- Abstract: A concise summary of a study that communicates the essential information about the study.
- Assumption: A statement based on logic or reason whose correctness or validity is taken for granted.
- Data: Units of information.
- Descriptive research: Non experimental research designed to discover new meanings and to provide new knowledge when there is little known about the phenomena of interest.
- Hypothesis: A statement of predicted relationship between two or more variables in a research study. An educated or calculated guess by the researcher.
- Informed consent: Voluntary agreement by a study subject to participate in the research study after being fully informed about the study.
- Phenomena: Facts or events that can be observed or scientifically described because they are known through senses rather than thoughts or intuition.
- Reliability: Stability of a measuring item overtime. A measure of the extent to which random variation may have influenced the stability and consistency of results.
- Validity: Ability of the test item to measure what it is expected to measure. Extent to which research findings represent reality.
- Variable: An attribute or characteristic that can have more than one value, such as height, weight and blood pressure.
- Dependent variable: The variable that changes as the independent variable is manipulated by the researcher.
- Independent variable: The variable that is purposely manipulated or changed by the researcher.
- Confounding variable: Variable outside the purpose of the study that could influence the study’s results.
- Qualitative data: Data characterized by words rather than numbers.
- Quantitative data: Data characterised by numbers.
- Population: A total group of individual people or things meeting the designed criteria of interest to the researcher.
- Sample: A smaller part of the population selected to represent the whole population.
A clear, concise summary of a study, usually found at the beginning of an article in research journals.
- To help readers quickly see if the paper is relevant to their own research.
- To share key findings with those who can't read the full paper.

Units of information, facts, figures, or knowledge collected during a study. This is also known as processed information.
Data is classified into major categories/scales:
- Nominal data
- Ordinal data
- Discrete data
- Continuous data
- Interval data
- Ratio data
Based on the provided visuals, data can be broadly divided and then further broken down:
Non-numerical data that describes characteristics, such as yes/no responses or eye colour.
- Description: Data used for naming variables, without any order or hierarchy.
- Examples: Hair colour, Gender, Letters, Colours, Symbols, Words.
- Description: Data used to describe the order of values, where categories have a meaningful sequence, but the differences between ranks are not necessarily equal.
- Examples: Opinion (Agree, neutral, disagree), Tumour Grade (1, 2, 3), Time of day (Morning, Noon, Night), 1 = happy, 2 = neutral, 3 = unhappy.
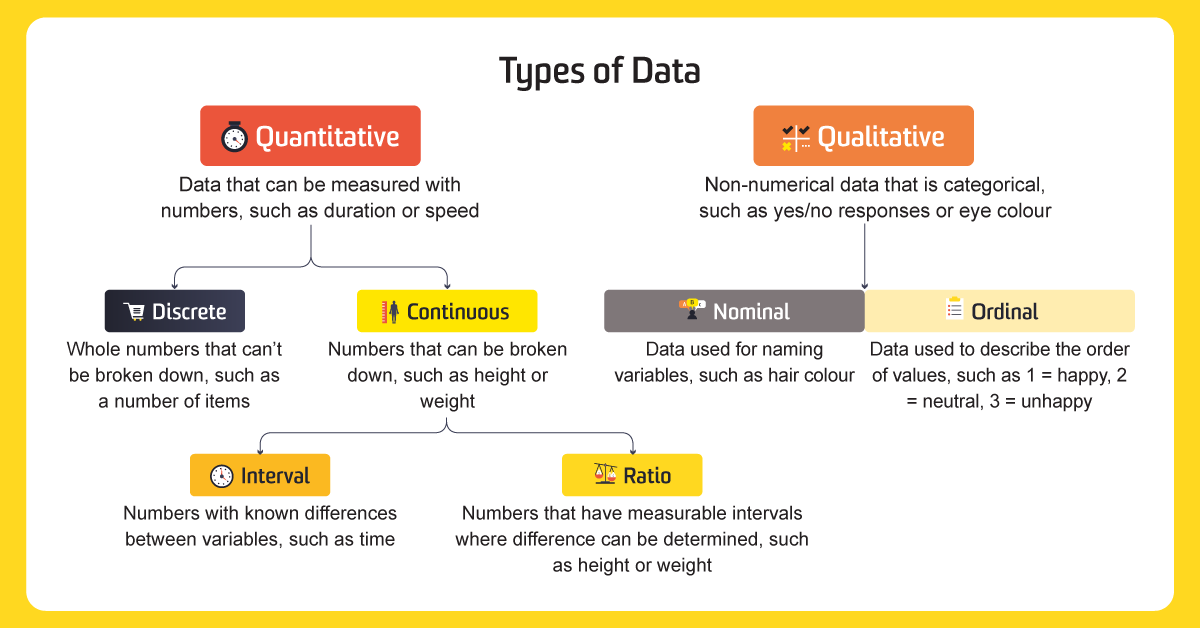
Data that can be measured with numbers, such as duration or speed.
