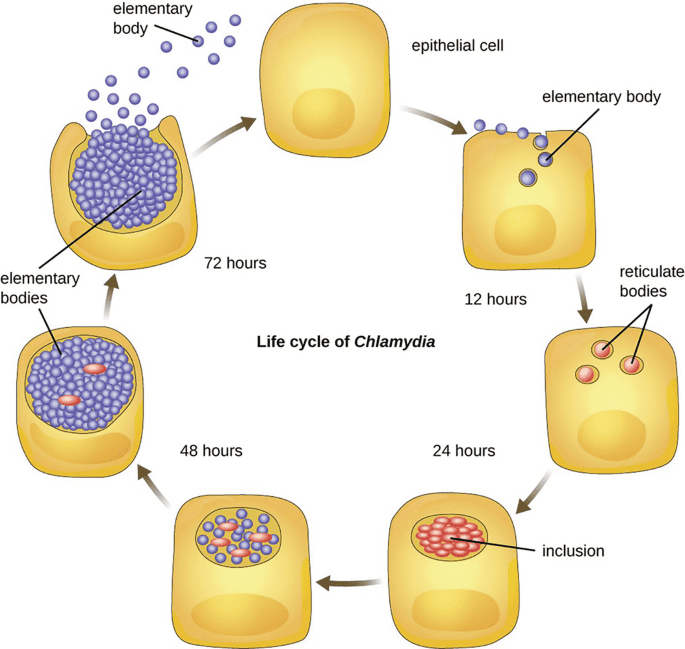
Introduction to Parasitology
Introduction to Medical Parasitology
By the conclusion of this exhaustive master guide, you will be deeply conversant with:
- The historical milestones that shaped modern medical parasitology.
- The rigorous taxonomic classification of parasites (Protozoa vs. Helminths vs. Arthropods) and their morphological features.
- The complex dynamics of Host-Parasite Relationships, including the specific types of hosts (Definitive, Intermediate, Paratenic) and the exact terminology of zoonotic transmission.
- The diverse modes of transmission, life cycles, and profound pathogenic mechanisms that parasites utilize to cause human disease.
- The sophisticated immune evasion strategies employed by parasites to survive for years within the human body.
- The comprehensive suite of laboratory diagnostic modalities, ranging from classical microscopy to modern molecular xenodiagnosis.
I. Introduction & Historical Perspective
Medical parasitology is the dedicated branch of microbiology that deals with the parasites which cause human infections, the intricate clinical diseases they produce, and the epidemiological networks that sustain them. It is broadly divided into two main, structurally distinct parts:
- Protozoology: The study of single-celled (unicellular) parasites. Though microscopic, these single cells perform all necessary physiological functions for survival, replication, and pathogenesis.
- Helminthology: The study of multi-cellular parasitic worms (metazoa). These organisms possess complex organ systems, including primitive nervous, excretory, and reproductive tracts.
Historical Perspective: The Pioneers of Parasitology
The field of parasitology has evolved through centuries of meticulous microscopic observation and epidemiological detective work.
- Antonie von Leeuwenhoek (1681): The pioneer Dutch microscopist and "Father of Microbiology" who first introduced the single-lens microscope. He made the seminal observation of Giardia lamblia trophozoites swimming in his very own diarrheal stools, providing the first description of a human protozoan parasite!
- Louis Pasteur (1870): Famous for germ theory, Pasteur also first published a scientific study on a protozoal disease leading to its control and prevention. He investigated pébrine, an epidemic silkworm disease caused by a microsporidian parasite in Southern Europe, saving the French silk industry.
- Patrick Manson (1878): While working in China, Manson made a seminal discovery about the role of Culex mosquitoes in transmitting the microfilariae of Wuchereria bancrofti (the cause of elephantiasis).
Significance: This was the very first concrete evidence of vector-borne disease transmission in medical history, fundamentally changing how we understand disease spread. - Alphonse Laveran (1880): A French army surgeon who discovered the malarial parasite (Plasmodium) inside the red blood cells of a patient in Algeria. He won the Nobel Prize for this discovery.
- Ronald Ross (1897): Building on Manson's and Laveran's work, Ross conclusively showed the transmission of malaria by Anopheles mosquitoes while working in Secunderabad and Calcutta, India, proving the complete transmission cycle.
Modern Era: By the mid-twentieth century, with dramatic, rapid advances in broad-spectrum antibiotics, antiparasitic chemotherapy, insecticides (like DDT), and improved global sanitation, all infectious diseases seemed amenable to control. However, due to emerging drug resistance and global travel, parasitic diseases remain a massive global health burden today.
II. Defining Parasites & Classification
In the strictest biological sense, parasites are living organisms which depend entirely on a living host for their nourishment, shelter, and survival. They multiply or undergo essential developmental stages within the host, usually at the host's physical or metabolic expense.
The term 'parasite' in human medicine is conventionally restricted to Protozoa (unicellular organisms belonging to Kingdom Protista) and Helminths (multicellular organisms belonging to Kingdom Animalia).
