Embolism in Pathology
Embolism Pathology
By the conclusion of this exhaustive master guide, you will be deeply conversant with:
- The precise pathophysiological definition of an Embolism and its direct linkage to tissue infarction.
- The detailed classification of emboli based on physical state, source, and infectious status.
- The distinct, divergent pathways and clinical outcomes of Arterial (Systemic) versus Venous thromboembolism.
- The exact hemodynamic collapse mechanisms seen in Pulmonary Embolism (PE), including acute cor pulmonale.
- Highly specialized embolic phenomena: Paradoxical, Retrograde, Fat, Amniotic Fluid, and Gas embolisms, complete with diagnostic criteria and underlying physics.
I. Definition & The Infarction Link
In the study of hemodynamic disorders, it is critical to distinguish between the mechanism of injury and the final resulting lesion. While an infarction is the ultimate pathological result (the localized area of ischemic tissue necrosis), an embolism is the primary mechanical cause that initiated the cascade.
Embolism Defined
- Embolism: The pathophysiological process of partial or complete obstruction of a part of the cardiovascular system by any foreign mass carried within the bloodstream.
- Embolus: The transported intravascular mass itself. It is a detached material that has broken free from its original site of origin and is now traveling freely through the circulatory system.
- Physical States: An embolus is not exclusively a blood clot. It can be a detached intravascular solid (clots, tumor clumps), a liquid (fat droplets, amniotic fluid), or a gaseous mass (air bubbles, nitrogen).
The Key Pathophysiological Sequence:
- Formation: An embolus forms or is introduced into the vasculature (e.g., a deep vein thrombosis breaks loose, or a fractured bone releases marrow fat).
- Transit: The embolus travels seamlessly through the larger upstream vessels of the bloodstream.
- Impaction: It eventually reaches a downstream vessel where the lumen diameter narrows to the point that it is physically too small for the mass to pass.
- Obstruction & Ischemia: The mass tightly wedges into the vessel, instantly halting anterograde arterial blood flow, leading directly to localized tissue hypoxia and ischemia.
- Infarction: If the occlusion is severe, sudden, or prolonged—and if collateral circulation is inadequate—the ischemic cells pass the point of no return, culminating in irreversible coagulative necrosis (infarction).
💡 The Golden Rule of Embolism vs. Thrombosis
A Thrombus forms locally and stays exactly where it was created, growing firmly attached to the endothelial wall of the vessel. An Embolus travels. It is a rogue, detached piece of material that breaks off, hops onto the cardiovascular "highway," and violently crashes when the road narrows. All thromboemboli started as thrombi, but not all thrombi become emboli!
II. Classification of Emboli
Emboli are highly heterogeneous. Pathologists classify them based on the matter they are composed of, whether they carry an infectious payload, their specific anatomical source, and their eventual destination.
- Solid: Detached blood clots (Thromboemboli - which astonishingly make up 90-99% of all clinical emboli), fragmented atheromatous plaques (cholesterol crystals), aggressive tumor cell clumps, necrotic tissue fragments, parasites, or bacterial vegetations.
- Liquid: Globules of marrow fat, amniotic fluid during childbirth, or inadvertently injected foreign fluids.
- Gaseous: Atmospheric air bubbles (introduced via IV lines or trauma) or other gases like Nitrogen precipitating out of the blood in decompression sickness.
- Bland: Completely sterile, containing no viable bacteria. They cause pure ischemic damage.
- Septic: Heavily infected with pyogenic bacteria. When a septic embolus lodges in distant tissue, it doesn't just cause ischemia; it actively seeds the dying tissue with bacteria, rapidly forming a highly destructive metastatic abscess.
- Cardiac emboli: Arising from the chambers of the heart (primarily the left atrium or left ventricle).
- Arterial emboli: Originating from systemic arteries (e.g., an ulcerated plaque in the internal carotid artery firing emboli into the cerebral circulation).
- Venous emboli: Originating from systemic veins (e.g., deep leg veins) and traveling to the right heart.
- Lymphatic emboli: Usually composed of metastatic tumor cells traveling through the lymphatic vessels to regional lymph nodes.
The 8 Major Clinical Types of Embolism:
In clinical practice, these are the primary culprits causing morbidity and mortality:
- Pulmonary embolism (PE)
- Systemic (Arterial) embolism
- Fat embolism
- Air embolism
- Decompression sickness (Nitrogen gas embolism)
- Amniotic fluid embolism
- Atheroembolism (Cholesterol embolism)
- Tumour embolism
III. Thromboembolism (90%+ of all Emboli)
A detached thrombus, or a fragment of a thrombus that breaks off, constitutes the overwhelming majority of emboli. Because the cardiovascular system is a closed loop split into two distinct halves (pulmonary and systemic), the origin of the thrombus absolutely dictates its deadly destination.
