HPV and Cervical Cancer
Microbiology: Oncogenic Viruses and Human Papillomavirus (HPV)
This comprehensive master guide details the clinically vital study of tumor-causing viruses. We will thoroughly explore the molecular virology, pathophysiology, clinical manifestations, diagnosis, and prevention of Human Papillomavirus (HPV). You will understand exactly how a microscopic pathogen hijacks cellular machinery to transform healthy tissue into fatal carcinomas.
1. An Overview of Oncogenic Viruses
Oncogenic viruses (tumor viruses) are specialized viruses that produce tumors in their natural hosts, in experimental animals, or induce malignant transformation of cells when placed in a laboratory culture.
The Concept of Transformation
Transformation represents the various molecular, genetic, and phenotypic changes that accompany the conversion of a normal, healthy cell into a malignant (cancerous) cell. It is important to note that a virus does not "want" to cause cancer; cancer is actually a biological accident. The virus simply wants to force the host cell to replicate its DNA, but in doing so, it permanently breaks the cell's normal growth controls.
Tumor viruses are broadly categorized into two distinct classes: DNA tumor viruses and RNA tumor viruses. Both classes possess immense oncogenic potential.
All known DNA tumor viruses either possess a DNA genome or generate a DNA provirus after the infection of host cells. In many cases, their DNA integrates directly into the host's chromosomes. These viruses are classified among the following families:
- Papilloma: e.g., Human Papillomavirus (HPV).
- Polyoma: e.g., Merkel cell polyomavirus, SV40.
- Adeno: Certain Adenoviruses.
- Herpes: e.g., Epstein-Barr Virus (causes Burkitt's Lymphoma) and Human Herpesvirus 8 (causes Kaposi's Sarcoma).
- Hepadna: e.g., Hepatitis B Virus (causes Hepatocellular carcinoma).
- Pox-virus group.
Most RNA tumor viruses belong to the Retrovirus family. Because human cells cannot read RNA to make DNA, these viruses bring their own special machinery.
- Mechanism: Retroviruses carry an RNA-directed DNA polymerase known as Reverse Transcriptase. This enzyme constructs a DNA copy of the viral RNA genome.
- Integration: The newly synthesized DNA copy (the provirus) becomes physically and permanently integrated into the host cell's DNA. From this integrated DNA copy, all viral proteins are translated.
- Examples: Human T-cell Leukemia Viruses (HTLV-1 causes adult T-cell leukemia), Leukosis Sarcoma Viruses.
2. Introduction to Human Papillomavirus (HPV)
Human Papillomavirus (HPV) is the most common sexually transmitted infection globally. Most sexually active men and women will be exposed to the virus at some point during their lifetime.
General Characteristics
- Nomenclature: They got their name because certain types cause benign tumors or warts known medically as papillomas.
- Epitheliotropic Nature: They are widely distributed in nature and are strictly species-specific, host-specific, and tissue-specific. HPV only infects humans, specifically targeting and thriving in squamous epithelia and mucous membranes.
- Diversity: There are more than 100 different kinds (strains/genotypes) of HPV. About 30 to 40 of these specifically cause genital infections.
- Infectivity: The rate of transmission can be exceedingly high—up to 26% after a single unprotected sexual encounter.
- Incubation: Genital warts typically appear 6 weeks to 8 months after contact with an HPV-infected person, though the virus can lie dormant for years.
The Cancer Link & Historical Context
The Danger: HPV is the single most important risk factor for developing cervical cancer, with over 99% of carcinoma of the cervix cases directly linked to HPV infection. Current epidemiological estimates state that up to 80% of sexually active women will be infected by age 50.
Historical Milestones:
- Papillomaviruses were first identified in the early 20th century when it was shown that skin warts could be transmitted between individuals by a filterable infectious agent.
- In 1935, Francis Peyton Rous demonstrated a cancer-causing sarcoma virus in chickens, and concurrent research showed that a papilloma virus could cause skin cancer in infected rabbits (the Shope papillomavirus). This was the first demonstration that a virus could cause cancer in mammals.
- Professor Harald Zur Hausen: Received the Nobel Prize in Medicine in 2008 for being the first to conclusively demonstrate HPV-DNA sequences in human cervical cancer biopsies. He fought against the prevailing medical dogma of the time (which incorrectly blamed Herpes Simplex Virus for cervical cancer).