Taxonomic Classification of Parasites
Classified heavily based on their primary organs of locomotion:
- Amoebae (Sarcodina): Move using pseudopodia (temporary projections of the cytoplasm, or "false feet").
Examples: Entamoeba histolytica (causes amoebic dysentery), Naegleria fowleri (the brain-eating amoeba). - Flagellates (Mastigophora): Move using rapid, whip-like flagella.
Examples: Giardia lamblia (intestinal), Trichomonas vaginalis (urogenital), Leishmania and Trypanosoma (blood and tissue). - Sporozoa (Apicomplexa): Possess an "apical complex" used to penetrate host cells. They have non-motile adult stages and are strictly obligate intracellular parasites.
Examples: Plasmodium (Malaria), Babesia, Toxoplasma gondii, Cryptosporidium. - Ciliates (Ciliophora): Move using thousands of tiny, hair-like cilia covering their surface.
Example: Balantidium coli (the largest protozoan and the only ciliate known to be pathogenic to humans). - Microspora: Very small, spore-forming, obligate intracellular parasites that extrude a unique polar tube to inject infective material into host cells (e.g., Microsporidia, common in HIV/AIDS patients).
Complex worms, classified into three main groups based on body shape:
- Nematodes (Roundworms): Unsegmented, cylindrical, elongated worms with separate sexes (dioecious). Complete digestive tracts.
Examples: Ascaris lumbricoides (giant roundworm), Ancylostoma duodenale (hookworm), Trichuris trichiura (whipworm). - Cestodes (Tapeworms): Segmented, flat, ribbon-like worms lacking a digestive tract (they absorb nutrients through their skin). They are hermaphroditic. Consist of a head (scolex) and body segments (proglottids).
Examples: Taenia solium (pork tapeworm), Echinococcus (hydatid worm). - Trematodes (Flukes): Leaf-shaped, unsegmented flatworms with incomplete digestive tracts. Most are hermaphroditic (except blood flukes).
Examples: Fasciola hepatica (liver fluke), Schistosoma spp. (blood flukes).
Members of the Phylum Arthropoda (Class Insecta, Arachnida, Crustacea) possess jointed appendages and exoskeletons. In medical parasitology, they serve largely as vital vectors (transmitters) rather than the primary infectious agents themselves, though some cause direct disease (e.g., Sarcoptes scabiei causing scabies).
The Helminth Worms
To easily remember the distinct morphological shapes of the multicellular worms, use: "Nema-Round, Trema-Leaf, Cesto-Tape".
- Nematodes = Round, cylindrical and smooth like a garden hose.
- Trematodes = Flukes, shaped flat like a Leaf.
- Cestodes = Tapeworms, extremely long, flat, and segmented like a measuring Tape.
III. Classification by Habitat & Dependence
Classification by Anatomical Location:
- Ectoparasite: Inhabits only the external body surface of the host without deeply penetrating the underlying tissue.
Examples: Lice (Pediculus), ticks, fleas, and mites. The specific term infestation is often employed for parasitization with ectoparasites (e.g., you are clinically "infested" with lice, not "infected"). - Endoparasite: Lives internally within the body, tissues, or organs of the host. This inherently causes an infection. The vast majority of protozoan and helminthic parasites causing human disease are endoparasites.
- Free-living parasite: Refers to nonparasitic stages of active existence which live entirely independent of the host in the external environment.
Extra Example: The active amoebic and cystic stages of the brain-eating amoeba, Naegleria fowleri, living freely in warm freshwater lakes until they accidentally enter a human nose.
Classification by Metabolic Dependence:
- Obligate parasite: Completely metabolically dependent; it absolutely cannot exist, complete its life cycle, or reproduce without a suitable host.
Examples: Toxoplasma gondii, Plasmodium spp. - Facultative parasite: A highly adaptable organism which may either live as a parasitic form inside a host OR as a completely free-living form in the soil.
Extra Example: Strongyloides stercoralis, a nematode that can multiply indefinitely in the soil or infect humans. - Accidental parasites: Infect an unusual host that is not part of their normal evolutionary life cycle.
Example: Echinococcus granulosus normally cycles strictly between dogs and sheep. It infects man only accidentally through close contact with dogs, giving rise to dead-end hydatid cysts. - Aberrant (Wandering) parasites: Infect a host where they wander aimlessly because they cannot develop further or find their correct anatomical target.
Example: Toxocara canis (the dog roundworm) infecting humans. The larvae hatch in the human gut, get "lost," and migrate randomly through the human liver, lungs, or eyes, causing a severe condition known as Visceral Larva Migrans.
IV. Host Types & Relationships
A host is an organism which harbors the parasite, provides essential nourishment and shelter, and is relatively much larger than the parasite itself. Identifying the specific type of host is crucial for understanding disease epidemiology and lifecycle interruption.
- Definitive Host: The ultimate host in which the adult parasite lives and undergoes sexual reproduction. In the majority of human parasitic infections, man is the definitive host (e.g., Wuchereria bancrofti/filaria, Ascaris/roundworm, hookworm).