A. Arterial (Systemic) Thromboembolism
- Origin: Around 80% of systemic arterial emboli arise from intracardiac mural thrombi. These predominantly occur in a diseased left heart (e.g., mural thrombi forming on the scarred wall of the left ventricle following a massive myocardial infarction, stagnant blood in a fibrillating left atrium, or vegetative endocarditis on the mitral and aortic valves). The remaining 20% derive from aortic aneurysms, carotid atherosclerosis, or paradoxical emboli.
- Destination: Because it starts in the arterial system, it is propelled OUT to the systemic capillary beds of the body. They invariably cause localized infarction at the sites of lodgement.
Specific Effects & Locations of Arterial Emboli:
The ultimate clinical consequence depends heavily upon the size of the embolus, the specific site of lodgment, and the adequacy of local collateral circulation.
- Lower Limbs (70-75%): The most common destination. Results in Acute Limb Ischemia. Clinicians look for the classic "6 Ps": Pain, Pallor (paleness), Pulselessness, Paresthesia (tingling/numbness), Paralysis, and Poikilothermia (the limb feels ice cold). If collateral circulation is inadequate and surgery is delayed, the infarction progresses to irreversible Gangrene.
- The Brain (10%): Lodging in the cerebral circulation (often the Middle Cerebral Artery), causing a massive ischemic stroke and potentially sudden death.
- Internal Viscera (10%): Splenic infarctions, renal infarctions (causing flank pain and hematuria), or severe acute mesenteric ischemia in the intestines (a highly lethal condition causing excruciating abdominal pain out of proportion to the physical exam).
- Myocardial Infarction: An extremely rare event where an embolus is swept directly into the coronary arteries.
B. Venous Thromboembolism (VTE)
- Origin: The vast majority (over 95%) of venous emboli originate from Deep Vein Thrombosis (DVT) within the large, deep veins of the lower extremities (specifically the popliteal, femoral, and iliac veins). Less common sources include severe varicosities, pelvic venous plexuses (especially post-surgery or post-partum), or the intracranial cavernous sinus.
- Destination: Because it originates in the systemic veins, the clot flows with venous return, gradually moving into larger and larger vessels (up the Inferior Vena Cava). It dumps directly into the right atrium, passes into the right ventricle, and is violently pumped out into the pulmonary arterial tree.
- The Ultimate Consequence: Rapid, acute obstruction of the pulmonary arterial circulation, causing Pulmonary Embolism (PE).
Arterial vs. Venous Emboli Destination
Remember the simple directional plumbing of the heart to never confuse the two on an exam:
- Venous emboli go to the Ventilators: (Lungs = Pulmonary Embolism).
- Arterial emboli go to the Appendages and Organs: (Brain, Kidneys, Legs = Stroke / Acute Limb Ischemia).
IV. Pulmonary Embolism (PE)
Pulmonary embolism is the most common, preventable, and highly fatal form of venous thromboembolism. It involves the sudden, acute occlusion of the pulmonary arterial tree by a traveling blood clot.
Etiology & Risk Factors (Virchow's Triad)
The formation of the initial DVT is driven by Virchow's Triad: endothelial injury, hypercoagulability, and venous stasis.
- Highly prevalent in hospitalized, post-operative, or bed-ridden patients due to severe venous stasis in the legs.
- Ambulatory patients with underlying genetic hypercoagulable states (e.g., Factor V Leiden mutation).
- Specific Hormonal Triggers: Late pregnancy, the puerperal (post-partum) state, and the use of oral contraceptive pills. Estrogen fundamentally alters the liver's production of clotting factors, tipping the blood into a pro-thrombotic state.
Pathogenesis (The Hemodynamic Journey):
- Detachment of a fragile, propagating thrombus from the deep leg veins.
- The thrombo-embolus flows effortlessly through the widening venous drainage into the Inferior Vena Cava (IVC).
- Drains seamlessly through the Right Atrium and into the Right Ventricle.
- Pumped out forcefully into the pulmonary artery, where the vascular tree begins to branch and rapidly narrow.
Outcome A (Saddle Embolus): If the thrombus is massive (often shaped like a long snake reflecting the leg vein it came from), it gets impacted directly at the main bifurcation (the "crotch") of the pulmonary artery. It straddles both the left and right main pulmonary arteries like a saddle on a horse.
Outcome B (Multiple Small Emboli): Multiple smaller emboli, or a large one that fragments through the mechanical churning of the right ventricle, will bypass the main bifurcation and impact in a number of smaller, peripheral vessels, heavily favoring the highly-perfused lower lobes of the lungs.
Consequences of Pulmonary Embolism:
The severity of the resulting syndrome depends heavily upon the size of the occluded vessel, the sheer number of emboli showering the lungs, and the pre-existing baseline cardiovascular health of the patient.
A massive pulmonary embolism (like a Saddle Embolus) results in virtually instantaneous death, often without the patient even having time to complain of chest pain or dyspnea!
Pathophysiology: It physically blocks 100% of blood from leaving the right heart. Consequently, the left heart receives absolutely no blood. Systemic cardiac output instantly drops to zero, leading to cardiovascular collapse and Pulseless Electrical Activity (PEA). If death is slightly delayed, the clinical features mimic a massive myocardial infarction (severe chest pain, sweating, profound cardiogenic shock).
