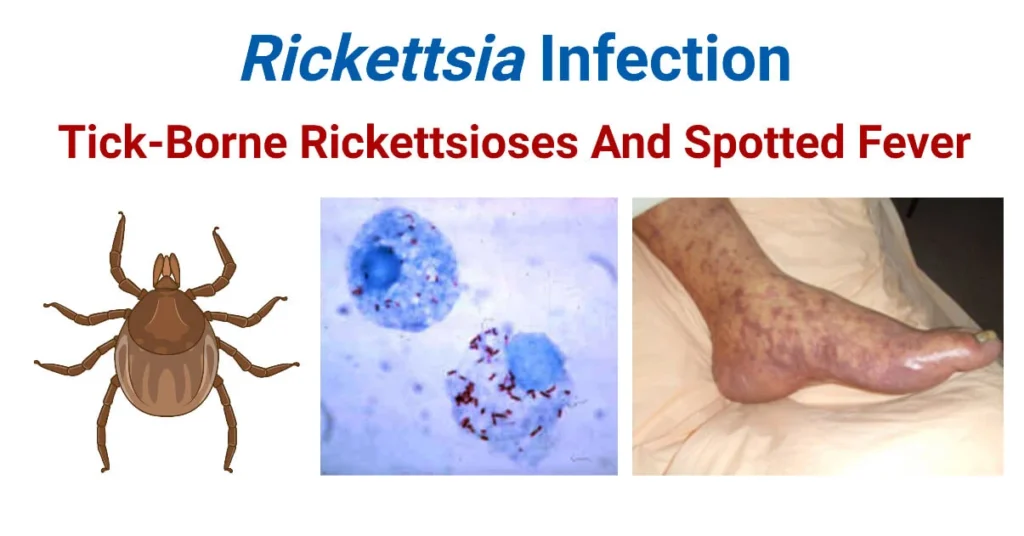
Rickettsiae, Spotted Fevers & Typhus
Rickettsiae, Spotted Fevers, & Typhus
By the conclusion of this highly detailed master guide, you will be deeply conversant with:
- The unique microbiological identity and evolutionary biology of the Rickettsiaceae family.
- The profound pathophysiological mechanisms of endothelial infection, explaining the classic triad of fever, headache, and rash.
- The exhaustive classification and geographic distribution of the Spotted Fever Group, Typhus Group, and related organisms (Ehrlichia/Anaplasma).
- The definitive clinical timeline, deadly complications, and diagnostic pitfalls of Rocky Mountain Spotted Fever (RMSF).
- The precise pharmacological interventions, including pediatric exceptions and alternative therapies, alongside vector control strategies.
I. Introduction to Rickettsiae & Historical Context
Rickettsiae represent a highly specialized, unique group of bacteria that conceptually bridge the evolutionary gap between classic, free-living bacteria and obligate intracellular viruses. Named after Dr. Howard Taylor Ricketts (who tragically died of typhus in 1910 while studying the very organisms that bear his name), they are historically classified as the devastating causative agents of epidemic typhus and severe spotted fevers.
Despite being among the oldest recognized infectious agents in modern medicine—responsible for altering the course of human history and wars through massive typhus outbreaks—rickettsial diseases remain a massive, ongoing cause of undifferentiated febrile illnesses (fevers of unknown origin) worldwide, particularly in resource-limited, tropical, and heavily forested regions.
Core Biological Identity:
- Obligate Intracellular Parasites: They are fundamentally incapable of surviving, metabolizing, or multiplying outside of a living host cell. They must hijack the host's cellular machinery to live.
- Gram-Negative Architecture: Structurally, they possess an outer membrane, a thin peptidoglycan layer, and an inner membrane, classifying them biologically as Gram-negative.
- Zoonotic Transmission: They are primarily arthropod-borne. They rely heavily on vectors such as hard and soft ticks, fleas, human body lice, and mites (chiggers) to move from host to host.
- Mammalian Reservoirs: The primary reservoir (where the bacteria naturally live, multiply, and maintain their life cycle in the wild) consists of various mammals, including rodents, dogs, and humans.
- Pathological Target (The Endothelium): Once introduced into the human bloodstream, they exhibit a profound tropism (specific target affinity) for the vascular endothelium—the single layer of squamous cells lining the interior of blood vessels. This leads directly to severe systemic vasculitis, microscopic hemorrhages, and characteristic petechial rashes.
II. General Characteristics & Microbiology
Because they are obligate intracellular organisms, investigating them in a clinical or research laboratory requires highly specialized techniques that differ drastically from standard bacteriological protocols.
