Bacterial Growth, Genetics, and Structure
Microbiology Foundations: Bacterial Growth, Genetics, and Structure
Before a bacterium can divide and cause an infection, it needs the right fuel and environment. Think of bacteria as microscopic factories; they need raw materials (nutrition) and ideal factory conditions (environment). Understanding these mechanisms is the fundamental basis of targeted antibiotic therapy and infectious disease management.
1. Bacterial Growth & Nutritional Requirements
A. Nutrient Requirements (The "Raw Materials")
Just like human cells, bacterial cells are highly complex and require specific building blocks to construct their membranes, DNA, and proteins.
- Water: Essential for all biochemical reactions. It is the universal solvent in which all intracellular metabolic processes occur.
- Carbon Source (C): The backbone of all living molecules (carbohydrates, lipids, proteins). Bacteria are often classified by how they get carbon (e.g., heterotrophs get it from organic compounds like glucose; autotrophs get it from CO2).
- Nitrogen Source (N): Crucial for building amino acids (which make up proteins) and nucleic acids (which make up DNA/RNA).
- Inorganic Salts, Sulfur (S), & Phosphorus (P):
- Phosphorus is needed to synthesize ATP (energy currency) and the phospholipid bilayer of the cell membrane.
- Sulfur is strictly needed for certain sulfur-containing amino acids (like cysteine and methionine) which hold proteins together via disulfide bonds.
- Growth Factors: Essential vitamins and amino acids that the bacteria cannot synthesize on their own. If the environment lacks these, the bacteria cannot survive.
B. Environmental Factors (The "Factory Conditions")
- Temperature: Most human pathogens grow best at 37°C (normal human body temperature). These are known as mesophiles. (Clinical Note: This is why the human body generates a fever—it raises the temperature above 37°C to make the environment uncomfortably hot and hostile for the invading bacteria!)
- Gas (Oxygen): Determines if they can breathe in air or if air is toxic to them (detailed in the next section).
- pH: Most bacteria prefer a neutral pH (around 7.0), though some have adapted to survive extreme acid.
Example: Helicobacter pylori in the stomach survives the highly acidic gastric juice (pH ~2.0) by secreting an enzyme called urease, which creates a neutralizing "cloud" of ammonia around the bacteria. - Osmotic Pressure: Salt and sugar concentrations in the environment.
Clinical Example: High salt environments usually pull water out of bacteria, killing them (which is why curing meat with salt prevents rotting). However, Staphylococcus aureus is a "halophile" (salt-lover) and can easily survive on the salty surface of human skin, making it a major cause of surgical wound infections.
2. Oxygen Requirements: Aerobic vs. Anaerobic Bacteria
A patient steps on a rusty nail. The deep puncture wound closes over quickly, trapping bacteria inside with no oxygen. This is the perfect, deadly environment for an obligate anaerobe (like Clostridium tetani) to thrive and cause tetanus. Understanding oxygen requirements tells you exactly where an infection can survive in the human body!
Oxygen is highly reactive. When metabolized, it creates deadly byproducts called Reactive Oxygen Species (ROS), such as superoxide radicals (O2-) and hydrogen peroxide (H2O2). To survive in oxygen, a bacteria MUST have specific enzyme "shields" (Catalase and Superoxide Dismutase - SOD) to neutralize these toxins.