Occurs when numerous small emboli suddenly obstruct over 60% of the total pulmonary circulation. The right ventricle, which is normally a thin-walled, low-pressure pump, suddenly faces an impenetrable wall of resistance. It must pump incredibly hard, resulting in rapid, acute right heart failure (severe acute dilatation of the right ventricle). This pushes the intraventricular septum leftward, further crushing left ventricular filling.
Occurs upon obstruction of relatively small-sized, peripheral pulmonary arterial branches. Interestingly, because the lung has a dual blood supply (Pulmonary arteries + Bronchial arteries), true infarction only occurs if the patient's underlying cardiovascular status is already compromised (e.g., existing heart failure). Clinical features include severe pleuritic chest pain (pain worsens upon taking a deep breath), dyspnea, and hemoptysis (coughing up blood) as the necrotic lung tissue bleeds into the alveoli.
Obstruction of terminal branches (end-arteries). The dual bronchial blood supply manages to keep the tissue alive (no infarction), but the ischemic endothelial damage causes the capillaries to leak massively, leading to central pulmonary hemorrhage. Features include hemoptysis and dyspnea, but less pleuritic chest pain due to the central location away from the sensitive pleura.
The vast majority (60-80%) of very small, clinically silent pulmonary emboli are effectively dissolved and cleared by the body's natural fibrinolytic defense system (plasmin aggressively dissolving the fibrin clot network within days to weeks).
These are the devastating long-term sequelae of multiple, recurrent small thromboemboli that undergo organization (fibrotic scarring) rather than fibrinolytic resolution. Over years, these organized scars permanently narrow and stiffen the pulmonary vascular bed, driving pulmonary pressures steadily upward until the right heart ultimately fails.
V. Contrasting Pulmonary Thrombosis vs. Thromboembolism
Although extremely rare, localized thrombosis can occur directly within the pulmonary arteries (usually secondary to existing pulmonary atherosclerosis, severe pulmonary hypertension, or local trauma). It is a diagnostic necessity for a pathologist to differentiate a primary local thrombus from a traveling embolus during an autopsy.
| Diagnostic Feature | Pulmonary Thrombosis (Primary) | Pulmonary Thromboembolism (Secondary) |
|---|---|---|
| Pathogenesis | Locally formed at the exact site of occlusion. | Travelled from a distant source (usually deep leg veins). |
| Anatomical Location | Typically found in small arteries and distal branches where flow is naturally sluggish. | Found abruptly lodged in major arteries and main bifurcations (can be massive in size). |
| Wall Attachment | Firmly, biologically adherent to the vessel wall. Difficult to scrape off. | Loosely attached, wedged tightly, or lying completely free within the lumen. |
| Gross Appearance | Distinct architecture: the head is pale (platelet-rich), the tail is dark red (RBC trapped). | No distinct distinction in head/tail; usually has a smooth-surfaced, dry, dull, cast-like appearance matching the leg vein. |
| Microscopic Architecture | Platelets and fibrin are laid down in beautifully distinct, alternating microscopic layers. Lines of Zahn are clearly and prominently seen. | Haphazardly mixed with blood clot due to churning in the heart. Lines of Zahn are generally rare, disrupted, or entirely absent. |
VI. Detection and Diagnosis of Emboli and Infarction
Because emboli (especially a massive PE) can lead to rapid right ventricular strain, profound cardiovascular collapse, and sudden death within minutes, immediate and highly accurate diagnosis is a life-saving critical priority.
Clinical Signs & Symptoms (High Suspicion):
- Tachypnea: An abnormally fast breathing rate (>20 breaths/min), seen in an overwhelming 96% of cases.
Pathophysiology: The embolus creates a massive Ventilation-Perfusion (V/Q) mismatch. It creates "alveolar dead space"—areas of the lung where oxygen enters the alveoli, but absolutely no blood is flowing past to pick it up. The brain detects low oxygen and forces the body to hyperventilate to try and compensate. - Tachycardia: A rapid, pounding heart rate (>100 beats/min) as the heart struggles to maintain cardiac output against the blockage.
- DVT Signs: Unilateral leg swelling, throbbing pain, warmth, and erythema (tenderness usually localized deep in the calf muscle).
- Cyanosis: A disturbing bluish discoloration of the lips and nail beds due to profound, systemic hypoxemia.
Advanced Imaging Tests (The Gold Standards):
- CT Pulmonary Angiography (CTPA): The absolute most preferred and highly accurate diagnostic test! It utilizes rapid 3D CT imaging combined with an intravenous iodine contrast dye to physically visualize the pulmonary tree. An embolus appears as a dark, definitive "filling defect" cutting off the bright white dye.
- Ventilation-Perfusion (V/Q) Scan: A highly specialized nuclear medicine scan used to map air flow (ventilation) against blood flow (perfusion) in the lungs. Particularly vital if a CTPA cannot be performed (e.g., in a pregnant patient to avoid high-dose chest radiation, or in a patient with severe kidney failure where CT iodine contrast is highly nephrotoxic). A PE shows normal ventilation but totally absent perfusion.