- Intermediate Host: The host in which the larval stage lives, or where asexual multiplication takes place. Some highly complex parasites require 2 completely different intermediate hosts to complete consecutive larval stages (known precisely as first and second intermediate hosts, e.g., Diphyllobothrium latum uses a crustacean then a fish).
Note: Man acts as an intermediate/secondary host for: Plasmodium spp., Babesia spp., Toxoplasma gondii, Echinococcus granulosus, Taenia solium (when causing cysticercosis), and Spirometra spp. - Paratenic (Transport) Host: A host in which the larval stage of the parasite enters and remains viable, but undergoes absolutely NO further developmental stages. It merely acts as a living transport vehicle to move the parasite geographically or up the food chain to reach the definitive host.
Extra Example: Humans eating undercooked frogs or fish containing Gnathostoma spinigerum larvae. - Reservoir Host: In an endemic geographical area, a parasitic infection is continuously kept alive by the presence of an animal host which harbors the exact same parasite and acts as an important, continuous source of infection to other susceptible hosts (including humans).
Example: The domestic dog is the primary reservoir host for Leishmania donovani and hydatid disease. - Accidental Host: The host in which the parasite is not usually found and from which it is unlikely to transmit further (e.g., man is a dead-end accidental host for cystic echinococcosis).
💡 High-Yield Board Exam Trap: Malaria's Definitive Host
Medical students constantly get this wrong! In Malaria (Plasmodium infection), the human feels the severe clinical symptoms and intuitively seems like the "main" or definitive host. This is entirely false!
The Female Anopheles Mosquito is the Definitive Host. Why? Because the strict biological definition of a definitive host is where sexual reproduction occurs. In malaria, the fusion of male and female gametocytes (macrogametes and microgametes) occurs exclusively inside the mosquito's stomach! The human is strictly the Intermediate Host, because only massive asexual replication (schizogony) occurs in the human liver and red blood cells.
V. Zoonosis
The word zoonosis was originally introduced by the legendary pathologist Rudolf Virchow in 1880 to include diseases shared in nature by man and animals. In 1959, the World Health Organization (WHO) refined the definition of zoonosis as "those diseases and infections which are naturally transmitted between vertebrate animals and man."
Types of Zoonoses:
- Protozoal zoonoses: e.g., toxoplasmosis (from cats), leishmaniasis (from dogs/rodents), balantidiasis (from pigs), and cryptosporidiosis (from cattle).
- Helminthic zoonoses: e.g., hydatid disease (from dogs/sheep), taeniasis (from pigs/cattle), trichinellosis (from bears/pigs).
Direction of Transmission:
- Anthropozoonoses: Infections transmitted fundamentally to man from lower vertebrate animals. (e.g., cystic echinococcosis from dogs to humans, or rabies from bats to humans).
- Zooanthroponoses: Infections transmitted in reverse, from man to lower vertebrate animals. (e.g., human tuberculosis transmitted to susceptible cattle or domestic dogs).
- Amphixenoses: (Extra detail) Infections maintained equally in both humans and lower animals, transmitted freely in both directions (e.g., Trypanosoma cruzi / Chagas disease).
VI. Host-Parasite Relationships
When two distinct organisms live in close, prolonged physical association, the biological relationship is broadly termed symbiosis. This umbrella term breaks down into three specific medical categories based on who benefits and who suffers:
- Symbiosis (Mutualism): Both the host and the parasite are strictly dependent upon each other. They both benefit, and absolutely none of them suffers any harm from the association.
Extra Example: Termites and their gut flagellates; the flagellates get a home, and the termite gets enzymes to digest wood. - Commensalism: Only the parasite derives active benefit (food, shelter) from the association, but does so entirely without causing any physiological injury or harm to the host. The host is neutral. A commensal is generally capable of living an independent life also.
Example: Entamoeba coli living harmlessly in the human colon, eating gut bacteria. - Parasitism: The parasite derives immense benefits (nutrition, immune protection, reproduction sites) and the host is always harmed (tissue damage, nutrient theft, immune exhaustion) due to the association. The parasite cannot live an independent life.
VII. Life Cycle of Parasites
A parasite's life cycle encompasses all developmental stages from its inception to maturity and reproduction. Cycles range from startlingly simple to incredibly complex.
1. Direct Life Cycle:
Occurs when a parasite requires only a single host (typically humans) to complete its entire sexual and asexual development. Transmission is often direct via the fecal-oral route or direct skin penetration from contaminated soil.
- Protozoa with direct life cycles: Entamoeba histolytica, Giardia lamblia, Trichomonas vaginalis, Balantidium coli, Cryptosporidium parvum, Cyclospora, Isospora belli, Microsporidia.
- Helminths with direct life cycles: Ascaris lumbricoides (ingested eggs hatch in gut, migrate to lungs to molt, and return to gut), Enterobius vermicularis (pinworm), Trichuris trichiura (whipworm), Ancylostoma duodenale & Necator americanus (hookworms).
Extra Note: Hymenolepis nana is incredibly unique as it is the ONLY human tapeworm that is fully capable of completing a direct life cycle without an intermediate host!