| Type of Bacterium | Effect of Oxygen | Growth Pattern in a Tube | Enzyme Status (The "Shields") | Classic Clinical Examples |
|---|---|---|---|---|
| Obligate Aerobes | Only aerobic growth; O2 is strictly required. | Growth occurs only at the very top of the tube where O2 is highest. | Have Catalase and Superoxide Dismutase (SOD) to neutralize toxic oxygen radicals. | Mycobacterium tuberculosis (This is why TB infections classically target the APEX of the lungs, where oxygen concentration is highest!) |
| Facultative Anaerobes | Adaptable! Both aerobic and anaerobic growth. Greater growth with O2, but can survive without it. | Growth is best at the top, but occurs throughout the entire tube. | Have Catalase and SOD to neutralize toxic oxygen. | Escherichia coli (E. coli) and Staphylococcus aureus. |
| Obligate Anaerobes | Only anaerobic growth. Oxygen is highly toxic/lethal. | Growth occurs only at the very bottom of the tube where there is zero O2. | Lacks enzymes to neutralize harmful forms of O2. | Clostridium tetani, Bacteroides fragilis (Found deep in the gut). |
| Aerotolerant Anaerobes | Only anaerobic growth, but it can continue growing in the presence of O2. | Growth occurs evenly throughout the entire tube; O2 has no effect. | Presence of one enzyme (SOD) allows them to partially tolerate O2. | Lactobacillus and Streptococcus pyogenes. |
| Microaerophiles | Only aerobic growth, but strictly requires LOW concentrations of oxygen. | Growth occurs right in the middle of the tube (where O2 is low but not zero). | Produce lethal amounts of toxic oxygen if exposed to normal atmospheric air. | Helicobacter pylori and Campylobacter jejuni. |
3. The Bacterial Growth Curve
When bacteria invade a host or are put in a culture tube, they follow a predictable, 4-stage life cycle. Exam Trap: Know exactly what happens in the Log phase vs. Stationary phase!
The "Prep" Phase
Bacteria are sensing their environment, gathering nutrients, and turning on specific enzymes needed to digest local food sources. There is NO increase in the number of living bacterial cells during this phase.
The "Population Boom"
There is an exponential increase in the number of living cells. The bacteria are replicating at maximum speed.
Clinical Pearl: This is when bacteria are rapidly building new cell walls and dividing. Therefore, this is the exact phase where antibiotics that target cell wall synthesis (like Penicillin or Cephalosporins) are most spectacularly effective!
The "Plateau"
Nutrients are running out, and toxic metabolic waste is building up. The rate of cell division exactly equals the rate of cell death. (Deep Dive: In this phase, bacteria like Clostridium and Bacillus realize they are starving and will trigger the formation of Endospores to survive the upcoming famine). Because cell wall synthesis slows down drastically here, Penicillin becomes much less effective against bacteria in an abscess (which are usually in the stationary phase).
The "Collapse"
There is an exponential decrease in the number of living cells due to complete nutrient depletion and a lethal overload of toxic waste.
4. Fastidious Bacteria (The "Picky Eaters")
Definition: Fastidious microorganisms are extremely difficult to grow in the laboratory because they have highly complex or restricted nutritional/environmental requirements (specific temp, pH, O2, special nutrients). They will simply die if these stringent needs aren't met.
Exam Tip: Memorize these classic examples. If you see them on a test, know they require special agars (like Chocolate agar) to grow!
- Neisseria gonorrhoeae (Causes Gonorrhea). Requires highly specific "Thayer-Martin" agar (chocolate agar with antibiotics added to kill competing bacteria).
- Haemophilus influenzae (Causes respiratory infections/meningitis). Requires Chocolate Agar, which contains heated, lysed red blood cells that release strict growth factors: Factor X (hemin) and Factor V (NAD).
- Treponema pallidum (Causes Syphilis - actually so fastidious it can't be grown on standard lab media at all! It must be grown in animal testicles).
- Legionella pneumophila (Causes Legionnaires' disease).
- Bordetella pertussis (Causes Whooping cough).
- Campylobacter jejuni (Requires microaerophilic conditions).
- Helicobacter pylori (Requires microaerophilic and acidic adaptations).
- Brucella species.
- Francisella tularensis.
- Bartonella henselae (Cat scratch disease).
- Mycoplasma pneumoniae & A. pleuropneumoniae.
5. Bacterial Cell Division & Generation Time
Prokaryotic cells divide by Binary Fission. One cell elongates, duplicates its DNA, a cross-wall forms, and it splits exactly into two identical daughter cells (One into two, two into four, four into eight). Because of this, cell growth is mathematically exponential.
Generation Time (Doubling Time): The time it takes for a bacterial population to double in number. This varies wildly among species and has huge health consequences.
Specific Dividing Times to Know:
- Escherichia coli: Very fast! ~52.0 to 86.6 mins. (Clinical translation: A patient with an E. coli UTI can develop overwhelming, life-threatening sepsis overnight because the bacteria duplicate so rapidly).
- Proteus vulgaris: 28.2 mins.
- Enterococcus faecalis: 25.9 mins.
- Bacillus cereus: 49.0 mins.
- Fungi/Yeasts (Saccharomyces cerevisiae): ~99 - 107 mins.