Morphology & Size:
- They are extremely small, pleomorphic coccobacilli (meaning they can alter their shape, appearing somewhere between a sphere and a tiny rod).
- Their microscopic size ranges from 0.3 to 0.5 μm in width to 0.8 to 2.0 μm in length, making them barely visible under standard light microscopy.
Staining & Culturing (High-Yield Board Concept):
- The Gram Stain Failure: Although they possess a Gram-negative cell wall architecture, they stain incredibly poorly with a standard Gram stain. This is due to their minuscule size, the lipid-rich nature of their outer membrane, and the fact that they hide deep within the cytoplasm of host cells.
- Specialized Stains Required: To visualize them microscopically, pathologists must utilize specialized tissue stains such as the Giemsa stain, Gimenez stain, or Machiavello stain.
- Culturing Limitations: Rickettsiae cannot grow on artificial, cell-free agar media (such as standard Blood agar, Chocolate agar, or MacConkey agar). To culture them, the reference laboratory must utilize living tissue cultures (e.g., Vero cells), embryonated chicken eggs (yolk sac inoculation), or susceptible live laboratory animals (like guinea pigs).
Replication & Evolutionary Genetics:
- They divide strictly by binary fission, but this division occurs entirely within the nutrient-rich cytoplasm (and sometimes the nucleus) of the host cell. They have a very slow generation time (8 to 12 hours).
- Reductive Evolution: Rickettsiae have an incredibly small genome (ranging from 1.1 to 1.6 Mb) that is heavily loaded with "pseudogenes" (broken, fragmented, non-functional genes). Because they have lived safely inside host cells for millions of years, they underwent reductive evolution—they literally threw away their own genes for synthesizing amino acids, nucleotides, and energy.
- The ATP/ADP Translocase: Instead of making their own energy, they possess a unique membrane transport protein (ATP/ADP translocase) that allows them to parasitize the host by physically stealing ATP directly from the host cell's cytoplasm, trading it for depleted ADP!
Vascular Leakage & Virchow's Triad
Why do Rickettsial infections cause such deadly drops in blood pressure, distinct rashes, and organ failure? It comes down to the destruction of the blood vessel lining.
When the bacteria aggressively infect the endothelial cells lining the capillaries and arterioles, the host cells eventually undergo severe necrosis (death) and detach from the blood vessel wall. This physical destruction creates microscopic "holes" in the capillaries.
- Red Blood Cell Leakage: RBCs leak out of the ruptured vessels into the dermal skin layers, causing a non-blanching, petechial/purpuric rash (a rash that does not turn white when you press on it, because the blood is trapped outside the vessel).
- Plasma Leakage: Fluid leaks massively into the interstitial tissues. This causes severe total-body edema, profound hypovolemia (low intravascular blood volume), and ultimately, hypotensive shock.
- Microvascular Thrombosis: The exposed, damaged sub-endothelial collagen powerfully activates the body's clotting cascade. Micro-clots form throughout the body's capillary beds (consumptive coagulopathy/DIC), leading to ischemic microinfarcts in the brain, kidneys, and digits. This systemic vasculitis is the primary mechanism of multi-organ failure and death.
III. Exhaustive Classification of Rickettsial Diseases
Rickettsial diseases are notoriously confusing to categorize. They are strictly and historically grouped based on their clinical presentation, their genetic relatedness, and the specific arthropod vector that transmits them to humans.
Characterized by ticks and mites, causing severe fevers and classic spreading rashes.
- Rocky Mountain Spotted Fever (RMSF): Caused by Rickettsia rickettsii. Transmitted by hard ticks (Dermacentor species). Carries a devastating 5-10% mortality rate if untreated. Found primarily in the Americas.
- Mediterranean Spotted Fever (Boutonneuse fever): Caused by R. conorii. Transmitted by the brown dog tick (Rhipicephalus sanguineus).
Clinical Hallmark: Frequently presents with a "Tache Noire" (a black, necrotic eschar/scab at the exact site of the tick bite). - African Tick Bite Fever: Caused by R. africae. Transmitted by Amblyomma ticks. Common in travelers returning from sub-Saharan African safaris. Also presents with a prominent inoculation eschar and regional swollen lymph nodes.
- Rickettsialpox: Caused by R. akari. The Exception: This is the notable exception in the spotted fever group because it is transmitted by a MOUSE MITE (Liponyssoides sanguineus), not a tick. Presents with a vesicular rash resembling chickenpox.