- Description: Whole numbers that can't be broken down, often representing counts.
- Examples: Number of items, Dates (200; 1000; 1500), Temperature in specific increments (30°C; 45°C; 60°C), pH (1.2; 4.5; 7.2), IQ (80; 120; 140).
- Description: Numbers that can be broken down into smaller units, representing measurements along a continuous scale.
- Examples: Height, Weight, Temperature range, Distance travelled, Time interval, Age range.
- Description: Numbers with known differences between variables, but without a true zero point (zero doesn't mean the absence of the characteristic).
- Examples: Time (from the visual's first diagram), Temperature (from the visual's second diagram, also showing as discrete/interval).
- Description: Numbers that have measurable intervals and a true zero point (zero means the absence of the characteristic), allowing for meaningful ratios.
- Examples: Height, Weight (from the visual's first diagram), Distance travelled, Time interval, Age range (from the visual's second diagram, also showing as continuous/ratio).
Qualities or quantities, properties, or characteristics of people, things, or situations that change or vary. They can have more than one value, such as height or weight.
- Variables purposely manipulated or changed by the researcher. Also called a "Manipulative Variable."
- Example: In "factors influencing the uptake of family planning services," the "factors influencing" are the independent variables (e.g., educational level, access to clinics, cultural beliefs).
- The variable influenced by the independent variable. It's the outcome variable that the researcher aims to understand, explain, or predict.
- Example: Following the previous example, "uptake of family planning services" is the dependent variable.
- Undesirable variables that influence the relationship between the independent and dependent variables. They are not the focus of the study but can affect the outcome.
- Example: In a study examining the effect of a new teaching method on student performance, students' prior knowledge or motivation could be extraneous variables.
- A type of extraneous variable that is related to both the independent and dependent variables, making it difficult to determine the true relationship between the IV and DV.
- Example: In a study looking at the relationship between coffee consumption and heart disease, smoking could be a confounding variable if coffee drinkers are also more likely to smoke, and smoking itself increases heart disease risk.
- Hypothetical variables that help explain the relationship between the independent and dependent variables but cannot be directly observed or measured. They intervene between the IV and DV.
- Example: In a study where increased job satisfaction (IV) leads to increased productivity (DV), "motivation" could be an intervening variable. Job satisfaction leads to increased motivation, which in turn leads to increased productivity.
- A variable that influences the strength or direction of the relationship between an independent variable and a dependent variable. It "moderates" the relationship.
- Example: In a study on the relationship between stress (IV) and job performance (DV), social support could be a moderating variable. The negative effect of stress on performance might be weaker for individuals with high social support.
- Variables that are kept constant or accounted for by the researcher to minimize their potential effect on the relationship between the independent and dependent variables.
- Example: In an experiment testing the effectiveness of a new fertilizer on plant growth, factors like sunlight exposure, soil type, and water amount would be controlled variables.
- Variables that can be divided into distinct categories, but do not have a natural numerical order.
- Example: Gender (male, female, non-binary), blood type (A, B, AB, O), religious affiliation.
- Variables that can take on any value within a given range, and can be measured with infinite precision.
- Example: Height, weight, temperature, time.
- A type of categorical variable that can only take on two values or categories.
- Example: Yes/No, True/False, Alive/Dead, Pass/Fail.
The specific way a researcher defines and measures variables in a study. It specifies how study variables will be observed or measured in the actual research situation.
- Example: If "pain" is a variable, its operational definition might be "the score on a 0-10 numerical pain rating scale reported by the patient."
A statement of the predicted relationship between two or more variables in a research study; an educated or calculated guess by the researcher.
- Example: "Increased nurse-to-patient ratios will lead to decreased patient fall rates."
Restrictions in a study that may decrease the credibility and generalizability of the research findings. These are flaws or shortcomings, perhaps due to limited resources, small sample size, or an imperfect methodology. Acknowledging limitations demonstrates honesty and a comprehensive understanding of the topic.