The Extreme Exception (Mycobacterium)
- Mycobacterium smegmatis (non-pathogenic): ~3 hours.
- Pathogenic Mycobacterium (like M. tuberculosis): 18 to 24 hours!
Clinical Scenario: Because its doubling time is so slow, a patient with Tuberculosis must wait up to 4-6 weeks for lab cultures to grow a visible colony. Furthermore, because antibiotics work best on rapidly dividing cells, treatment for TB takes 6 to 9 months because the bacteria replicate so sluggishly!
6. Morphology: Size and Shape
Size
Bacteria generally range from 0.1 to 5 µm in diameter. They are much smaller than human Eukaryotic cells, but significantly larger than viruses. You need a Light Microscope to see bacteria, but a high-powered Electron Microscope to see viruses.
- Haemophilus influenzae: 0.25 × 1.2 µm (Very small)
- Escherichia coli: 1.3 × 3 µm (Average)
- Cyanobacteria: 5 × 40 µm (Giant for a bacteria)
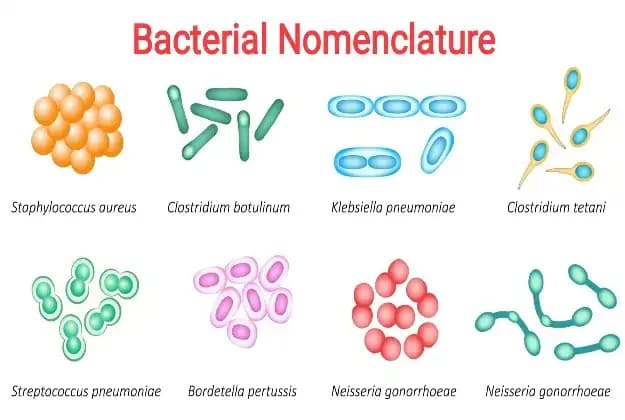
Shapes & Arrangements
Pathologists use these shapes to instantly narrow down the cause of an infection.
- Cocci (Spheres):
- Diplococci: Pairs. (e.g., Streptococcus pneumoniae, and Neisseria gonorrhoeae which is famously a Gram-negative diplococci).
- Streptococci: Chains. (Looks like a string of pearls under the microscope).
- Staphylococci: Grape-like clusters. (e.g., Staphylococcus aureus. If a doctor sees Gram-positive clusters on a blood culture, they immediately suspect Staph!).
- Tetrads (groups of 4) & Sarcina (3D cubes of 8).
- Bacilli (Rods): Coccobacillus (plump, oval rod), Chain of bacilli (Bacillus anthracis), Flagellate rods (Salmonella typhi), Spore-formers (Clostridium botulinum).
- Others:
- Vibrios: Comma-shaped (Vibrio cholerae).
- Spirilla / Spirochaetes: Corkscrew shaped (Helicobacter pylori, Treponema pallidum).
- Filamentous: Long, branching threads resembling fungal hyphae. Examples include Mycobacteria (visible on ZN/Ziehl-Neelsen Acid-Fast stain), Actinomyces, and Nocardia.
7. Eukaryotic vs. Prokaryotic Cell Comparison
Exam Trap: You must know the absolute differences. This is the entire foundation of Selective Toxicity in pharmacology! We want drugs that kill bacteria (prokaryotes) without harming human host cells (eukaryotes).