- Emerging Spotted Fevers: R. parkeri (US), R. 364D (Pacific Coast tick fever), R. japonica (Japanese spotted fever).
Characterized by lice and fleas, notorious for explosive, devastating historical outbreaks in crowded populations.
- Epidemic Typhus: Caused by Rickettsia prowazekii. Transmitted by the human body louse (Pediculus humanus corporis). Highly contagious in crowded, freezing, unsanitary conditions (e.g., refugee camps, wartime trenches). Anne Frank famously died of epidemic typhus in the Bergen-Belsen concentration camp.
Clinical Phenomenon: The bacteria can lay dormant in the reticuloendothelial system (lymph nodes/macrophages) for decades and reactivate later in life when the patient's immunity wanes. This milder, reactivated form is known as Brill-Zinsser disease. - Murine Typhus (Endemic Typhus): Caused by Rickettsia typhi. Transmitted by rat fleas (Xenopsylla cheopis). Often seen in urban areas with high rodent populations.
A distinct geographical entity.
- Scrub Typhus: Caused by Orientia tsutsugamushi (formerly classified as a Rickettsia, but genetically reclassified). Transmitted by the bite of the chigger (the microscopic larval stage of the Leptotrombidium mite).
- Geography: Highly prevalent in the "Tsutsugamushi Triangle" (the Asia-Pacific region, including Japan, India, Australia, and the Pacific Islands). Patients frequently present with a blackened eschar, deafness, and tinnitus alongside the fever.
Closely related intracellular bacteria that target leukocytes (white blood cells) rather than endothelial cells.
- Human Monocytic Ehrlichiosis (HME): Caused by Ehrlichia chaffeensis. Transmitted by the Lone Star tick (Amblyomma americanum). Targets monocytes.
- Human Granulocytic Anaplasmosis (HGA): Caused by Anaplasma phagocytophilum. Transmitted by Ixodes ticks (the exact same tick that transmits Lyme disease). Targets neutrophils.
- Sennetsu Fever: Caused by Neorickettsia sennetsu. Uniquely transmitted to humans by ingesting a trematode (fluke) found in raw, undercooked fish in Japan.
Vectors of the Rickettsial World
To easily keep the bugs and their specific vectors straight for board exams:
- R. rickettsii = Ticks (RMSF).
- R. typhi = Fleas (Murine/Endemic Typhus) - "Typhi flies on Fleas."
- R. prowazekii = Lice (Epidemic Typhus) - "Pro wars cause Epidemics (and body lice)."
- O. tsutsugamushi = Chiggers (Scrub typhus).
- R. akari = Mites (Rickettsialpox).
IV. Cellular Pathogenesis: The Hijacking Mechanism
How do these tiny bacteria cause such massive damage at the cellular level? The process is a highly choreographed molecular invasion.
- Invasion & Cellular Adherence: Rickettsiae utilize outer membrane proteins, specifically OmpA and OmpB, to firmly adhere to specific receptors (like Ku70) on the host endothelial cell surface. Surface Cell Antigen (Sca) proteins further facilitate the actual endocytosis (swallowing) into the cell.
- Phagosomal Escape: Once swallowed, the bacteria find themselves trapped in a phagosome (a cellular stomach). To avoid being digested by lysosomes, the bacteria immediately secrete phospholipase D and hemolysin C, enzymes that rapidly dissolve the phagosome membrane. The bacteria escape directly into the safety of the host's cytoplasm to multiply freely.
- Actin-Based Motility (The Comet Tail): Some species (most notably R. rickettsii) do not wait to lyse and destroy the cell to escape. Instead, they express a surface protein called RickA, which hijacks the host cell's Arp2/3 complex. This forces the host to rapidly polymerize actin filaments directly behind the bacteria, creating a powerful "actin comet tail." This tail physically rockets the bacteria through the cytoplasm and propels it directly through the cell membrane into the adjacent neighboring cell.
Clinical Significance: This allows the bacteria to spread from cell to cell rapidly without ever entering the extracellular blood space, completely hiding from the host's circulating antibodies! (Note: Listeria monocytogenes and Shigella use this exact same actin-rocket mechanism).
V. Rocky Mountain Spotted Fever (RMSF)
RMSF is the most common, most severe, and most frequently fatal tick-borne disease in the United States and the Americas. It requires immediate, aggressive clinical intervention based purely on clinical suspicion.
