HBsAg Positive consistently for > 6 months.
- Elevated Serum HBV DNA (quantifiable via PCR).
- Liver Enzymes: Persistent or intermittent elevation of ALT > 2x the Upper Limit of Normal (ULN) for 3 to 6 consecutive months.
- Liver Biopsy: Showing definitive chronic hepatitis with moderate or severe necroinflammation (Knodell score ≥ 4).
Initial Evaluation of Patients:
- Comprehensive History and Physical Examination (looking for signs of advanced disease like spider angiomas or ascites).
- Detailed Family History of liver disease or Hepatocellular Carcinoma (HCC).
- Comprehensive laboratory tests to assess baseline liver function (Albumin, Bilirubin, Prothrombin time).
- Specific tests for HBV replication status: HBeAg, anti-HBe, and heavily quantifying the exact HBV DNA load.
- Co-infection Screening (Mandatory): Must definitively rule out other viral co-infections that complicate treatment—anti-HCV, anti-HDV, and anti-HIV in those at risk.
- HCC Screening: Baseline Abdominal Ultrasound (USG) and Alpha-Fetoprotein (AFP) tumor marker levels, especially in older, high-risk patients.
- Consider a liver biopsy to precisely grade and stage liver disease for patients who meet the criteria for chronic hepatitis but have confusing blood panels.
XIII. Antiviral Pharmacology: Treatment Options
The primary clinical goals of CHB treatment are to aggressively suppress HBV replication to totally undetectable levels, decrease hepatic necroinflammation and fibrosis, and definitively prevent progression to cirrhosis, liver failure, and HCC. Currently, there are seven major antiviral agents approved globally.
| Class of Drug | Specific Drugs & Dosing | Mechanism of Action | Key Advantages & Disadvantages |
|---|---|---|---|
| 1. Immunomodulators (Injectables) | Interferon alpha-2b (IFN) and Pegylated-interferon alpha-2a/2b (PEG-IFN). Dose: PEG-IFN is 180 µg once per week (subcutaneous injection). |
They do NOT kill the virus directly. They act as massive immune-system boosters. They enhance the phagocytic activity of macrophages, inhibit viral replication inside already virus-infected cells, and dramatically increase the aggressive cytotoxicity of T-lymphocytes against the infected liver cells. | Advantages: Finite treatment duration (usually 48 weeks), zero risk of viral resistance, and higher rates of achieving actual HBeAg and HBsAg loss (a true "cure"). Disadvantages: Highly toxic. Less than 50% actually respond, very high cost, requires painful injections, and causes severe, debilitating side effects (flu-like symptoms, bone marrow suppression, suicidal depression, alopecia/hair loss). |
| 2. Nucleoside/Nucleotide Analogues (Oral NAs) | Tenofovir (TDF), Entecavir (ETV), Lamivudine (3TC), Adefovir (ADV), Telbivudine (LdT). Doses: Tenofovir (300 mg once daily), Entecavir (0.5 mg daily; 1.0 mg if decompensated). |
These are highly advanced "fake DNA building blocks." They act by specifically inhibiting the HBV DNA polymerase (Reverse Transcriptase) enzyme. When the virus tries to build new DNA, it accidentally inserts the drug instead of a real nucleotide, causing instant chain termination and a massive, rapid decrease in viral replication. | Advantages: Extremely simple one-pill-a-day oral administration with very few side effects. Highly effective at dropping viral load fast. Disadvantages: The major limitation is the development of viral resistance (the virus mutates to avoid the drug), requiring strict lifelong, uninterrupted therapy in most cases to keep the virus suppressed. |
Absolute Contraindications for Interferon (IFN) Therapy
Because IFN causes massive immune system stimulation, it is absolutely contraindicated and extremely dangerous in patients with: Decompensated cirrhosis (it will push the failing liver over the edge), hypersplenism, uncontrolled thyroid disease, active autoimmune diseases (it will worsen the autoimmune attack), severe coronary artery disease, organ transplants (it will cause the body to reject the transplant!), pregnancy, severe psychiatric illness (due to profound depression side effects), and infants < 1 year of age.
XIV. WHO Treatment Guidelines (Updated Benchmarks)
A. First-Line Therapies & Managing Resistance
- The WHO strongly, universally recommends Tenofovir (TDF) or Entecavir (ETV) as absolute first-line therapy because both possess an exceptionally high genetic barrier to drug resistance (the virus rarely mutates to beat them).