| Feature | Fungi / Human (Eukaryote) | Bacteria (Prokaryote) | Pharmacological Relevance |
|---|---|---|---|
| Nuclear Structure | True nucleus with a well-defined nuclear membrane. | No nuclear membrane (Nucleoid region only, DNA is free-floating). | Bacterial DNA replication is directly exposed in the cytoplasm, allowing drugs like Fluoroquinolones to easily target DNA gyrase. |
| Organelles | Mitochondria, Endoplasmic Reticulum, Golgi apparatus, Vacuoles. | None. Lacks all membrane-bound organelles. | Bacteria must perform cellular respiration directly on their inner cell membrane instead of inside a mitochondrion. |
| Cell Membrane | Sterols present (e.g., Cholesterol in humans, Ergosterol in fungi). | Sterols absent (Except in the unique bacteria Mycoplasma). | Antifungal drugs (like Amphotericin B or Fluconazole) specifically attack Ergosterol. They kill fungi but ignore human cholesterol and bacterial membranes! |
| Cell Wall | Polysaccharides (Glucans, mannans, chitin in fungi). NO peptidoglycan in humans/fungi. | Made of highly specific Peptidoglycan. | Penicillin destroys peptidoglycan. Because humans lack peptidoglycan entirely, Penicillin can kill billions of bacteria without bursting a single human cell! |
| Spores | Sexual and asexual reproductive spores. | Endospores (For harsh survival ONLY, NOT for reproduction). | Bacterial spores are practically indestructible and heavily complicate hospital sanitization protocols. |
8. Bacterial Genetics: DNA, Transcription, & Translation
A. Bacterial DNA
Most bacteria have a haploid genome (only one copy of their genes, meaning any mutation shows up immediately, with no backup copy to hide a lethal recessive trait). The genome is a single chromosome consisting of a circular, double-stranded DNA molecule.
Plasmids: Extra, small circular DNA pieces are also often present. Plasmids are not essential for basic life, but they carry "superpowers" like antibiotic resistance genes or toxin genes. Bacteria can pass these plasmids to each other via conjugation (like sharing a flash drive of data).
Exceptions to the Rule (Exam Favorites!):
- Linear chromosomes exist in Gram-positive Borrelia and Streptomyces.
- Agrobacterium tumefaciens (Gram-negative) has one linear AND one circular chromosome!
B. The Central Dogma & RNA Processing
DNA replicates → DNA is transcribed into mRNA → mRNA is translated by ribosomes into Protein.
Crucial Difference: In Eukaryotes (humans), DNA has "junk" sequences called introns that must be spliced (cut) out, leaving only exons. Bacteria generally do NOT have introns and do not require RNA splicing. Their mRNA is ready to be translated immediately, allowing them to adapt to new environments at lightning speed.
C. Bacterial Ribosomes (The Protein Factories)
Bacterial ribosomes are small particles composed of ribosomal protein and rRNA.
- Size: They are exactly 70S in size (composed of a 50S large subunit and a 30S small subunit).
Clinical Pharmacology Pearl: Human ribosomes are larger, at 80S (made of 60S and 40S subunits). This structural difference is heavily exploited in medicine! Drugs like Tetracyclines and Aminoglycosides specifically bind to and jam the 30S subunit. Drugs like Macrolides (Azithromycin) specifically target the 50S subunit of the 70S bacterial ribosome. Because humans don't have 70S ribosomes, these powerful drugs paralyze the bacteria without stopping human protein synthesis!
9. The Cell Envelope: Cytoplasmic Membrane & Cell Wall
The envelope is everything surrounding the cytoplasm. It consists of the Cell (Plasma) Membrane and the Cell Wall.
A. Cytoplasmic / Cell / Plasma Membrane
It is a Phospholipid bi-layer (hydrophilic heads facing out, hydrophobic tails facing in). Because bacteria lack internal organelles, this thin outer membrane has to do 5 crucial jobs:
- Selective permeability barrier: Keeps nutrients in, keeps toxins out.
- Electron transport and oxidative phosphorylation: Since bacteria have NO mitochondria, the cytochromes and dehydrogenase enzymes for the respiratory chain (ATP making) are embedded directly in the cell membrane!
- Excretion: Pumps out hydrolytic enzymes and pathogenic toxins into the host body.
- Biosynthetic function: Contains the enzymes that build the cell wall.
- Chemotactic systems: Receptors that bind attractants (food) and repellants. Example: E. coli has 20 different chemoreceptors on its membrane to navigate its environment!
*Note: Antibacterial agents like Polymyxins and Ionophores specifically destroy the bacterial cell membrane, causing the cell's contents to leak out and die.
B. The Cell Wall (Peptidoglycan)
The highly rigid layer outside the membrane. Its main functions are: Shape and cellular integrity (prevents the cell from popping due to high internal water pressure), essential role in cell division, serves as a primer for its own synthesis, and is a major site for antigen determinants.
Structure of Peptidoglycan (The Brick and Mortar):
- The Backbone (Bricks): Two alternating sugar derivatives: N-acetylmuramic acid (NAM) and N-acetylglucosamine (NAG).
- Tetrapeptide side chains: Attached to NAM.