2. Indirect Life Cycle:
Occurs when a parasite mandates 2 or more species of host (e.g., a human and a snail, or a human and an insect vector) to complete its required developmental stages.
- Protozoa with indirect life cycles:
- Plasmodium spp. (Definitive: Mosquito | Intermediate: Man)
- Babesia (Definitive: Tick | Intermediate: Man)
- Leishmania (Definitive: Man/Dog | Intermediate: Sandfly)
- Trypanosoma brucei (Definitive: Man | Intermediate: Tsetse fly)
- Trypanosoma cruzi (Definitive: Man | Intermediate: Triatomine/Kissing bug)
- Toxoplasma gondii (Definitive: Feline/Cat | Intermediate: Man, rodents, birds)
- Cestodes with indirect life cycles: Taenia solium (Pig), Taenia saginata (Cattle), Echinococcus (Dog/Man), Diphyllobothrium latum (Copepod and Fish).
- Trematodes with indirect life cycles: Fasciola hepatica (requires an aquatic Snail and then encysts on aquatic plants), Schistosoma spp. (requires a specific freshwater Snail).
- Nematodes with indirect life cycles: Wuchereria bancrofti (requires a Mosquito vector), Dracunculus medinensis (requires an aquatic Cyclops/water flea).
VIII. Sources and Modes of Infection
A. Sources of Infection:
Where does the parasite physically come from before it enters the human body?
- Contaminated soil and water:
- Soil heavily polluted with embryonated, hardy eggs (roundworm, whipworm) may be ingested via unwashed hands or raw vegetables.
- Infective filariform larvae lurking in damp soil may actively penetrate intact, exposed skin (hookworm, Strongyloides).
- Infective protozoan cysts present in drinking water may be ingested (amoeba, Giardia).
- Water containing the microscopic intermediate host may be swallowed completely (e.g., swallowing a Cyclops containing a guineaworm larva).
- Infected free-swimming larvae in water may directly penetrate exposed skin of swimmers/waders (cercariae of schistosomes).
- Free-living parasites in water may forcefully enter vulnerable anatomical sites (Naegleria entering the nose, crossing the cribriform plate into the brain).
- Food:
- Ingestion of contaminated food or unwashed vegetables containing the infective stage (amoebic cysts, Toxoplasma oocysts from cat feces, Echinococcus eggs).
- Ingestion of raw, under-cooked, or smoked meat harboring encysted infective larvae (e.g., eating "measly pork" containing Cysticercus cellulosae, the larval stage of Taenia solium, or eating raw bear meat containing Trichinella spiralis).
- Insect Vectors:
- Biological (True) Vectors: The vector is absolutely critical. It not only assists in transfer, but the parasite undergoes mandatory developmental stages or massive multiplication inside the vector's body.
Examples: Mosquito (Malaria, filariasis), Sandflies (Kala-azar), Tsetse flies (Sleeping sickness), Reduviid bugs (Chagas’ disease), Ticks (Babesiosis, Lyme). - Mechanical Vectors: The vector assists purely in physical, passive transfer and is NOT essential in the life cycle (e.g., passive transport of cysts on the hairy legs or mouthparts of a bug).
Example: A housefly landing on human feces and then landing on your food (transmitting amoebiasis or typhoid).
- Biological (True) Vectors: The vector is absolutely critical. It not only assists in transfer, but the parasite undergoes mandatory developmental stages or massive multiplication inside the vector's body.
- Animals:
- Domestic: Cow (T. saginata), Pig (T. solium), Dog (Echinococcus), Cat (Toxoplasma).
- Wild: Game animals/antelope (African trypanosomiasis), wild felines (Paragonimus), freshwater fish (fish tapeworm D. latum), molluscs/snails (liver flukes), copepods (guineaworm).
- Other Persons & Self (Autoinfection):
- Other persons may be asymptomatic chronic carriers or transmit infections vertically (mother-to-child congenital infections).
- Autoinfection: A vicious cycle where a patient re-infects themselves. Finger-to-mouth transmission after scratching (e.g., pinworm Enterobius) or internal reinfection where larvae hatch and penetrate the gut wall without ever leaving the body (e.g., hyperinfection syndrome in Strongyloides stercoralis). Other highly autoinfective parasites include Hymenolepis nana, Taenia solium, Capillaria, and Cryptosporidium.
B. Modes of Infection:
How exactly does the parasite breach the body's defenses?
- Oral transmission: The absolute most common method globally. Ingestion of cysts, embryonated eggs, or encapsulated larval forms via contaminated food, water, soiled fingers, or dirty fomites (doorknobs, toys).
- Skin transmission: Hookworm larvae actively penetrate intact skin when walking barefoot on contaminated soil; Schistosome cercariae secrete enzymes to dissolve and penetrate skin when swimming in contaminated lakes.
- Vector transmission: Transmitted by insect bite (via saliva injection like Malaria, or via rubbing infected feces into the bite wound like Chagas disease).
- Direct transmission: Person-to-person physical contact (e.g., deep kissing transmitting gingival amoebae, or sexual intercourse explicitly transmitting the flagellate Trichomonas vaginalis).