Epidemiology & Vector Dynamics:
- Vectors: Transmitted primarily by Dermacentor variabilis (the American dog tick, found heavily in the Eastern and South-Central US, notably North Carolina, Oklahoma, and Arkansas) and D. andersoni (the Rocky Mountain wood tick, found in the Western US).
- Seasonality: Highly seasonal. Over 90% of cases peak between April and September when ticks are in their nymph and adult stages, actively seeking mammalian blood meals.
- Transmission Time: The tick must generally be attached and feeding for 6 to 24 hours to successfully reactivate and transmit the dormant bacteria into the human bloodstream.
Clinical Features & The Deadly Timeline:
RMSF is a rapidly progressing disease. Understanding the day-by-day evolution is critical for survival.
- Incubation Period: 2 to 14 days (Average: 7 days) following the tick bite. Many patients never even recall being bitten by a tick.
- Early Phase (Days 1-3): Sudden, abrupt onset of extremely high fever (often 103-104°F), severe unrelenting frontal headache, profound myalgia (muscle pain), and nausea/vomiting.
Crucial Clinical Note: There is absolutely NO RASH during the first three days! Because of this, patients are tragically misdiagnosed with a severe viral illness or a migraine and sent home without antibiotics. - The Rash Phase (Days 3-5): Begins as a faint, macular, blanching rash (pink spots that turn white when pressed).
- Centripetal Spread Pattern: It characteristically starts on the extreme periphery—the wrists, forearms, and ankles—and then spreads centripetally (inward) toward the trunk and chest over the next 24 hours.
- Palmar/Plantar Involvement: It heavily and famously involves the palms of the hands and the soles of the feet.
- Petechial Evolution: By day 5 or 6, as the vasculitis deeply worsens and red blood cells leak out, the rash becomes petechial or purpuric (dark red/purple hemorrhagic dots that do NOT blanch when pressed).
- Late Phase (Severe Complications, Days 7+): The widespread, uncontrolled vascular leakage leads to massive systemic collapse:
- Neurological: Altered mental status, confusion, seizures, and meningoencephalitis.
- Pulmonary: Non-cardiogenic pulmonary edema leading to Acute Respiratory Distress Syndrome (ARDS).
- Renal: Acute Kidney Injury (AKI) driven by hypovolemia (pre-renal) and micro-clots (intra-renal).
- Digital Necrosis: The profound lack of blood flow causes fingers and toes to turn black and gangrenous, frequently requiring surgical amputation.
- Mortality: Untreated mortality historically approaches 20-25%. Even with treatment, modern mortality is 5-10% if antibiotics are delayed past day 5.
The "Spotless" Exception
Approximately 10-15% of RMSF patients NEVER develop a rash, or the rash is highly atypical. This condition is termed "Spotless RMSF". Older patients and darker-skinned individuals are particularly at risk for missing the rash. These patients paradoxically have a significantly higher mortality rate simply because their diagnosis is severely delayed, as physicians wait for a rash that never comes before starting antibiotics.
Rashes on the Palms and Soles
Very few infectious diseases in all of medicine cause a distinct rash on the palms of the hands and the soles of the feet. When you observe this clinical sign, immediately think of driving CARS:
- Coxsackievirus A (Hand, Foot, and Mouth Disease)
- A (Ignore - used for phonetic flow, though some use 'A' for atypical measles/syndromes)
- Rocky Mountain Spotted Fever
- Syphilis (Specifically Secondary Syphilis)
VI. Laboratory Diagnosis
Rickettsial diseases progress incredibly fast. Clinical diagnosis must drive treatment. Do NOT wait for laboratory confirmation to start antibiotics, or the patient may suffer irreversible damage or death.
- Serology (IFA) - The Gold Standard:
- Detects specific antibodies formed against rickettsial antigens via the Indirect Immunofluorescence Assay (IFA).
- Definitive diagnosis requires paired sera showing a fourfold rise in antibody titers between the acute phase (week 1) and the convalescent phase (week 3-4).
- The Limitation: Diagnostic IgG and IgM antibodies typically do not appear in detectable quantities until 7 to 14 days after the onset of illness. Therefore, serology is essentially useless for making the initial life-saving decision in the emergency department. It is used retrospectively to confirm what you already treated. A single titer of ≥1:64 with a compatible clinical illness confirms the diagnosis.
- Direct Detection:
- Skin biopsy of the petechial rash evaluated with immunohistochemistry (IHC) or direct immunofluorescence. This is the fastest way to confirm RMSF in the acute phase, but it has low sensitivity (the bacteria are patchy).