- Doppler Ultrasound: Used to scan the deep veins of the legs for a DVT. Finding the primary source strongly suggests the presence of a PE in a symptomatic patient without needing to radiate the chest.
- Echocardiogram: An ultrasound of the heart that cannot usually see the lung arteries directly, but can definitively identify right heart strain (the right ventricle ballooning outward = acute cor pulmonale) or, occasionally, actually visualize a massive clot actively "in transit" whipping around inside the right atrium or ventricle.
Laboratory Blood Tests:
- D-Dimer Test: Measures the presence of D-dimer, a highly specific protein fragment released exclusively when the body's plasmin is actively breaking down a cross-linked fibrin clot.
Clinical Utility & Caveat: A perfectly normal/negative D-dimer safely and definitively rules OUT a clot (it has an exceptionally high negative predictive value). However, a high/positive level is extremely non-specific! D-dimer elevates in pregnancy, cancer, severe inflammation, and post-surgery. Therefore, a high D-dimer demands a CTPA to actually confirm the diagnosis. - Arterial Blood Gas (ABG): Measures exact oxygen and carbon dioxide levels in the deep arterial blood. In a classic PE, the ABG reveals severe hypoxemia (low oxygen) and a profound respiratory alkalosis (low CO2) because the patient is hyperventilating and blowing off all their acid.
❓ Applied Clinical Question: The Post-Op Patient
Case: A 55-year-old female is recovering in the hospital 4 days after undergoing major orthopedic surgery on her right knee. She suddenly calls frantically for the nurse, complaining of sharp, stabbing chest pain specifically when she breathes in (pleuritic pain), and she begins coughing up bright red blood (hemoptysis). Her respiratory rate is 28, and her right calf is visibly swollen, red, and warm to the touch.
What is the most likely diagnosis, what was the exact path the object took to get there, and what is the gold-standard imaging test you should order immediately?
Answer: She is suffering from an acute Pulmonary Embolism that has progressed to a pulmonary infarction (which is the direct cause of the hemoptysis and pleuritic pain).
The Path: A Deep Vein Thrombosis (DVT) formed in the stagnant veins of her immobilized right leg -> it broke free -> traveled up the Iliac vein -> into the Inferior Vena Cava (IVC) -> Right Atrium -> Right Ventricle -> Pulmonary Artery -> and permanently lodged in a small peripheral lung branch.
The Test: The physician must immediately order a CT Pulmonary Angiography (CTPA) to visualize the clot and initiate immediate systemic anticoagulation therapy (e.g., Heparin).
VII. Septic Embolism & Systemic Embolism
While we explored systemic arterial emboli previously, the fundamental nature of the material traveling through the arteries can drastically, and dangerously, alter the clinical outcome.
Systemic Arterial Embolism (A Brief Review):
These originate predominantly from sterile thrombi residing in a diseased left heart. Because they travel out through the aorta, they invariably cause pure, sterile ischemic infarction at the highly perfused sites of lodgement (legs, brain, viscera).
Septic Emboli: The Infected Missiles
- These are highly dangerous emboli containing dense, active colonies of viable bacteria.
- Origin: They originate classically from Vegetative Mural Endocarditis (massive, friable clumps of highly active bacteria and fibrin aggressively growing on and destroying the inner heart valves, notably the mitral or aortic valves, frequently caused by Staphylococcus aureus or Streptococcus viridans).
- Pathology: When these infected fragments break off and lodge in distant, healthy tissues, they do not merely cause ischemic necrosis; they actively seed the dying tissue with millions of bacteria. This dual-action injury rapidly liquifies the dead tissue, forming a highly destructive metastatic abscess filled with purulent exudate (pus) wherever the embolus lands (e.g., brain abscesses, renal abscesses).
Clinical Sign of Septic Emboli: Janeway Lesions
If a patient is suffering from Acute Bacterial Endocarditis, tiny fragments of the infected valve vegetations can shoot down the arteries of the arm and lodge tightly in the tiny capillary beds of the hands and feet.
This creates Janeway Lesions: Small, completely painless, non-tender subcutaneous maculopapular (flat to slightly raised) hemorrhagic lesions typically found on the thick pulp of the fingers or the palms.
Pathophysiological Distinction: Because Janeway lesions are essentially painless, physical micro-abscesses resulting from traveling bacteria, they are clinically distinguished from Osler's Nodes. Osler's Nodes are intensely painful, raised, immune-complex deposits found on the finger pads in subacute endocarditis! (Memory Trick: Osler's nodes equal Ouch!).
VIII. Emboli Dependent Upon the Flow of Blood
Usually, emboli predictably follow the standard, directional plumbing of the cardiovascular system. However, two highly specialized types completely and astonishingly defy normal circulatory logic.