3. Morphology and Structure of HPV
Understanding the microscopic physical structure of HPV is critical because it directly explains its resilience in the outside environment and its pathogenesis inside the body.
- Viral Architecture (Shape & Size): It is a small, spherical, icosahedral virus with a diameter of 52–55 nm.
- The Envelope (Naked Virus): HPV is a non-enveloped (naked) virus. It completely lacks a lipid envelope.
- Clinical Significance: Because it lacks a fragile lipid envelope (which is easily destroyed by alcohol or soap), HPV is incredibly hardy. It is highly resistant to drying, heat, and standard alcohol-based sanitizers. This allows it to survive on fomites (inanimate objects) for extended periods.
- The Capsid: The viral shell is made of a rigid protein capsid composed of 72 pentameric capsomers. The capsid contains two virally encoded structural proteins:
- L1 (Late 1) protein: The major capsid protein. It facilitates the primary attachment to host epithelial cells. (Note: This is the exact protein used to make the HPV vaccine!)
- L2 (Late 2) protein: The minor capsid protein. It assists in viral entry and transport of the viral genome into the cell nucleus.
- Genetic Material: Contains a single, double-stranded circular DNA molecule of about 7900 to 8000 base-pairs (bp). Interestingly, this viral DNA wraps itself around cellular histones stolen directly from the host cell to pack itself tightly!
4. Classification of HPV
The International Committee on the Taxonomy of Viruses (ICTV) classifies these viruses into two separate families: Papillomaviridae (which includes HPV) and Polyomaviridae. Clinically, HPV strains are rigidly divided based on their oncogenic (cancer-causing) potential.
- Examples: HPV 6, 11, 42, 43, 44.
- Clinical Presentation: These types tend to infect the skin and mucous membranes to cause benign genital lesions and warts (Condylomata acuminatum).
- Appearance: Warts can appear as small, cauliflower-like growths on or around the genitals, hands, feet, and anus. They rarely, if ever, progress to cancer.
- Examples: HPV 16, 18, 31, 33, 34, 35, 39, 45, 51, 52, 56, 58, 59, 66, 68, 70.
- Clinical Presentation: Found preferentially in precancerous and cancerous specimens. They cause persistent, silent infections leading to precancerous cellular changes (dysplasia) and full-blown invasive carcinoma.
- The Deadliest: HPV 16 and 18 alone are the most dangerous, causing the vast majority (~70%) of all HPV-related cancers worldwide.
Low Risk: Think of a V6 engine or an 11-year-old child (HPV 6 & 11) — annoying (warts) but generally safe and not lethal.
High Risk: Think of the legal ages of adulthood and consent (HPV 16 & 18) — serious, grown-up consequences (Cancer).
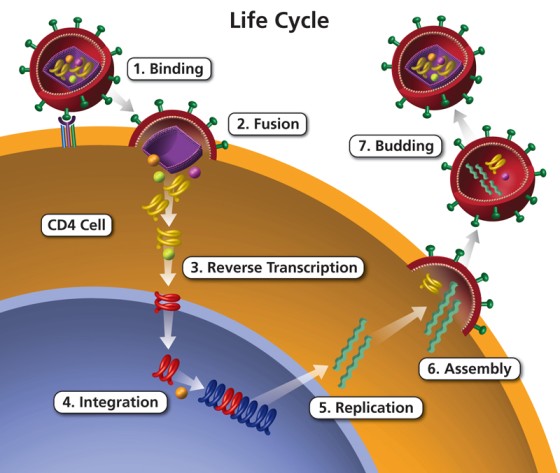
5. Pathogenesis & Viral Life Cycle
Entry and Initial Infection
- The Breach: The virus cannot bind to dead, intact surface tissue. HPV strictly enters the body through micro-abrasions or micro-trauma in the skin or mucosa that exposes the deep basement membrane.
- The Target: It specifically infects the deep, dividing basal cells of stratified squamous epithelium (e.g., in the cervix, skin, and oropharynx).
- Receptor Binding: The HPV virion (via the L1 protein) associates with specific cellular receptors such as alpha-integrins, laminins, and annexin A2. It then enters the cell through endocytosis.