- PCR (Polymerase Chain Reaction) of the blood or skin biopsy is highly specific but suffers from variable sensitivity because the bacterial load in the circulating blood is actually quite low.
- Culture:
- Extremely dangerous to laboratory staff due to the risk of aerosolization. Culturing is restricted strictly to Reference Laboratories requiring high-security BSL-3 (Biosafety Level 3) containment. Utilizes the shell vial technique with cell monolayers.
- The Weil-Felix Test (Historical Context):
- An obsolete, historical agglutination test you may encounter in older literature. It relied on the phenomenon of immunological cross-reactivity: antibodies produced against Rickettsia will accidentally agglutinate (clump) the O-antigens of certain innocuous strains of Proteus bacteria (OX-19, OX-2, OX-K strains).
- It possesses terrible sensitivity and specificity, yielding numerous false positives and false negatives, and has been completely abandoned in modern, evidence-based medicine.
VII. Treatment Pharmacology
A delay in treatment is the single largest cause of mortality in Rickettsial diseases. Empirical therapy must be initiated immediately based on clinical suspicion, often before the rash even fully develops.
1. Doxycycline (The Undisputed Drug of Choice):
- Mechanism: A broad-spectrum tetracycline antibiotic. It is bacteriostatic, entering the host cell and binding reversibly to the 30S ribosomal subunit of the rickettsial bacteria, totally shutting down their protein synthesis.
- Dosage: 100 mg BID (twice daily) intravenously or orally for 5 to 7 days, or strictly until the patient has been completely afebrile (fever-free) and clinically improving for at least 3 consecutive days.
❓ Applied Clinical Question: Pediatric Pharmacology Exception
Case: A 6-year-old boy presents to the Emergency Department with a 103°F fever, severe headache, and a petechial rash spreading from his wrists to his chest. His mother recently pulled an engorged tick off his neck after a camping trip in North Carolina. The attending physician orders Doxycycline IV. The new pediatric nurse questions the order, stating, "Doxycycline is strictly contraindicated in children under 8 years of age due to the risk of permanent enamel hypoplasia and severe grey/yellow tooth discoloration." How should the charge nurse respond?
Answer: The charge nurse must firmly explain that while Doxycycline is indeed normally contraindicated in young children for routine infections, Rocky Mountain Spotted Fever is a rapidly fatal, life-threatening emergency. The CDC and the American Academy of Pediatrics specifically dictate that Doxycycline is the absolute first-line treatment for RMSF for patients of ALL ages, including infants and young children. The risk of neurological devastation, limb amputation, and death from the disease vastly, undeniably outweighs the minimal, often merely cosmetic risk of dental staining from a short 5-to-7-day course of the drug. There is no comparable second-line drug.
2. Alternative Agents:
- Chloramphenicol: The classic historical alternative, particularly used in pregnant women. However, it is highly toxic, binding to the 50S ribosomal subunit. It is heavily associated with severe, unpredictable bone marrow toxicity (idiosyncratic, fatal aplastic anemia) and Grey Baby Syndrome in neonates (due to the baby's lack of liver UDP-glucuronyl transferase to metabolize the drug, leading to cyanosis, cardiovascular collapse, and an ashen grey skin color). It is rarely used in the US today.
- Azithromycin / Fluoroquinolones (Ciprofloxacin): Can be used with varied success for very mild rickettsial diseases (like Mediterranean Spotted fever), but are NOT recommended for severe RMSF.
- Rifampin: Highly effective specifically for severe, doxycycline-resistant cases of Scrub typhus in Southeast Asia.
VIII. Ehrlichiosis and Anaplasmosis
These diseases are closely related to Rickettsiae genetically, present with similar vague febrile symptoms, and are treated with the exact same drugs, but they target completely different cells in the human immune system.
Pathological Target Cells (The Leukocytes):
- Ehrlichia chaffeensis: Actively infects Monocytes and Macrophages.
- Anaplasma phagocytophilum: Actively infects Neutrophils (Granulocytes).
The "Morulae" Phenomenon
Once inside the white blood cells, these bacteria avoid lysosomal destruction and multiply rapidly inside host vacuoles. They pack tightly together into massive, distinct, membrane-bound, mulberry-like clusters called Morulae (from the Latin word for mulberry) inside the cytoplasm of the WBC. Finding these morulae on a peripheral blood smear is diagnostic.
Clinical Presentation & Co-Infection Risks:
- Symptoms: Non-specific flu-like illness: high fever, profound headache, severe myalgia, and malaise.