A. Paradoxical Embolus: The "Crossed" Clot
- Definition: An embolus which is carried from the venous side of the circulation directly to the arterial side (or vice versa), entirely bypassing the massive capillary filter network of the lungs! This is termed a paradoxical or "crossed" embolus.
- Mechanism: It absolutely requires the presence of an abnormal, congenital arteriovenous communication (a right-to-left intracardiac shunt). The most common structural defects allowing this are a Patent Foramen Ovale (PFO), an Atrial Septal Defect (ASD), or a Ventricular Septal Defect (VSD).
- Clinical Scenario: A patient has a massive, silent DVT in their leg. They go to the bathroom and bear down aggressively (the Valsalva maneuver). This heavy straining temporarily raises right atrial pressure above left atrial pressure. In that split second, the right-to-left shunt opens. The leg clot arrives, bypasses the right ventricle, slips through the PFO directly into the left atrium, drops into the left ventricle, and shoots straight up the carotid artery into the brain, causing a massive, unexplainable ischemic stroke in an otherwise healthy young person!
B. Retrograde Embolus: Flowing Backwards
- Definition: An embolus which miraculously travels in the exact opposite direction of the normal forward flow of blood.
- Classic Example: Widespread metastatic tumor deposits found inexplicably in the high thoracic and cervical spine originating from an early-stage Prostate Carcinoma.
- The Mechanism: The prostatic venous plexus normally drains cleanly into the internal iliac vein. However, it also heavily interconnects with the vertebral venous plexus (Batson's Plexus) that runs the entire length of the spine.
- These complex intraspinal/vertebral veins uniquely operate under incredibly low pressure and, crucially, they possess absolutely NO one-way valves.
- During conditions of high, sustained intra-pelvic or intra-abdominal pressure (such as severe, chronic coughing, straining, or heavy lifting), blood is aggressively forced backward (retrograde movement) from the prostate gland directly up into the spinal column. Insidious tumor cells ride this backward vascular wave, effortlessly leading to severe, destructive bone metastasis far from the primary tumor!
IX. Fat and Tumour Embolism
Not all occlusions are driven by coagulated blood. Lipids and rapidly dividing neoplastic cells frequently hijack the vasculature.
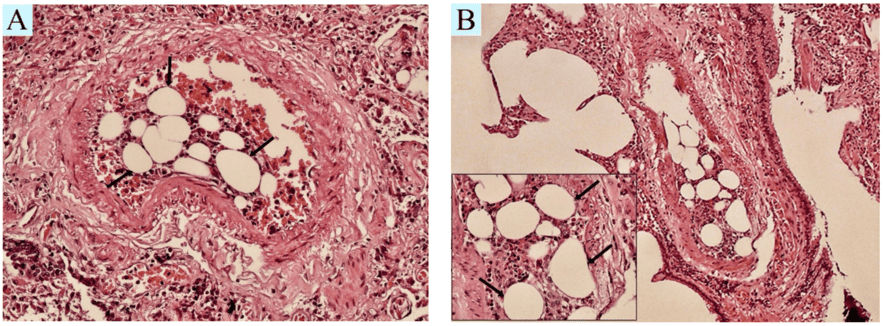
Fat Embolism
Defined as the lethal obstruction of arterioles and capillaries by circulating fat globules. If the obstruction is caused by actual, intact microscopic fragments of adipose (fat) tissue, it is specifically termed a fat-tissue embolism.
- Traumatic Causes: The most common, defining cause is severe physical, crushing trauma to the skeletal bones (e.g., severe long bone fractures like a shattered femur or crushed pelvis in a high-speed car accident). The violent fracture forcefully releases large volumes of semi-liquid, yellow bone marrow fat directly into the freshly ruptured, low-pressure venous sinusoids of the bone.
- Non-Traumatic Causes: Severe fatty liver disease, Diabetes Mellitus (DM), aggressive liposuction procedures, or severe acute pancreatitis.
💡 High-Yield Clinical Triad: Fat Embolism Syndrome (FES)
Fat embolism is uniquely destructive due to both a mechanical and a highly toxic biochemical pathway. If a patient severely fractures their femur and 24 to 72 hours later develops a highly specific triad of symptoms, they have FES:
- Hypoxemia (Acute Respiratory Distress): The fat droplets physically clog the massive pulmonary capillary bed, leading to severe shortness of breath. Furthermore, the biochemical theory states that pulmonary lipase enzymes aggressively break down the fat droplets into highly toxic Free Fatty Acids, which chemically burn and destroy the delicate alveolar pneumocytes, causing Acute Respiratory Distress Syndrome (ARDS).
- Neurologic Abnormalities: The tiny fat micro-globules squeeze through the pulmonary filter and shower the brain, causing sudden confusion, agitation, delirium, or deep coma.
- Petechial Rash: The hallmark sign. A fine, pinpoint, non-blanching red rash appears specifically on the chest, neck, conjunctiva, and armpits (axillae). This is caused by fat micro-emboli rupturing the dermal capillaries, combined with a sudden massive drop in blood platelets as they aggregate around the fat droplets.