Viral Replication & Evasion
HPV replication is tightly, brilliantly linked to the natural differentiation (aging) of host epithelial cells. It uses the cell's natural life cycle to its advantage.
- Latent Phase: In the deep basal cells, the viral DNA exists as an episome (a free-floating circle of DNA). It is maintained in very low copy numbers. The virus is quiet here.
- Amplification Phase: As the infected host cells naturally mature, move upward, and differentiate into superficial skin cells, viral DNA replication rapidly increases.
- Assembly & Release: The "Late" structural genes (L1, L2) are finally expressed in the uppermost layers. Massive numbers of new virions are assembled. The virus is naturally released when the dead, superficial squamous cells are naturally sloughed/shed off during daily friction.
Immune Evasion Mechanisms ("The Stealth Virus")
HPV avoids immune detection for months or years by employing several stealth tactics:
- No Viremia: It never enters the bloodstream; it stays completely localized in the avascular surface epithelium. Therefore, circulating white blood cells rarely encounter it.
- Low Protein Expression: It hides in the basal cells by producing almost no viral proteins that the immune system could detect.
- Non-lytic release: It doesn't burst live cells (which would cause inflammation). It simply rides dead cells off the surface, triggering absolutely no inflammatory immune response! Without inflammation, dendritic cells are not alerted.
6. Molecular Virology: How HPV Causes Cancer
The outcomes of HPV infection are usually either a transient infection (cleared by the immune system, mostly low-risk types) or a persistent infection (high-risk types). Persistent infection with high-risk HPV is what leads to malignant transformation via its major virulence factors: the Viral Oncoproteins.
The Engine of Cancer: E6 and E7 Proteins
High-risk HPV types (like 16 and 18) produce early oncoproteins, E6 and E7, which completely disrupt normal host cell cycle control by attacking the body's natural tumor suppressor proteins. (You MUST memorize these exact mechanisms for your exams).
1. The E6 Protein destroys p53
- Normal Function: Normal p53 is the "Guardian of the Genome." It acts as a strict quality control inspector. If a cell's DNA is mutated, p53 halts the cell cycle to fix it, or induces apoptosis (programmed cell suicide) to prevent the mutation from spreading.
- HPV Intervention: The HPV E6 protein binds to a host protein called E6-associated protein (E6AP), which acts as a ubiquitin ligase. Together, they attach ubiquitin tags to the normal p53. Ubiquitin is a "kiss of death" tag that marks p53 for complete destruction in the cell's proteasome (the cellular garbage disposal).
- Result: The cell loses its p53 inspector. It completely loses its ability to undergo apoptosis, becoming essentially immortal and allowing massive DNA mutations to accumulate.
2. The E7 Protein destroys pRb
- Normal Function: Normal Retinoblastoma protein (pRb) is the "Brakes" of the cell cycle. It physically hugs and traps a transcription factor called E2F, preventing the cell from transitioning from the G1 phase to the S phase (DNA synthesis).
- HPV Intervention: The HPV E7 protein acts as the primary transforming protein. It aggressively binds to pRb with extremely high affinity, competing with and displacing the trapped E2F.
- Result: The "brakes" are cut. E2F is set free to travel to the nucleus and transactivate its targets, pushing the cell cycle forward into unrestrained, uncontrollable cellular proliferation.
Other Important Viral Proteins
- E4 Protein: Plays a crucial role in causing G2 arrest in HPV-infected cells (pausing the cell cycle at a specific point highly favorable for viral genome amplification) and helps break down the cellular cytoskeleton to aid virus release.
- E5 Protein: Enhances growth factor signaling in the host cell (specifically the Epidermal Growth Factor Receptor), supporting the early stages of cellular transformation.
Conclusion: The combined, devastating actions of E5, E6, and E7 lead to massive cellular proliferation, allowing for the accumulation of host genetic mutations, and ultimately driving the progression from benign dysplasia to invasive carcinoma.
7. Transmission & Consequences of HPV Infection
Transmission Routes
- Direct Skin-to-Skin Contact: The primary mode of transmission. In children, this easily transmits skin warts from hands or feet.
- Sexual Transmission: Having vaginal, anal, or oral sex with someone who has the virus. It is most commonly spread during vaginal or anal sex. The virus can be passed even when an infected person has absolutely no visible signs or symptoms (asymptomatic shedding).