- Peptide cross-bridges (Mortar): Link the side chains together to make a tough, chain-link fence.
D-Alanine vs. Beta-Lactam Rings
The building block of the bacterial cell wall cross-bridge usually ends in two specific amino acids: D-Alanine-D-Alanine. Beta-Lactam antibiotics (like Penicillin, Cephalosporins, Carbapenems) contain a chemical ring that looks exactly like a fake D-Alanine-D-Alanine.
The bacterial enzymes (Penicillin-Binding Proteins / PBPs) mistakenly grab the antibiotic instead of the real D-Alanine to build their wall. The wall fails to cross-link, the structure weakens, and the bacteria explosively pops under its own osmotic pressure!
(Note: MRSA is deadly because it mutated its PBP enzyme so it no longer falls for the Penicillin trick!)
Exceptions: Archaebacteria lack peptidoglycan. Some eukaryotic cells have walls made of cellulose (plants) or chitin (fungi).
10. Gram-Positive vs. Gram-Negative Envelopes
This structural difference is why Gram staining works (Gram-positives trap the purple crystal violet dye in their thick walls, while Gram-negatives lose it and stain pink), and it fundamentally decides what antibiotics a doctor will prescribe.
| Feature | Gram-Positive Bacteria | Gram-Negative Bacteria |
|---|---|---|
| Overall Structure | Simple structure. | Complex and multilayered. |
| Peptidoglycan Layer | THICK: Up to 40 sheets, comprising 50% to 90% of the cell wall materials. | THIN: Only 1 or 2 sheets, comprising just 5% to 20% of the cell wall. |
| Unique Wall Components | Contains Teichoic acid and Lipoteichoic acid interwoven like rebar in concrete, giving the wall massive structural strength and a negative charge. | Has an Outer Membrane: An extra lipid bilayer completely covering the thin peptidoglycan. |
| Periplasmic Space | Virtually none. | Present: The large gap between the inner and outer membrane. (Often contains enzymes that destroy antibiotics, like beta-lactamase!) |
| Pores/Channels | Nutrients diffuse directly through the thick wall. | Porins: Special protein channels in the outer membrane that allow small molecules to pass. (Large antibiotics like Vancomycin cannot fit through these porins, making all Gram-negatives inherently resistant to Vancomycin!) |
11. Lipopolysaccharides (LPS) - The Gram-Negative Weapon
LPS is found exclusively in the outer leaflet of the outer membrane of Gram-negative bacteria. It consists of 3 specific parts:
- Complex Lipid A (The Endotoxin): Made of fatty acids (caproic, lauric, myristic, palmitic, and stearic acids). Notice it does NOT contain glycerol.
Clinical Scenario (Septic Shock): Lipid A is highly toxic. When intact inside the bacteria, it does little harm. However, if you give a patient strong antibiotics and burst open millions of Gram-negative bacteria (like E. coli or Salmonella) in the blood, massive amounts of Lipid A are released. Human macrophages detect Lipid A via Toll-like receptor 4 (TLR4), causing a massive immune overreaction (cytokine storm of TNF-alpha and IL-1) leading to severe fever, a deadly drop in blood pressure, and catastrophic Septic Shock. - Core Polysaccharide: Similar across all Gram-negative bacteria of the same genus. Connects Lipid A to the outer chain.
- Terminal O-Polysaccharides (O-Antigen): A repeating series of sugar units sticking out into the environment. This is the major surface antigen recognized by host antibodies. Because it is highly variable, bacteria use it to evade the immune system.
Fact: There are >2500 different antigenic types in Salmonella alone! Public health scientists use this to track outbreaks (e.g., the deadly strain of E. coli known as O157:H7 is named entirely after its specific O-Antigen and Flagellar H-antigen!).
12. Capsules, Slime Layers, and Appendages
A. Capsules and Slime Layers (Glycocalyx)
A slimy/gummy extracellular material secreted by prokaryotes. It is almost always an extracellular polymer of highly hydrated polysaccharide.
The ONE Exam Exception: The capsule of Bacillus licheniformis (and the deadly Bacillus anthracis) is uniquely made of protein (poly-D-glutamic acid), not polysaccharide!