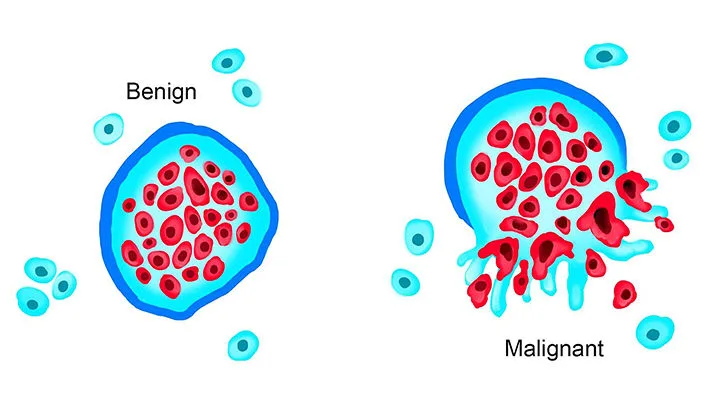
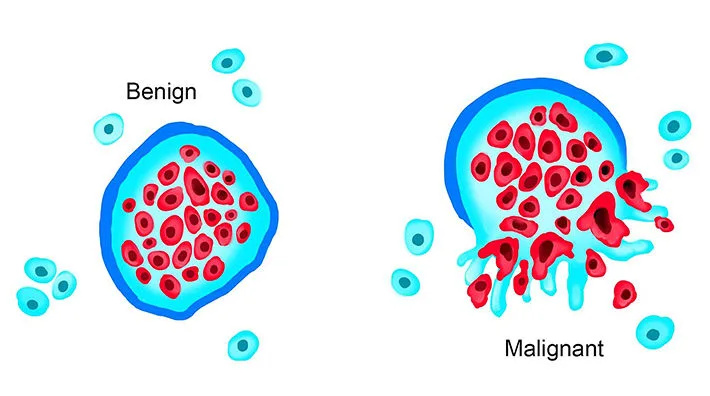
Tumour Embolism
Malignant, highly aggressive tumor cells utilize the bloodstream for distant colonization. They secrete enzymes that degrade the extracellular matrix, allowing them to actively invade local, thin-walled blood vessels (especially veins). Clumps of these cells break off to form circulating tumor emboli, lodging in distant capillary beds and establishing secondary metastatic tumor deposits.
Notable Examples:
- Clear cell carcinoma of the kidney: A notorious cancer that actively invades the renal vein, forms an organized embolic mass, and literally grows backward like a solid snake entirely up the Inferior Vena Cava (IVC), sometimes reaching all the way into the Right Atrium!
- Carcinoma of the lung: Frequently sending embolic metastases to the brain and adrenal glands.
- Malignant Melanoma: Extremely aggressive, utilizing both lymphatic and hematogenous embolic spread widely throughout the body.
X. Amniotic Fluid Embolism (AFE)
A rare (approx. 1 in 40,000 deliveries), highly unpredictable, catastrophic, and exceptionally lethal obstetric complication of pregnancy and childbirth. Mortality rates historically exceed 60-80% if untreated immediately.
- Definition: AFE occurs when amniotic fluid—which is heavily contaminated with fetal squamous skin cells, lanugo (fetal hair), vernix, meconium, and dense cellular debris—aggressively and inappropriately enters the maternal pulmonary circulation. This typically happens through a tear in the placental membranes or deeply ruptured uterine veins during intense labor, complicated delivery, or immediate post-partum trauma.
Pathophysiology (Far beyond a simple physical blockage!)
The profound lethality of AFE is not merely due to fetal debris physically plugging the maternal lung vessels. It triggers a massive, systemic, two-phased immune and hemodynamic collapse:
- Phase 1: Severe Pulmonary Vasospasm & Anaphylaxis. The maternal immune system recognizes the fetal debris as a massive foreign antigen attack. It triggers a profound, systemic inflammatory response (similar to severe anaphylactic shock). The pulmonary arteries violently spasm shut, leading to immediate, severe hypoxemia, acute right heart failure, and rapid cardiogenic shock.
- Phase 2: Disseminated Intravascular Coagulation (DIC). The amniotic fluid is incredibly, dangerously rich in Tissue Factor (thromboplastin). When massive amounts of Tissue Factor enter the mother's central blood supply, it completely overwhelms the coagulation cascade. It aggressively forces the mother's body to form millions of microscopic blood clots everywhere, instantly consuming and depleting all of her platelets and clotting factors.
The Devastating Result: Because all clotting factors are utterly exhausted by the microscopic clots, the mother immediately transitions into uncontrollable, profuse, widespread bleeding from every orifice and surgical site (DIC). She suffers simultaneous hypoxic respiratory failure, cardiogenic shock, and exsanguination.
XI. Air and Gas Embolism
It is a common misconception that only solid objects cause blockages. Air, nitrogen, and other gases can produce highly resilient physical bubbles within the circulation. Due to surface tension, these gas bubbles act exactly like solid, impenetrable physical clots, relentlessly obstructing blood vessels and causing severe downstream tissue hypoxia. There are two main forms: Air Embolism and Decompression Sickness.