- Fomites (Very Rare): Sharing of contaminated objects (e.g., gymnasium apparatus, damp towels, or swimming pool decks). Though the virus is hardy, fomite transmission for genital types is exceedingly rare compared to cutaneous types.
- Vertical Transmission: From an infected mother to child during passage through an infected birth canal. This can lead to a dangerous condition in the infant called laryngeal papillomatosis.
The "Silent" Infection
A person can develop symptoms years or even decades after being infected. Because the immune system usually clears the virus (or forces it into a dormant latency) in most people, it is notoriously hard to know exactly when or from whom you first became infected.
Clinical Consequences of Infection
While most HPV goes away on its own within 1-2 years via cellular immunity, persistent infection causes:
- Cutaneous warts (skin).
- Genital warts.
- Oral/laryngeal warts.
- Precancerous changes of the cervix, vagina, rectum, or anus.
- Frank malignancy (Cervical, penile, anal, or oropharyngeal cancer).
8. Cutaneous and Anogenital Warts
A. Cutaneous Warts (Skin)
These lesions are strictly benign. They involve hypertrophy (thickening) of all layers of the skin. They frequently affect the hands and feet, especially in children and adolescents.
- Plantar warts: Found on the soles of feet and palms (HPV 1, 4). Described as hard, grainy, painful growths on the heels or balls of the feet.
- Flat warts: Found on the face, neck, and hands (HPV 3, 10). Generally affect children/young adults; appear as flat-topped, slightly raised lesions darker than normal skin color. Often spread in a line by scratching (autoinoculation).
- Common warts: Can occur anywhere on the skin, oral mucosa, hands, and around the mouth (HPV 2, 4, 7). Rough, raised bumps mostly on fingers and elbows.
- Immunosuppression: There is an exponentially increased risk of severe, widespread cutaneous warts in persons with depressed cell-mediated immunity (e.g., patients with renal transplants receiving chronic steroid therapy, or HIV patients).
- Epidermodysplasia verruciformis: Also known as "Treeman syndrome." This is a rare genetic immunodeficiency leading to massive susceptibility to normal cutaneous HPV types, causing tree-bark-like warts that carry a high risk of transforming into squamous cell skin cancers.
B. Anogenital Warts (Condylomata Acuminata)
- Presentation: May appear as a small bump, a cluster of bumps, or stem-like protrusions. They can range in size and appearance (large, small, flat, or cauliflower-shaped) and may be white or flesh-toned.
- Location: In women (vulva, cervix, vagina, anal, and perianal region). In men (shaft of the penis, scrotum, anal, and perianal region).
- Causative Agents: HPV types 6 and 11 are the most common causes of these benign genital warts in both sexes.
Do not confuse Condylomata Acuminata (HPV Genital Warts) with Condylomata Lata!
Acuminata = "Accumulating" cauliflower-like warts caused by HPV.
Lata = "Flat", velvety, moist lesions caused by Secondary Syphilis (Treponema pallidum).
C. Orolaryngeal Lesions
- Recurrent Respiratory Papillomatosis (RRP): Aerodigestive benign tumors, most commonly laryngeal papillomas (caused by HPV 6 and 11). These can grow rapidly on the vocal cords and obstruct the airway of children or adults, requiring repeated laser surgical removal.
- Focal epithelial hyperplasia: Benign neoplastic condition in the mouth.
- Oral papillomas: Benign epithelial tumors around the mouth and tongue.
9. HPV, Cervical Cancer, and HIV Co-infection
The Progression to Cervical Cancer
Malignant disease of the cervix is always preceded by a neoplastic change in the surface epithelium, a pre-cancerous warning condition known as Cervical Intraepithelial Neoplasia (CIN). This involves new, abnormal, disorganized growth of the cervix epithelium, marked by cells with large, dark nuclei (high nuclear-to-cytoplasmic ratio).
- Natural History: Normal Cervix → HPV Infection → Viral Persistence → Progression to Pre-cancer (CIN) → Invasion (Cancer).
- Causative Agents: Nearly all cervical cancer is due to HPV. HPV 16 and HPV 18 account for 70% of cases. (Other high-risk types include 45, 31, 33, 52, and 58).