- Vertical transmission: Transplacental transmission from an infected mother directly to the developing fetus (e.g., Congenital Malaria, Congenital Toxoplasmosis causing severe brain damage).
- Iatrogenic transmission: Medically induced, accidental transmission via contaminated blood transfusions, sharing of contaminated IV drug needles, or infected organ transplantation (e.g., transfusion-induced malaria or Chagas disease).
❓ Applied Clinical Question: Pork vs. Beef Tapeworm
Case: A patient enjoys eating rare steaks and undercooked pork chops. They present with abdominal pain, and stool analysis reveals tapeworm proglottids. Why is a Taenia solium (pork) infection considered vastly more dangerous to the human than a Taenia saginata (beef) infection?
Answer: Humans are the natural definitive host for both worms (we get the giant adult tapeworm living in our gut simply by eating undercooked meat containing larval cysts).
However, Taenia solium is incredibly dangerous because it is capable of Autoinfection! If a human accidentally swallows T. solium eggs from their own feces (or someone else's) via the fecal-oral route, the human's body acts as the intermediate host (like the pig). The eggs hatch in the stomach, the larvae cross the gut wall, enter the bloodstream, and migrate into the brain tissue, encysting there to cause Neurocysticercosis (the leading parasitic cause of severe seizures, epilepsy, and death worldwide). T. saginata (beef) eggs cannot infect humans in this way.
IX. Pathogenesis of Parasitic Infections
Parasitic infections do not always result in immediate, fulminant illness. They may remain completely inapparent (asymptomatic carriers) or give rise to severe clinical disease. A few highly adapted organisms, such as Entamoeba histolytica, may even live as surface commensals in the gut lumen for years without invading the tissue until triggered. When clinical infection is produced, it may take many forms—acute, subacute, chronic, latent, or recurrent.
Pathogenic Mechanisms (How parasites cause damage):
- Lytic Necrosis: Toxic enzymes produced by some parasites actively dissolve and destroy host tissue to feed.
- Example: E. histolytica secretes potent pore-forming peptides called "amoebapores" and histolysins that rapidly lyse intestinal epithelial cells, producing the classic, deep "flask-shaped amoebic ulcers" in the colon.
- Trauma: Direct physical, mechanical damage to host tissues.
- Example: The aggressive attachment of hookworms (via sharp cutting plates in N. americanus or teeth in A. duodenale) onto the jejunal mucosa leads to traumatic maceration of the villi and continuous, oozing bleeding at the site of attachment, ultimately causing profound iron-deficiency anemia in the patient.
- Allergic Manifestations: Severe clinical illness caused entirely by the host's own extreme, hyperactive immune response to the foreign parasite proteins.
- Example 1: Eosinophilic pneumonia (Löffler's syndrome) in Ascaris infection, which occurs precisely as millions of microscopic larvae migrate through and burst out of the delicate lung capillaries into the alveoli.
- Example 2: Severe, immediate, and potentially life-threatening anaphylactic shock upon the accidental rupture of a massive hydatid cyst (Echinococcus), suddenly dumping huge amounts of foreign, highly antigenic hydatid fluid directly into the bloodstream.
- Physical Obstruction: Complete or partial blockage of vital hollow organs, ducts, or blood vessels by the sheer physical mass and volume of the parasite.
- Example 1: Tangled, knotted masses of dozens of giant roundworms (Ascaris) causing fatal mechanical intestinal obstruction at the ileocecal valve, or migrating into and physically blocking the tiny common bile duct.
- Example 2: Plasmodium falciparum radically alters the surface of infected red blood cells, deploying sticky proteins (PfEMP1) that cause the cells to clump together (rosetting) and adhere to blood vessel walls (cytoadherence). This produces a fatal physical blockage of tiny brain capillaries, resulting in Cerebral Malaria and coma.
- Inflammatory Reaction: Chronic, long-term irritation by adult worms or trapped eggs triggering endless inflammation and consequent massive fibrosis/scarring.
- Example 1: Chronic lymphadenitis and severe lymphatic fibrosis induced by adult Wuchereria worms blocking lymph vessels, ultimately leading to massive limb swelling (Elephantiasis).
- Example 2: Hundreds of thousands of trapped Schistosoma eggs inciting intense granulomatous inflammation, leading to "pipe-stem" fibrosis of the liver or massive fibrosis of the urinary bladder wall.
- Neoplasia (Cancer): A few unique chronic parasitic infections have been conclusively shown to continuously damage DNA and induce cellular malignancy over decades.
- Example 1: The Asian liver fluke, Clonorchis sinensis, lives in the biliary tract and directly induces bile duct carcinoma (Cholangiocarcinoma).
- Example 2: S. haematobium chronic bladder wall irritation causes squamous cell metaplasia, eventually leading to highly fatal squamous cell carcinoma of the urinary bladder.
X. Immunity in Parasitic Infections
Exactly like bacterial and viral infectious agents, parasites elicit both robust humoral (antibody-mediated) and cellular (T-cell and macrophage) immune responses in the host. However, immunological protection and clearance against parasitic infections is generally much less efficient and less sterilizing than it is against bacterial or viral infections.