- Entecavir is specifically highlighted and recommended for use in children aged 2–11 years.
- Older NAs with a notoriously low barrier to resistance (Lamivudine, Adefovir, Telbivudine) predictably lead to rapid drug resistance and are no longer recommended as first-line options.
- Rescue Therapy: If a patient previously on old drugs develops profound resistance to Lamivudine (developing the notorious YMDD mutation), the required treatment adaptation is to immediately switch the patient to Tenofovir (TDF), which remains effective against the mutant.
B. Who TO Treat (Treatment Priority)
- Priority 1 (Mandatory): All adults, adolescents, and children with CHB who show clinical evidence of cirrhosis (or an APRI score > 2 in adults) MUST be treated immediately, completely regardless of what their ALT levels, HBeAg status, or HBV DNA levels are! The liver is scarred and needs immediate protection.
- Priority 2: Adults with CHB who do NOT yet have cirrhosis, but are > 30 years old, and demonstrate persistently abnormal ALT levels AND highly active viral replication (HBV DNA > 20,000 IU/mL).
C. Who NOT To Treat (But continue to aggressively monitor)
- Antiviral therapy is strictly deferred in persons without cirrhosis (APRI ≤ 2) who have persistently normal ALT and low/undetectable HBV DNA (< 2000 IU/mL). (This is the safe Immune Control phase).
- Also strongly defer treatment in young patients (≤ 30 years old) without cirrhosis who possess insanely high DNA (> 20,000) but maintain persistently normal ALT. (This is the classic Immune Tolerant phase! Giving drugs here is futile and wastes resources).
D. When to STOP Treatment
- Lifelong Therapy: Unconditionally required for all persons with established cirrhosis. They should never discontinue therapy because the inevitable viral reactivation rebound can cause severe acute-on-chronic liver injury, massive decompensation, and rapid death.
- Discontinuation: May be considered highly exceptionally in non-cirrhotics ONLY if there is strict serological evidence of HBeAg loss AND clear seroconversion to protective anti-HBe, followed by at least one full additional year of "consolidation" treatment.
- If stopped, relapse may fiercely occur. Retreatment is immediately recommended if HBsAg/HBeAg becomes positive again, ALT begins to rise, or DNA becomes heavily detectable again.
E. Strict Monitoring Guidelines
- Disease Progression: Monitor ALT, HBsAg, HBeAg, HBV DNA, and non-invasive fibrosis tests (APRI/FibroScan) at least annually. If the patient is untreated and blood values are fluctuating wildly, monitor tightly every 3 months.
- Toxicity Monitoring: The premier drugs TDF and ETV are cleared exclusively by the kidneys. Therefore, baseline renal function (creatinine) must be assessed before starting. Renal function must be monitored annually (due to the risk of insidious nephrotoxicity), and bone growth must be monitored carefully in children. Mandatory dose reductions and adjustments are required for patients with a creatinine clearance < 50 mL/min.
- HCC Surveillance (Cancer Checks): Routine, lifelong surveillance utilizing high-resolution Abdominal Ultrasound and Alpha-Fetoprotein (AFP) blood testing every 6 months is heavily recommended for:
- All persons with cirrhosis.
- Persons with a known family history of HCC.
- Persons > 40 years old without cirrhosis but with high, smoldering HBV DNA (> 2000 IU/mL).
XV. Special Populations & Clinical Coinfections
Treating HBV becomes exponentially more complicated when the patient has overlapping diseases or unique physiological conditions.
Because they share the exact same transmission routes (blood/sex), coinfection is extremely common. In these individuals, Antiretroviral Therapy (ART) should be initiated in ALL those with severe liver disease (completely regardless of how high their CD4 count is), and in all those with a CD4 count ≤ 500 cells/mm³.
Recommended Regimen: Tenofovir + Lamivudine (or Emtricitabine) + Efavirenz as a combined fixed-dose. (Note: Tenofovir and Lamivudine are highly effective at treating BOTH HIV and HBV simultaneously!).
Persistent HDV replication is the absolute most important predictor of severe mortality. Bachelor's Physiology: HDV is a "defective" RNA virus. It cannot survive on its own; it literally steals the HBsAg envelope generated by HBV to coat itself and infect cells!
Treatment: PEG-IFN (Interferon) is the ONLY drug currently effective against HDV. Highly advanced oral NAs (like Tenofovir) have virtually zero effect on HDV replication because HDV is an RNA virus that doesn't use reverse transcriptase!