A. Air Embolism
Occurs when ambient atmospheric room air is accidentally, or traumatically, introduced into either the venous or arterial circulation.
1. Venous Air Embolism:
Occurs when air is inadvertently sucked into the low-pressure systemic venous system. It can occur under the following specific circumstances:
- Operations on the head & neck / Severe Trauma: Accidental opening of a major vein (like the large internal jugular vein). Because venous pressure in the neck is actually sub-atmospheric (lower than the outside air pressure) when a patient is sitting upright, air is rapidly and forcefully drawn into the open vessel like a vacuum.
- Obstetrical operations & trauma: During normal vaginal delivery, caesarean sections, or aggressive abortions, massive, fatal air embolism may occasionally result from pressurized air entering the huge, opened-up uterine venous sinuses and deep endometrial veins.
- Intravenous (IV) Infusion / Iatrogenic: Can occur if improper, un-purged IV lines are used, or if positive pressure is incorrectly employed to push blood or fluid bags that run completely empty.
- Angiography / Catheterization: During a venogram or central line placement, air may be accidentally entrapped in the massive catheter lumen and injected directly into the deep central venous system.
Factors determining the extreme severity of Venous Air Embolism:
- Amount of air introduced: Highly variable, but usually 100 to 150 ml of rapid air entry is considered definitively fatal in a healthy adult. (In severely ill or compromised patients, as little as 40 ml can be highly lethal). The air enters the Right Ventricle and mixes with the blood to create a dense, frothy, incompressible foam that acts as an "air lock," physically preventing the ventricle from pumping any blood forward into the lungs.
- Rapidity of entry: A sudden massive bolus is far more lethal than a slow, steady leak that the body can gradually absorb.
- General cardiovascular condition of the patient.
- Position of the patient: If the patient's head is elevated higher than their trunk (upright/sitting position), air bubbles—being lighter than blood—will ascend rapidly directly up the superior vena cava and reach the cerebral venous sinuses, causing massive neurological damage!
2. Arterial Air Embolism:
Occurs when air directly enters the pulmonary vein (via chest trauma or lung surgery) or its tributaries. It travels instantly to the left heart and is pumped directly out to the delicate systemic organs. It is extraordinarily highly fatal even in miniscule amounts if the tiny bubbles reach the extremely sensitive coronary arteries (causing instant myocardial infarction) or cerebral arteries (causing severe embolic stroke).
Durant's Maneuver (The Life-Saving Position)
If you are a nurse or physician and you suspect a massive venous air embolism (e.g., a massive central line becomes completely disconnected, and you hear a distinct "hissing" sound), why do doctors instantly instruct you to place the patient in the Left Lateral Decubitus position with the head tilted severely down (Trendelenburg)?
The Physical Rationale: You urgently want the massive, lethal air bubble to rise and trap itself safely against the non-obstructing lateral wall and apex of the Right Ventricle! If the patient sits up or lies flat, the lighter air bubble will float directly up into the pulmonary outflow tract, causing an impenetrable "air lock." This totally stops all forward blood flow to the lungs, dropping cardiac output to zero, causing instant death. By tilting them left and down, you trap the air away from the exit valve, allowing blood to flow underneath it until the air can be slowly absorbed or surgically aspirated via a catheter.
XII. Decompression Sickness (Nitrogen Gas Embolism)
This is a highly specialized, physics-driven form of gas embolism known historically by various names: Caisson's Disease, Divers' Palsy, The Bends, or Aeroembolism. It relies entirely on the complex physics of dissolved atmospheric gases reacting under extreme environmental pressure.
The Physics (Henry's Law)
Henry's Law dictates that the physical solubility of a gas in a liquid is directly proportional to the immense pressure of that gas resting above the liquid. Nitrogen (N2) is a largely inert gas that makes up roughly 78% of normal breathing air.
When a deep-sea diver descends into the depths (or an industrial worker in a highly pressurized caisson/underwater diving-bell performs heavy labor), the immense, crushing physical pressure of the surrounding water forces massive, unnatural amounts of Nitrogen gas to dissolve completely and invisibly into the diver's blood and deep tissue fluids.
Pathogenesis of the Embolism
- The highly dissolved Nitrogen is entirely harmless as long as the diver remains at depth and the pressure remains constantly high.
- The Danger (Ascending too rapidly): If the individual decompresses too suddenly (e.g., panicking and swimming from high-pressure depths to the normal-pressure surface in seconds, or an astronaut flying in an unpressurized cabin rapidly from normal ground level to low-pressure extreme altitudes), the dissolved nitrogen gases physically cannot remain in solution. They instantly precipitate out, expanding violently as thousands of minute gas bubbles directly within the blood and solid tissues. (Think exactly of rapidly unscrewing the cap on a highly shaken, pressurized bottle of soda!).
- These minute, expanding bubbles quickly coalesce (join together) in the venous system to form massive, highly obstructive Nitrogen emboli.