- Example: "The small sample size of this study (n=30) limits the generalizability of these findings to a broader population of critical care nurses."
The entire set of individuals or objects sharing common characteristics, selected for a research study.
- Target Population:
- The entire group of individuals (or objects) that the researchers are interested in and to whom they want to generalize their findings.
- Example: All pregnant women in a specific country.
- Accessible Population:
- The subgroup of the target population that is available to the researcher for a particular study.
- Example: All pregnant women attending a particular antenatal clinic during the study period.
A part or subset of the population selected to participate in the research study.
- Representative Sample:
- A sample whose characteristics closely mirror those of the population from which it is drawn. This allows for better generalization of findings.
The act, process, or technique of selecting a representative part of a population (a sample) to determine characteristics of the whole population.
- Probability Sampling:
- Selecting subjects or sampling units from a population using a random procedure, ensuring each member has a known chance of selection.
- Examples: Simple Random Sampling, Stratified Random Sampling.
- Non-Probability Sampling:
- Selecting subjects or sampling units from a population using a non-random procedure, where not every member has an equal chance of selection.
- Examples: Convenience Sampling, Purposive Sampling.
The degree of consistency or accuracy with which an instrument measures the attributes it is designed to measure. It refers to the stability and consistency of results over time or across different administrations.
- Example: A blood pressure cuff is reliable if it consistently gives similar readings when taken multiple times in a short period on the same person, assuming the person's blood pressure hasn't changed.
The degree to which an instrument truly measures what it is intended to measure. It refers to the extent to which research findings represent reality.
- Example: A questionnaire designed to measure anxiety is valid if it actually measures anxiety and not, for example, stress or depression.
The stage in research where data collection instruments (like questionnaires) are tested on a small group of people from the target population before the main study. This identifies potential problems.
- Purpose:
- To find and solve problems with the data collection instrument.
- To ensure the tools are valid, leading to reliable results.
- To check if respondents can and will provide the needed information.
- To allow the assessor to test solutions to questionnaire problems.
- Principles:
- Should mimic the actual data collection conditions as closely as possible.
- Should involve participants similar to those in the main sample.
- Careful notes should be taken on problems and potential solutions.
A smaller version of a proposed study, conducted to refine the research methodology. It uses similar subjects, settings, treatments, and data collection/analysis techniques as the main study. It aims to test the feasibility of instruments and methods.
- Example: Before a large study on a new nursing intervention, a pilot study might test the intervention on a small group of patients to ensure the procedures are clear, the data collection tools work, and the study is manageable.
The method of organizing, sorting, and scrutinizing data to answer research questions or draw meaningful conclusions. It usually follows the presentation and interpretation of research findings.
- Example: Using statistical software to compare patient recovery times between two treatment groups or reading interview transcripts to identify common themes.
The ongoing process where participants learn key facts about a study before deciding to participate, and continue to receive information throughout. Doctors, nurses, or researchers explain study details to help individuals make an informed decision.
In epidemiology, a group of individuals who share a common characteristic or experience.
- Types:
- Prospective Cohort: Studies participants from the present into the future.
- Retrospective Cohort: Studies participants based on their past records or experiences.
- Example: A group of nurses who graduated from the same university in the same year (a cohort) might be followed for 20 years to study their career progression.
When a point of view prevents impartial judgment on issues related to that viewpoint. In clinical studies, bias is controlled through methods like blinding and randomization.
- Example: If a researcher believes a certain drug is superior, they might unconsciously interpret results in a way that favors that drug.
In a randomized clinical trial, "blind" (or masked) means participants are unaware of which treatment arm (e.g., experimental drug or placebo) they are assigned to.
- Example: In a "single-blind" study, only the participants don't know their treatment. In a "double-blind" study, neither the participants nor the researchers administering the treatment know.
Any undesired actions or effects of a drug or treatment. These negative or adverse effects can include headaches, nausea, hair loss, skin irritation, or other physical problems. Experimental drugs are evaluated for both immediate and long-term side effects.


Thanks so much I have read and understood.
Very good and precised notes be blessed
Clear and precise notes Easily understood
Thanks
Mr nurses revision I would like one day to meet you face by face am from kasese
Good work
That’s wonderful, l wish we had some assignment at the end of every section in form of objectives, thanks so much for making learning easy