- Attachment: E.g., Streptococcus mutans uses its heavy slime layer to firmly stick to the smooth enamel of teeth, initiating plaque and causing dental caries (cavities). Furthermore, slime layers allow bacteria (like Staph epidermidis) to form impenetrable biofilms on hospital catheters and IV lines.
- Anti-phagocytic: The capsule acts like a "greased pig." Immune cells (macrophages and neutrophils) try to grab and eat the bacteria, but they slip right out of the immune cell's grip. This makes the bacteria highly pathogenic. (Patients without a functioning spleen, like Sickle Cell patients, are highly susceptible to encapsulated bacteria like Streptococcus pneumoniae).
- Antigenic structure: Doctors use the specific sugars of the capsule to identify (type) the bacteria and to create life-saving vaccines (like the Pneumococcal polysaccharide vaccine).
B. Bacterial Appendages
- Fimbriae: Short, fine, rigid surface structures. Enable bacteria to stick to inert surfaces or form pellicles/scums on surface liquids. Neisseria gonorrhoeae uses fimbriae to tightly anchor itself to the mucosal lining of the urethra so it doesn't get washed away by urine.
- Pili: Longer than fimbriae, usually only 1 or a few present per cell. Made of protein subunits called pillins.
- Adherence: Grabbing onto host tissues (e.g., Uropathogenic E. coli uses special P-pili to climb up the urinary tract and cause severe kidney infections).
- Sex Pili (F-pili): Used like a hollow grappling hook to attach a donor cell to a recipient cell during bacterial conjugation (sharing DNA/plasmids).
- Antigenic Variation: Neisseria gonorrhoeae constantly alters the genetics of its pili proteins. By the time the host immune system creates an antibody to destroy the pili, the bacteria has already swapped out its pili for a new version, meaning the immune system can never create a lasting antibody against it!
- Flagella: Thread-like appendages composed entirely of flagellin protein arranged in a helical structure (12-13nm diameter).
- Function: The primary organ of locomotion (swimming). Bacteria spin these like microscopic boat propellers to move toward food. Note: some bacteria lack flagella and instead glide or use internal gas vesicles to move.
- Antigenic: Flagella are highly antigenic. In Salmonella and E. coli, this is known specifically as the H-antigen.
13. Bacteria Endospores: The Ultimate Survival Mechanism
When environmental conditions become harsh (severe nutritional depletion, high heat, dangerous radiation), certain bacteria (mainly the Gram-positive rods like Bacillus and Clostridium) form a dormant, virtually indestructible internal "escape pod" called an endospore. The vegetative (living, eating) cell undergoes autolysis (bursts open and dies) to release the durable spore into the environment.
- Properties: They are incredibly resistant to heat, drying, radiation, acids, and chemical disinfectants. Standard boiling water will NOT kill them. (Clinical note: Standard alcohol-based hand sanitizers in hospitals DO NOT kill Clostridium difficile spores. Doctors must physically wash their hands with soap and water to wash the spores down the drain!)
- Structure: Composed of a highly dehydrated Core (containing dipicolinic acid and calcium), Cortex, tough protein Spore coat, and Exosporium.
- Classification: Pathologists look at exactly where the spore forms inside the mother cell to identify the bacteria species under a microscope (Central, Subterminal, or Terminal). (For example, C. tetani has a classic terminal spore that looks like a tennis racquet).
Clinical & Microbiological Uses of Spores
- Geobacillus stearothermophilus (Formerly Bacillus stearothermophilus) spores: Because they are so incredibly, famously heat resistant, hospitals put vials of these living spores directly into their autoclaves. If the autoclave successfully kills these spores, the hospital knows the machine is working perfectly to sterilize surgical equipment!
- Bacillus anthracis spores: Extremely deadly if inhaled (causing pulmonary anthrax). Because they can survive for decades in the dirt and be easily processed into a fine powder dispersed in the air, they are unfortunately a top-tier weapon used in biological warfare and bioterrorism.
Quick Quiz
Bacterial Growth, Genetics, and Structure Quiz
Microbiology - mobile-friendly and focused practice.
Privacy: Your details are used only for quiz tracking and certificates.
Bacterial Growth, Genetics, and Structure Quiz
Microbiology
Preparing questions...
Choose your answer and keep your streak alive.
Great effort.
Here is your quick performance summary.
Bacterial Growth, Genetics, and Structure Read More »