- Cofactors for Progression: HPV alone is absolutely necessary, but NOT sufficient to cause cervical cancer. A combination of HPV and one or more cofactors dramatically increases the risk of progression. These include:
- Poor hygiene.
- High parity (multiple full-term pregnancies).
- Hormonal contraceptives (prolonged use of oral contraceptive pills).
- Smoking (increases risk 4 times!).
- Immunosuppression (HIV, Rheumatoid Arthritis, Cancer).
- Multiple sexual partners & early age at first intercourse (< 16 years).
- Other STIs (like Chlamydia).
- Familial/genetic predisposition.
Staging Pre-Cancer
Pathologists stage the progression of this disease on a biopsy based on how deep the abnormal cells have penetrated:
- CIN I (Mild): The number and depth of abnormal cells is low. Dysplasia is confined strictly to the bottom 1/3 of the epithelial thickness.
- CIN II (Moderate): Abnormal cell growth penetrates about 1/2 to 2/3 the thickness of the cervical epithelium.
- CIN III (Severe / Carcinoma In-Situ): Abnormal cell growth penetrates the entire thickness of the cervical epithelium. However, the critical basement membrane is completely intact.
- Invasive Cervical Cancer: The malignant cells break through and penetrate beyond the basement membrane into the underlying connective tissue, accessing blood vessels to metastasize.
HPV and HIV Co-Infection
Individuals infected with both HPV and HIV have an exponentially increased risk of developing cervical or anal cancer.
- Why? Immunosuppression (massive loss of CD4 T-cells) inhibits the body's ability to clear papillomaviruses and actively promotes HPV reactivation.
- The prevalence of HPV is 5 times higher in HIV-infected individuals than in the general population.
- These patients rapidly develop a greater number of precancerous lesions. In HIV-infected women undergoing colposcopy after an abnormal Pap smear, the detection of HPV is greater than 90%.
Epidemiology of Cervical Cancer
- It is the 2nd most common cancer in women worldwide.
- It is the most common cause of cancer death in females in developing countries.
- Stats Highlight: In India, over 200 women die every day from cervical cancer (roughly 72,000 females per year; 1 woman dies every 7 minutes).
- Symptoms: There may be absolutely no signs or symptoms until the cancer has progressed to a dangerous, invasive stage. When symptoms do occur, they include: abnormal vaginal bleeding (especially post-coital bleeding after intercourse, or bleeding other than during menstruation), abnormal foul-smelling vaginal discharge, and severe pelvic/back pain in advanced stages.
10. HPV and Head & Neck Cancer (OPSCC)
While HPV is famous for cervical cancer, the epidemiological landscape is changing. It is rapidly becoming the leading cause of Oropharyngeal Squamous Cell Carcinoma (OPSCC).
- Epidemiology: 6th most common cancer worldwide (>600,000 new diagnoses annually). >95% are Squamous Cell Carcinomas.
- In recent years, studies show that ~25% of all oropharyngeal carcinomas are associated with oncogenic high-risk HPV (specifically HPV-16, which is present in 94% of these specific cancers).
- Commonest Sites: The virus prefers the deep, crypt-like lymphoid tissues of Waldeyer's ring. The most common sites are the Tonsils, Base of the tongue, Lingual tonsil, and the Lateral wall of the oropharynx.
- The Shift: Patients with HPV-related SCCs often do NOT have the traditional risk factors (smoking, heavy alcohol consumption, or tobacco chewing). Instead, research links it to a higher number of sexual partners and a massive increase in oral sexual behavior.
- Future Outlook: By 2030, OPSCC will likely constitute the absolute majority (47%) of all Head and Neck cancers, surpassing the annual number of cervical cancers, with the vast majority occurring among men.
❓ Advanced Molecular Pathology: Why do HPV+ throat cancers survive better?
Clinical Paradox: Clinical studies overwhelmingly show that patients with HPV-positive Head and Neck cancers have a much better prognosis and better survival rates than patients with traditional (smoking/alcohol-induced) HPV-negative cancers. Why?
Answer: In smoking-induced cancers, the chemical carcinogens physically mutate and destroy the critical tumor suppressor gene p53 genetically. The gene is gone forever. In HPV-induced cancer, the wildtype (normal, healthy) p53 gene is perfectly intact! The HPV E6 protein is simply holding it hostage via degradation. When radiation or chemotherapy damages the cancer cells, it disrupts the virus, releasing the normal p53 to flood the cell and trigger massive, highly successful apoptosis (cancer cell suicide). Furthermore, the expression of foreign viral proteins triggers a strong, targeted anti-tumor immune response from the patient's own T-cells!