In patients with both viruses, HBV DNA levels are usually surprisingly low or totally undetectable. This is because the Hepatitis C Virus (HCV) is highly dominant and suppresses HBV, making HCV responsible for the active hepatitis.
Treatment: Patients should generally receive targeted treatment for HCV first. However, HBV DNA must be strictly and aggressively monitored during HCV therapy, because curing the HCV instantly removes the suppression, causing the silent HBV to terrifyingly reactivate!
Treating TB requires heavily toxic drugs (isoniazid, rifampicin, pyrazinamide). Drug-induced liver injury (hepatotoxicity) from these anti-TB drugs is three- to six-fold higher and far more dangerous in persons concurrently coinfected with HBV, HCV, or HIV. Extreme caution and LFT monitoring is required.
Treatment: Tenofovir (TDF) is the absolute preferred antiviral because it boasts a better resistance profile and extensive, proven safety data in pregnancy. Entecavir (ETV) safety is unknown, and IFN-based therapy is absolutely contraindicated.
Prevention of MTCT (Mother-to-Child): To prevent the 90% chronic infection rate in babies, deliver the first dose of the Hepatitis B vaccine as soon as physically possible after birth (strictly within 24 hours). Add HBIG (Hepatitis B Immune Globulin) if available to provide instant, passive circulating immunity (achieves >90% total efficacy).
Because oral NAs are cleared by the kidneys, all NAs require massive dose adjustments in dialysis patients to prevent toxic overdose.
HBsAg-positive persons successfully undergoing renal transplantation must receive prophylactic, preventative NA therapy. Why? Because the heavy immunosuppressants required to prevent kidney rejection will cause life-threatening HBV reactivation post-transplant! IFN is strictly prohibited here because it aggressively stimulates the immune system, virtually guaranteeing rejection of the new kidney!
XVI. Levels of Prevention & Vaccination
Levels of Prevention:
- Primary Prevention: Government advocacy, strict blood safety strategies (relying on voluntary non-remunerated donations to avoid infected paid donors), rigorous hospital infection control precautions, promoting safe injection/safe sex practices, and vital harm reduction programs (needle exchanges) for IV drug users.
- Secondary Prevention: Aggressive early diagnosis to provide medical support, prevent spread to partners, and counsel the patient to protect the already compromised liver from additional compounding harm (strictly abstaining from all alcohol, tobacco, and hepatotoxic drugs like excessive Tylenol).
- Tertiary Prevention: There is absolutely no surgical treatment for the virus itself. However, for fulminant hepatitis or end-stage advanced cirrhosis/liver failure, the only definitive treatment choice remaining to prevent death is a highly complex full Liver Transplant.
Hepatitis B Vaccination (The Ultimate Shield):
The HBV vaccine is the most overwhelmingly effective tool in preventing transmission. By successfully preventing Hepatitis B, the vaccine also completely eliminates the risk of acquiring Hepatitis D! It was originally introduced in the early 1980s.
- Types of Vaccines:
- Plasma Derived (Historical): Originally derived directly from the plasma of heavily infected HBsAg-positive donors. It utilized highly purified, formalin/heat-inactivated, alum-absorbed sub-virion surface particles (22nm). Because it was meticulously stripped of all detectable nucleic acid, it was completely non-infectious, though public fear of blood products remained high.
- Recombinant DNA (Modern): Developed to replace plasma vaccines. It is genetically engineered (the yeast Saccharomyces cerevisiae is reprogrammed to mass-produce pure HBsAg). This is the safest, most common type used today (e.g., Recombivax HB, Engerix-B).
- Combination Vaccines: To reduce needle sticks in children, HBsAg vaccines can be highly combined with BCG, measles, mumps, rubella, Hib, diphtheria, tetanus, and polio.
- Schedule & Dosage:
- The WHO heavily recommends universal infant vaccination with a 3-dose schedule: Month 0 (Birth), Month 1, and Month 6.
- Administered via deep intramuscular (IM) injection in the anterolateral thigh for newborns/infants, or the deltoid muscle in the shoulder for older children/adults. Crucial Rule: Never inject subcutaneously or in the glutes (buttocks), as injecting into deep fat drastically reduces the absorption and destroys the protective immune response!
- Sero-protection & Non-Responders:
- Complete, lifelong protection is guaranteed if a blood test shows an anti-HBs titer ≥ 10 mIU/mL.