Why is the immune response so weak against parasites?
- Size & Antigenic Complexity: Compared to simple bacteria and viruses, parasites are enormously larger (some tapeworms are 30 feet long!) and structurally/antigenically infinitely more complex. The immune system struggles to focus a coordinated attack on the correct protective surface antigens because there are thousands of distinct proteins.
- Intracellular / Cavity Location: Many protozoan parasites are strictly intracellular (hiding safely inside host cells, like Plasmodium hiding inside red blood cells which lack MHC-I molecules to alert the immune system, or Leishmania happily surviving inside the very macrophages meant to destroy them). Several protozoa and massive helminths live completely free inside external body cavities (like the gut lumen) where immune cells (WBCs) and circulating antibodies have almost zero access.
Premunition (Infection-Immunity):
Unlike viral infections that provide durable, lifelong sterilizing immunity after the virus is cleared (like a natural measles infection), once a parasitic infection is completely eliminated, the host frequently becomes fully susceptible to a brand new reinfection immediately.
Premunition is a highly unique type of immunity that depends entirely on the continued, ongoing presence of a residual, low-level, asymptomatic parasite population in the host. As long as a few parasites remain, the host's immune system is constantly stimulated and remains immune to severe new super-infections. Once the host is fully cured with drugs, the protective immunity vanishes instantly. (This is heavily observed in Malaria in endemic regions).
Immunoglobulin & Cellular Responses:
- IgM vs IgG: Antibodies from different immunoglobulin classes are heavily produced. Selective serological tests for IgM are incredibly helpful diagnostically in differentiating an active, current/acute infection from an old, fully resolved past infection (which only shows IgG memory antibodies).
- IgE & Eosinophilia: An absolutely massive, exaggerated IgE response is the hallmark diagnostic signature of tissue-invasive helminthiasis (worm infections). A characteristic cellular response in these infections is profound eosinophilia (both localized in the tissues and systemically elevated in the complete blood count). Eosinophils are recruited by IL-5, bind to the IgE coating the worm, and literally degranulate, dumping toxic Major Basic Protein directly onto the exterior of migrating larvae (like schistosomula) to kill them.
💡 High-Yield Board Concept: Helminths vs. Eosinophils
Why do ONLY tissue-invading Helminths cause massive, soaring Eosinophilia, while intestinal Protozoa (like Giardia or Entamoeba) generally do not?
Because eosinophils are a specialized evolutionary weapon designed explicitly to attack organisms that are physically too large to be phagocytized (eaten by macrophages or neutrophils). A macrophage can easily engulf and eat a tiny single-celled protozoan, but it absolutely cannot eat a 10-inch long living Ascaris worm. Therefore, the immune system uses IgE to flag the worm, triggering Eosinophils to swarm the massive target and destroy it externally via Antibody-Dependent Cellular Cytotoxicity (ADCC)!
XI. Immune Evasion Mechanisms
All animal pathogens, including complex parasitic protozoa and worms, have evolved highly effective, almost science-fiction-like mechanisms to successfully avoid, suppress, or misdirect elimination by the host defense system, allowing them to survive for decades inside a human.
| Parasite Escape Mechanism | Specific Examples & Pathophysiology |
|---|---|
| Intracellular Habitat (Hiding inside host cells) |
Malarial parasite (Plasmodium hides inside mature RBCs which lack immune-alerting MHC molecules). Leishmania purposely allows itself to be eaten by Macrophages, but secretes enzymes to block the lethal phagolysosome fusion, turning the immune cell into its home! |
| Encystment (Forming a tough, impenetrable biological shell) |
Toxoplasma gondii forms bradyzoite tissue cysts in the brain; Trypanosoma cruzi forms amastigote nests in heart muscle, walled off from immune surveillance. |
| Resistance to microbial phagocytosis | Leishmania surface lipophosphoglycan (LPG) physically protects it from complement-mediated lysis and oxidative bursts. |
| Masking of Antigens / Molecular Mimicry (Coating themselves in host molecules to look like "self") |
Adult Schistosomes are masters of disguise. As they swim in the human bloodstream, they literally steal and coat their entire outer skin (tegument) with human ABO blood group antigens and MHC proteins, making them completely invisible to passing T-cells! |
| Variation of Antigen (Constantly changing their surface coats) |
Trypanosoma brucei (Sleeping Sickness) possesses over 1,000 genes for Variable Surface Glycoproteins (VSGs). Just as the human body makes antibodies against coat A, the parasite sheds it and switches to coat B, perpetually staying one step ahead of the immune system. Also seen in Plasmodium and Giardia. |
| Suppression of immune response | Trichinella spiralis and Schistosoma mansoni secrete specific immunosuppressive cytokines that actively shut down local T-cell responses. The malarial parasite actively degrades memory B-cells. |
| Interference by Polyclonal Activation (Distracting the immune system) |
African Trypanosomes secrete chemicals that force the host's B-cells to randomly, massively activate. The host produces huge amounts of useless antibodies (hypergammaglobulinemia) that do not target the parasite, exhausting the immune system. |
| Continuous turnover and release of surface antigens (Shedding their skin as a decoy) |
Schistosomes rapidly shed their surface antigens into the blood. Host antibodies attack the floating debris (the decoys) rather than attacking the actual living worm. |
*Note on Immunodeficiency: Some severe infections produce massive, acquired immunodeficiency simply due to the extensive, physical destruction of the reticuloendothelial system (e.g., Visceral Leishmaniasis / Kala-azar destroying the spleen and bone marrow).