Because of this high cure rate, clinical trials are actively testing "Deintensification" (reducing the dose of radiation/chemo) for HPV+ patients to save them from the long-term severe morbidities of heavy radiation (like permanent dry mouth and swallowing difficulties).
11. Diagnosis & Detection of HPV
Traditional viral diagnostic methods like electron microscopy and cell culture are useless for HPV detection because HPV cannot be cultured in standard laboratory cell cultures (it requires fully differentiating, intact, 3-dimensional human skin to complete its complex life cycle).
Important modern diagnostic methods include:
- The PAP Smear (Papanicolaou Test): Developed by Dr. George N. Papanicolaou in the 1940s. It is the most common and successful cancer screening test in history. A sample of mucus and cells is scraped from the cervix/endocervix using a wooden scraper or cervical brush, rinsed into a vial, and examined by a cytologist on a slide. It detects early precancerous changes (dysplasia) long before symptoms arise. It has vastly reduced cervical cancer mortality.
- Acetic Acid Test: A vinegar (3-5% acetic acid) solution is applied to the genital areas. HPV-infected, dysplastic tissue has highly dense nuclei and will rapidly turn white (acetowhite) because the acid dehydrates the cells. This helps clinicians identify difficult-to-see flat lesions.
- Colposcopy: A procedure that allows illuminated, stereoscopic, magnified viewing of the cervix to pinpoint exactly where to biopsy after an abnormal Pap smear or acetowhite test.
- Biopsy: Taking a physical tissue sample for histological grading by a pathologist (staging it as CIN I, II, or III). If pre-cancer or cancer is found, definitive treatment is initiated.
- Molecular DNA Testing (The Gold Standard for Virology):
- PCR-based methods: HPV DNA is exponentially amplified selectively by a series of reactions. This detects the exact viral strain (e.g., typing for HPV 16 vs 11).
- DNA In-Situ Hybridization / Southern Blot.
- p16 Immunohistochemistry (IHC): A highly advanced stain. It detects the evidence of functioning Oncoprotein E7. (Because E7 destroys pRb, the cell massively overproduces a backup brake protein called p16 in a panic. Staining a slide brown for p16 essentially proves the virus is actively driving the cancer pathway).
- mRNA of E6/E7 testing.
Note on Serology: Serological assays (ELISA/Western blot) detect antibodies in the blood. They are not useful for diagnosing current/active HPV infection because the virus evades the blood, but they are useful for epidemiological studies showing evidence of past exposure in populations.
12. Treatment and Management
There is currently NO specific antiviral treatment to cure an HPV infection. Management relies entirely on removing the problematic warts or precancerous/cancerous cells and letting the patient's own immune system handle the rest.
Medical/Topical Treatment of Warts:
- Salicylic acid: Over-the-counter treatment that works by chemically peeling away layers of the wart a little at a time.
- Imiquimod (Aldara, Zyclara): A prescription cream that acts as an immune response modifier, heavily enhancing the body's local immune system (interferon production) to attack the HPV. (Side effects: severe local redness/swelling).
- Podofilox (Condylox): A topical cytotoxic prescription derived from plant extracts that directly arrests cell division and destroys genital wart tissue. (Side effects: pain/itching).
- Trichloroacetic acid (TCA): A harsh chemical treatment applied by a clinician that literally burns off warts on palms, soles, and genitals through protein coagulation.
- Interferons: Have been tried via injection, especially for stubborn CIN II and III lesions, though less common due to systemic side effects.
Surgical & Procedural Removal:
- Cryotherapy: Freezing the wart/dysplasia with liquid nitrogen or dry ice, causing it to blister and fall off.
- Electrocautery: Burning the tissue with a high-frequency electrical current.
- Laser surgery & Surgical excision (scalpel): Used for large warts or high-grade CIN (e.g., LEEP procedure - Loop Electrosurgical Excision Procedure).
- Management of Oropharyngeal Cancer: Standard single-modality treatment (surgery OR radiotherapy) is recommended for early (T1-T2, N0) disease.