- The Rash: Unlike RMSF, a rash is UNCOMMON in these diseases (occurring in <30% of Ehrlichiosis cases and <10% of Anaplasmosis cases). This heavily differentiates it clinically from RMSF.
- Laboratory Findings: Because they actively destroy white blood cells and platelets while inflaming the liver, patients present with a classic triad of profound Leukopenia (low WBCs), Thrombocytopenia (low platelets), and elevated hepatic transaminases (AST/ALT).
- Co-Infection Alert: Anaplasma is transmitted by the Ixodes scapularis tick in the Northeast and Midwest US. This is the exact same tick that transmits Lyme disease (Borrelia burgdorferi) and Babesiosis (Babesia microti). A patient bitten by one tick can be simultaneously infected with all three diseases, presenting with a highly complex, confusing, and severe clinical picture!
Diagnosis and Treatment:
- Diagnosis: Examining a peripheral blood smear under a microscope to physically see the morulae inside the WBCs (fast, but low sensitivity). NAAT (PCR of the blood) and Serology (IFA) are definitive.
- Treatment: First-line is strictly Doxycycline for all ages. Rifampin is utilized as an alternative exclusively for patients with true, severe, anaphylactic allergies to tetracyclines or for pregnant women.
IX. Prevention & Environmental Control Strategies
Because the pathophysiology of these diseases is so destructive, and because there are currently NO commercially available vaccines for any Rickettsial diseases, Ehrlichiosis, or Anaplasmosis, prevention relies entirely on rigid environmental and vector control mechanisms.
1. Tick Avoidance & Chemical Repellents:
- Clothing: Wear light-colored protective clothing (makes tiny ticks easier to spot), with long pants tucked securely into high socks to prevent ticks from crawling up the legs from tall grass.
- DEET (N,N-Diethyl-meta-toluamide): A highly effective chemical repellent applied directly to exposed skin. It disrupts the tick's olfactory receptors, masking the human's scent.
- Permethrin: A synthetic neurotoxin specifically lethal to arthropods but generally safe for mammals. It must be applied exclusively to clothing, boots, and camping gear, never directly to the skin. Ticks that crawl across permethrin-treated fabric die rapidly.
- Tick Checks: Conduct thorough full-body tick checks (especially in hair, groin, and axilla) immediately after outdoor activity in endemic areas.
2. Proper Tick Removal Technique:
Removing a tick incorrectly can be deadly. If you squeeze the tick's swollen abdomen, burn it with a hot match, or try to suffocate it with petroleum jelly, the stressed tick will aggressively regurgitate its infected salivary and gut contents directly into the patient's bloodstream, guaranteeing infection.
- The Correct Method: Use fine-tipped tweezers. Grasp the tick as close to the skin's surface (the mouthparts) as physically possible. Apply a steady, even, upward pull without twisting or jerking. Disinfect the bite site immediately after removal.
3. Louse Control (For Epidemic Typhus):
Body lice (unlike head lice) live and lay their eggs in the seams of clothing, only moving to the skin to feed. They thrive exclusively in unhygienic, impoverished, crowded conditions. Control focuses on improving basic sanitation, boiling clothes in hot water (killing both lice and nits), and utilizing systemic chemical delousing procedures (e.g., mass dusting with safe insecticidal powders in refugee situations).
References
- Centers for Disease Control and Prevention (CDC): Diagnosis and Management of Tickborne Rickettsial Diseases: Rocky Mountain Spotted Fever and Other Spotted Fever Group Rickettsioses, Ehrlichioses, and Anaplasmosis (MMWR Recommendations and Reports).
- Mandell, Douglas, and Bennett's: Principles and Practice of Infectious Diseases (9th Edition) - Section on Rickettsiaceae.
- Harrison's Principles of Internal Medicine: (21st Edition) - Chapters detailing tick-borne illnesses and systemic vasculitis.
- American Academy of Pediatrics (AAP): Red Book: Report of the Committee on Infectious Diseases - Guidelines regarding the pediatric administration of Doxycycline.
- World Health Organization (WHO): Guidelines on the management and vector control of Epidemic and Scrub Typhus in developing nations.
Quick Quiz
Bacteriology Intro Quiz
Microbiology - mobile-friendly and focused practice.
Privacy: Your details are used only for quiz tracking and certificates.
Bacteriology Intro Quiz
Microbiology
Preparing questions...
Choose your answer and keep your streak alive.
Great effort.
Here is your quick performance summary.
Rickettsiae, Spotted Fevers & Typhus Read More »