Effects & Severity Factors
The overall severity of the resulting tissue damage depends strictly on: 1) The extreme depth/altitude reached, 2) The total duration of high-pressure exposure, 3) The exact rate of ascent, and 4) The general physiological condition of the individual.
- Obesity Risk Factor: The physiological changes are vastly more serious and lethal in obese persons. Why? Because Nitrogen gas is highly lipophilic (it is up to five times more soluble in fat tissue than in normal body fluids). Obese divers absorb vastly higher, dangerous reserves of Nitrogen into their adipose tissue!
- Sudden decompression from high pressure to normal levels (divers) causes a much more massive gradient shift, and thus is significantly more pronounced and dangerous than moving from normal to low pressure (pilots).
Clinical Effects of Decompression Sickness
1. Acute Form (Immediate Presentation)
Occurs due to the sudden, acute obstruction of thousands of small blood vessels simultaneously.
- 'The Bends': The hallmark symptom. The patient literally doubles over in agony in bed due to excruciating, severe, sharp pain localized deep in the major joints (shoulders, knees), ligaments, and skeletal muscles, caused by gas bubbles aggressively tearing apart the tissue planes and blocking local ischemia.
- 'The Chokes': Massive accumulation of countless tiny nitrogen bubbles in the pulmonary microvasculature, resulting in severe acute respiratory distress, heavy coughing, and suffocating chest pain.
- Cerebral Effects: Nitrogen bubbles expanding directly within the brain or spinal cord may manifest rapidly as severe vertigo, vision loss, instant coma, and sometimes sudden death.
2. Chronic Form (Delayed Presentation)
Occurs in professional divers who suffer repeated, minor, poorly-treated decompression events over years.
- Avascular Necrosis of Bones (Caisson Disease of Bone): Chronic, tiny nitrogen emboli relentlessly occlude and destroy the delicate blood supply to the bones. Classic, highly tested locations include the heavy destruction of the head of the femur, the tibia, and the humerus, leading to early, crippling osteoarthritis.
- Neurological Symptoms: Severe, permanent ischemic damage to the spinal cord tracts includes intractable paraesthesia (numbness/tingling) and irreversible paraplegia (paralysis of the lower body).
- Lung Involvement: Chronic micro-infarctions can present as pulmonary hemorrhage, severe edema, emphysema, and lung atelectasis (collapse).
- Skin Manifestations (The Creeps): Cutaneous itching (pruritus), patchy red erythema, cyanosis (mottling), and pitting edema as bubbles block dermal lymphatics.
❓ Final Module Review Clinical Question
Case: A 35-year-old male, slightly overweight, presents to the ER via ambulance with excruciating, crippling hip and knee pain, extreme shortness of breath accompanied by a dry cough ("the chokes"), and distinct patchy erythema mottling his skin. He reports that he is a professional deep-sea pipeline welder and his oxygen equipment malfunctioned earlier today, forcing him to drop his gear and swim to the surface as fast as humanly possible.
Based entirely on the strict pathogenesis of his condition, what specific gas is currently obstructing his microvasculature, and what is the absolute definitive, life-saving medical treatment?
Answer: The specific gas obstructing his vessels is Nitrogen (N2), which rapidly bubbled out of solution due to his extremely rapid ascent causing a sudden drop in ambient pressure (Henry's Law). He is suffering from severe, acute Decompression Sickness (The Bends). The definitive, non-negotiable medical treatment is immediate transport to and placement inside a Hyperbaric Oxygen Chamber. The chamber safely and heavily re-pressurizes his entire body to forcefully crush the nitrogen bubbles back into a dissolved liquid solution within his blood, and then painstakingly slowly decompresses him over hours so his lungs can safely exhale the nitrogen normally.
XIII. References & Recommended Reading
- Kumar, V., Abbas, A. K., & Aster, J. C. (2020). Robbins and Cotran Pathologic Basis of Disease (10th ed.). Elsevier. (Chapters on Hemodynamic Disorders, Thromboembolism, and Shock).
- Loscalzo, J., Fauci, A., Kasper, D., Hauser, S., Longo, D., & Jameson, J. L. (2022). Harrison's Principles of Internal Medicine (21st ed.). McGraw Hill. (Sections on Pulmonary Embolism and Deep Vein Thrombosis).
- Hall, J. E., & Hall, M. E. (2020). Guyton and Hall Textbook of Medical Physiology (14th ed.). Elsevier. (Aviation, High Altitude, and Space Physiology; Deep-Sea Diving and Other Hyperbaric Conditions).
- Kearon, C., et al. (2016). Antithrombotic Therapy for VTE Disease: CHEST Guideline and Expert Panel Report. Chest, 149(2), 315-352.
Quick Quiz
Embolism Quiz
Pathology - mobile-friendly and focused practice.
Privacy: Your details are used only for quiz tracking and certificates.
Embolism Quiz
Pathology
Preparing questions...
Choose your answer and keep your streak alive.
Great effort.
Here is your quick performance summary.
Embolism in Pathology Read More »