- Some individuals fail to respond to the vaccine. Known factors for a decreased vaccine immune response include: Smoking, morbid obesity, HIV/immunocompromised state, hemodialysis, severe prematurity, genetic unresponsiveness, chronic disease, improper subcutaneous injection, or accidentally freezing the vaccine vial before use.
XVII. Dental Considerations for Hepatitis B
For practicing dentists and oral surgeons, HBV and HCV are critically important public health threats, as they represent the most common blood-borne infections transmitted via contaminated sharps, drills, and surgical instruments.
- Associated Clinical Features in the Mouth: Severe HBV infection and subsequent liver failure manifest highly specific signs in the oral cavity:
- Sjögren’s syndrome (severe dry mouth).
- Lichen planus (white, lacy patches on the mucosal lining).
- Glossitis (swollen, beefy red tongue) and/or angular cheilosis (cracking at the corners of the mouth) due to poor liver nutrient processing.
- Mucosal Ecchymosis (Spontaneous Bleeding): The liver is the factory responsible for producing critical blood clotting factors (Factors II, VII, IX, and X). As the liver fails, these factors disappear, leading to uncontrolled bleeding and massive bruising inside the mouth after minor trauma or brushing.
- Management for Dentists:
- Pre-exposure vaccination is absolutely, legally mandatory for all dental practitioners and students (e.g., Engerix-B 3-dose series).
- Strict, uncompromising adherence to sterilization and universal infection control (using disposable latex gloves, protective eyewear, heavy mouth masks, and rigid, puncture-proof sharps/needle disposal bins).
- Role of the Public Health Dentist: Actively educating people about hidden transmission routes, dispelling cultural myths, heavily encouraging vaccination drives, and promoting healthy lifestyles to prevent compounding liver diseases.
Occupational Needlestick Injury
Case: An unvaccinated, first-year dental student is rushing and accidentally violently sticks themselves deep in the finger with a sharp explorer tool that was just used on a patient. A rapid chart review reveals the patient is known to be highly infectious with chronic Hepatitis B (HBeAg heavily positive). What is the immediate, life-saving post-exposure prophylaxis (PEP) protocol?
Answer: Because the student has zero immunity and the viral load exposure is massive, the student must be rushed to occupational health and given a combination of Passive AND Active immunization.
- Passive: They must receive a large intramuscular injection of HBIG (Hepatitis B Immunoglobulin) strictly within 48 hours (and no later than 7 days). These are pre-made, donor-derived antibodies that will circulate immediately, acting as a temporary shield to hunt down the virus currently swimming in the student's blood.
- Active: Because HBIG only lasts a few weeks, the student MUST simultaneously receive their first dose of the Recombinant Hepatitis B Vaccine at a completely different anatomical site (e.g., opposite arm) to force the student's own immune system to begin building long-term, permanent immunity to finish the fight.
XVIII. Comprehensive References & Recommended Reading
- Kumar, V., Abbas, A. K., & Aster, J. C. (2020). Robbins & Cotran Pathologic Basis of Disease (10th ed.). Elsevier. (Definitive reference for HCC, pathology, and cellular ground-glass morphologies).
- World Health Organization (WHO). (2015). Guidelines for the Prevention, Care and Treatment of Persons with Chronic Hepatitis B Infection. Geneva: WHO Press. (Core reference for Priority 1 & 2 treatment algorithms and TDF/ETV recommendations).
- Jameson, J. L., Fauci, A. S., Kasper, D. L., Hauser, S. L., Longo, D. L., & Loscalzo, J. (2018). Harrison's Principles of Internal Medicine (20th ed.). McGraw-Hill Education. (Core reference for clinical manifestations, virology, and extra-hepatic complications).
- Katzung, B. G., & Trevor, A. J. (2021). Basic & Clinical Pharmacology (15th ed.). McGraw-Hill Education. (Pharmacodynamics and detailed mechanism of action for Nucleoside Analogues and Interferons).
- Lok, A. S., & McMahon, B. J. (2015). Chronic hepatitis B: update 2009. Hepatology, 50(3), 661-662. (AASLD practice guidelines supplementing serological interpretation).
Quick Quiz
Hepatitis B Virus Quiz
Microbiology - mobile-friendly and focused practice.
Privacy: Your details are used only for quiz tracking and certificates.
Hepatitis B Virus Quiz
Microbiology
Preparing questions...
Choose your answer and keep your streak alive.
Great effort.
Here is your quick performance summary.