Vaccination Reality: No highly effective, sterilizing vaccine for humans has so far been successfully deployed worldwide against complex parasites directly due to their massive, complex life cycles, multiple developmental stages, and extreme, rapid antigenic variation. However, massive global progress is currently being made in identifying protective antigens in malaria (e.g., the RTS,S and R21 vaccines) for eventual deployment.
XII. Laboratory Diagnosis of Parasites
Most parasitic infections simply cannot be conclusively diagnosed based on broad clinical features and physical examination alone (fever, chills, and diarrhea are exceedingly generic). Accurate laboratory diagnosis is absolutely paramount for prescribing highly specific, toxic antiparasitic drugs, and relies on multiple investigative modalities.
1. Microscopy (Direct Visualization):
The definitive, indisputable gold standard. Seeing the physical parasite proves the infection. Various bodily specimens are collected and examined depending strictly on the parasite's specific anatomical lifecycle.
- Stool Examination (Ova and Parasites - O&P): Vital for intestinal infections. Examined via wet mounts (saline/iodine) or permanent stains (Trichrome).
- Cysts/Trophozoites: Entamoeba histolytica, Giardia lamblia, Balantidium coli, Isospora, Cyclospora, Cryptosporidium (requires modified acid-fast stain).
- Eggs (Ova): Cestodes (Taenia, H. nana), Trematodes (Schistosoma mansoni/japonicum, Fasciola, Clonorchis), Nematodes (Trichuris/barrel-shaped, Enterobius, Ascaris, Hookworms).
- Larvae: Strongyloides stercoralis (rhabditiform larvae).
- Adult Worms: Gross examination of expelled Ascaris, Enterobius, or segments (proglottids) of Taenia.
- Blood Examination: Vital for parasites circulating in vessels. Demonstrates morphological stages. Thick smears are used to *detect* the presence of parasites (concentrates the blood), while thin smears are used to explicitly *speciate* the parasite based on RBC morphology. Detects: Plasmodium spp., Babesia (Maltese cross), Trypanosoma, Leishmania, and circulating microfilariae (Wuchereria, Loa loa, Brugia).
- Urine Examination: Detects characteristic terminal-spined eggs of Schistosoma haematobium, motile trophozoites of Trichomonas vaginalis, and microfilaria of W. bancrofti (specifically found in chylous/milky, lipid-filled urine).
- Sputum Examination: Heavy, golden-brown eggs of the lung fluke Paragonimus westermani. Occasionally reveals actively migrating larvae of Ascaris and Strongyloides during the pulmonary phase.
- Cerebrospinal Fluid (CSF): Examined via lumbar puncture. Detects highly motile T. brucei (Sleeping Sickness), Naegleria trophozoites, Acanthamoeba, and the rat lungworm Angiostrongylus.
- Tissue and Aspirates (Biopsies):
- Muscle biopsy: Coiled Trichinella spiralis larvae inside nurse cells.
- Brain histology: Trophozoites of Naegleria / Acanthamoeba in fatal encephalitis.
- Spleen/Bone Marrow aspirate: The ultimate diagnostic test for Kala-azar, revealing Leishman-Donovan (LD) bodies (amastigotes) packed inside macrophages.
- Liver pus aspirate: E. histolytica trophozoites found at the margins of the classic thick, brown, odorless "anchovy paste" pus.
- Genital Specimens: Jerky, motile T. vaginalis trophozoites in wet mounts of vaginal/urethral discharge. Enterobius vermicularis (pinworm) eggs are classically found on the perianal skin using the clear "scotch tape test" or anal swabs early in the morning.
❓ Applied Clinical Question: The Hematuria Mystery
Case: A young man recently returned from swimming and bathing in Lake Victoria (Uganda). He complains of painless, terminal hematuria (gross, visible blood at the very end of his urine stream). You correctly order a urine microscopy.
Question: What specific parasite are you highly suspicious of, and what exact morphological feature will the lab technician report to confirm the diagnosis?
Answer: You are highly suspicious of Schistosoma haematobium (the bladder blood fluke). The lab technician will specifically look for, and report, large, distinctly oval eggs containing a sharp, characteristic Terminal Spine in the centrifuged urine sediment.
(Mnemonic/Remember: S. haematobium has a Terminal spine at the tip, whereas the intestinal S. mansoni has a prominent Lateral spine on the side!)
XIII. Advanced Diagnostic Techniques
2. Culture Methods:
While exceptionally difficult and rarely used for routine clinical diagnosis, some adaptable parasites like Leishmania (grown on NNN medium), Entamoeba, and Trypanosoma can be cultured in the specialized laboratory in various axenic (pure, parasite-only) and polyxenic (mixed with bacteria for food) media.
3. Serological Tests (Antigen vs. Antibody Detection):
- Antigen Detection (Detects active, current, ongoing infection): Highly valuable because if the antigen is present, the living bug is present.
- Galactose lectin antigen (detects E. histolytica in stool/blood).
- Giardia specific antigen 65 (stool EIA).
- WKK and rK39 recombinant antigen (Highly specific for Leishmania donovani / Kala-azar).
- HRP-2 antigen (Histidine-Rich Protein 2 - This is the exact molecular target used in modern Malaria Rapid Diagnostic Tests / RDTs to explicitly diagnose Plasmodium falciparum!).
- pLDH (Parasite lactate dehydrogenase - detects Plasmodium vivax / falciparum).
- 200 KD Ag and OG4C3 (Detects circulating Wuchereria bancrofti adult worm antigen).
- Antibody Detection (Detects historical exposure or past infection): Useful for epidemiological surveys or when parasites are hiding deep in tissues. Uses complex methods like Complement Fixation Test (CFT), Indirect Hemagglutination (IHA), Immunofluorescent Antibody (IFA), and ELISA.
4. Skin Tests (Hypersensitivity testing):
Involves injecting a highly purified, sterile parasitic antigen directly intradermally into the patient's forearm. Immediate allergic reactions (wheal and flare) occur rapidly within 30 minutes (Type I hypersensitivity). Delayed cellular hypersensitivity (erythema/induration) occurs slowly after 48 hours (Type IV hypersensitivity).
Important Eponymous Skin Tests to Memorize:
- Casoni’s test: Highly specific for Hydatid disease (Echinococcus).
- Montenegro (Leishmanin) test: Used for Kala-azar/Cutaneous Leishmaniasis (Leishmania). A positive test actually indicates strong cell-mediated immunity and recovery!
- Frenkel’s test: Used for Toxoplasmosis.
- Fairley’s test: Used for Schistosomiasis.
- Bachman intradermal test: Used for Trichinellosis.
5. Other Specialized Diagnostics:
- Molecular Diagnosis: The modern frontier. DNA probes, highly sensitive PCR (Polymerase Chain Reaction), and microarrays. Capable of detecting the DNA of a single parasite in a sample. Highly sensitive and 100% specific.
- Animal Inoculation: Injecting patient blood or tissue samples into susceptible lab animals (like mice or hamsters) to detect and amplify slow-growing Toxoplasma, Trypanosoma, and Babesia.
- Xenodiagnosis: A remarkable, historical biological test used specifically for Chagas’ disease (Trypanosoma cruzi).
How it works: Clean, sterile, lab-raised reduviid (kissing) bugs are intentionally strapped to the patient's arm and allowed to feed on the patient's blood. Weeks later, the bug's intestines and feces are dissected and examined under a microscope for the massive multiplication of T. cruzi amastigotes/epimastigotes. It essentially uses a living vector as a biological incubator to amplify a low-level, undetectable infection inside the human! - Imaging Modalities: X-ray, Ultrasound (USG), CT scans, and MRI are extensively and routinely used for visualizing massive, life-threatening, space-occupying lesions deep in tissues, such as Neurocysticercosis (calcified brain cysts mimicking Swiss cheese) and massive Hydatid cyst disease (multilocular water-lily liver cysts).
- Hematology (Complete Blood Count Clues):
- Profound Anemia specifically in heavy hookworm disease (microcytic, iron deficiency) and acute malaria (normocytic, massive hemolytic destruction).
- Massive Eosinophilia in almost all invasive helminthic infections (tissue migration phase).
- Hypergammaglobulinemia (excessive antibody production) in visceral leishmaniasis.
- Leukocytosis (high WBCs) usually indicates a secondary bacterial infection or a severe acute amoebic liver abscess.
List of References for Further Study
- Paniker’s Textbook of Medical Parasitology (Late C.K. Jayaram Paniker) – Excellent for clear, concise life cycles and regional epidemiology.
- Diagnostic Medical Parasitology (Lynne S. Garcia) – The absolute gold standard for laboratory technologists, diagnostic criteria, and bench-side microscopy.
- Manson's Tropical Diseases (Jeremy Farrar, et al.) – The ultimate, comprehensive clinical reference for global tropical medicine, zoology, and parasite-induced pathophysiology.
- Foundations of Parasitology (Larry S. Roberts, John Janovy Jr.) – A highly detailed academic textbook focusing heavily on the biological and evolutionary aspects of parasitic organisms.
- CDC (Centers for Disease Control and Prevention) DPDx – The online Laboratory Identification of Parasitic Diseases of Public Health Concern database.
Quick Quiz
Parasitology Quiz
Microbiology - mobile-friendly and focused practice.
Privacy: Your details are used only for quiz tracking and certificates.
Parasitology Quiz
Microbiology
Preparing questions...
Choose your answer and keep your streak alive.
Great effort.
Here is your quick performance summary.
Introduction to Parasitology Read More »