13. Prevention & Vaccination
A. General Prevention
- Abstinence / Mutual Monogamy: Avoid skin-to-skin contact by abstaining from sex or maintaining a mutually monogamous relationship with an uninfected partner.
- Condoms and Dental Dams: Should be used every time. Crucial Note: Because HPV is transmitted via direct skin-to-skin contact (like rubbing against the base of the penis, scrotum, or vulva), condoms are not very good at completely preventing HPV (unlike HIV or Chlamydia, which require fluid exchange). Spermicide (nonoxynol-9) is also not protective against HPV. However, safer sex practices still significantly lower the chances of transmission and reduce viral load.
- Good personal hygiene: Particularly for preventing cutaneous warts.
- Secondary Prevention: Regular gynecologic exams and Pap smear screening to catch dysplasia before it turns into cancer.
B. Cervical Cancer Screening Guidelines
- First screen 3 years after first sexual intercourse or by age 21.
- Screen annually with regular Paps, or every 3 years with liquid-based tests.
- After three completely normal tests in a row, women can transition to screening every 5 years (often co-tested with HPV DNA testing).
- Stop screening at age 65-70 years if there is a consistent, documented history of negative tests.
C. The HPV Vaccines (A Major Public Health Triumph)
Three FDA-approved vaccines are available. They strictly prevent initial infection; they cannot be used to treat an infection, clear warts, or cure cancer after it has already developed.
Vaccine Mechanism of Action (MOA): They are non-infectious. They utilize Virus-Like Particles (VLPs) composed exclusively of the major L1 capsid protein. Because they contain absolutely no viral DNA, they cannot cause infection or replicate, but their perfectly icosahedral shape tricks the immune system into triggering a massive, highly protective neutralizing antibody response.
A Bivalent vaccine. Protects against HPV 16 and 18 (the two deadliest cancer-causing strains). Uses a proprietary ASO4 adjuvant to boost immune response.
A Quadrivalent vaccine. Protects against HPV 6, 11, 16, and 18. Prevents 90% of genital warts AND the major cancers. Uses an AAHS adjuvant.
A Nonavalent vaccine. Protects against 9 strains (6, 11, 16, 18, 31, 33, 45, 52, 58). Has the potential to prevent about 90% of all cervical, vulvar, vaginal, and anal cancers globally.
Vaccine Dosage & Administration
- Should be administered intramuscularly (IM) as 3 separate 0.5-mL doses according to a schedule of 0, 2, and 6 months (though newer guidelines allow for a 2-dose schedule if started early enough).
- Target age group: Routine vaccination in females and males starting at 11-12 years old (can start as early as 9 to ensure protection before sexual debut). Catch-up vaccination extends to age 26 (and in some guidelines, up to age 45 with clinical discretion).
- Duration: Efficacy lasts 5 to 10 years at minimum, and currently, no booster dose has been formally recommended.
- Clinical Warning: Care must be taken not to inject intravenously. The injection can lead to a syncopal (fainting) attack, so adolescents should be seated or lying down and observed for 15 minutes post-vaccination.
Special Populations
- Should boys be vaccinated? Yes, absolutely. It prevents penile, anal, and massive numbers of rising oropharyngeal (throat) cancers, and generates herd immunity to stop transmission to females.
- Can it be given to pregnant women? Generally avoided during pregnancy strictly due to a lack of robust safety data, but it is completely safe for lactating/breastfeeding mothers.
14. References
- Brooks, G. F., Carroll, K. C., Butel, J. S., Morse, S. A., & Mietzner, T. A. (2013). Jawetz, Melnick & Adelberg's Medical Microbiology (26th ed.). McGraw-Hill Education.
- Kumar, V., Abbas, A. K., & Aster, J. C. (2014). Robbins and Cotran Pathologic Basis of Disease (9th ed.). Elsevier Saunders.
- Centers for Disease Control and Prevention (CDC). (2021). Human Papillomavirus (HPV) Infection. Retrieved from CDC Guidelines.
- zur Hausen, H. (2002). Papillomaviruses and cancer: from basic studies to clinical application. Nature Reviews Cancer, 2(5), 342-350.
- World Health Organization (WHO). (2020). Cervical Cancer. WHO Fact Sheets.
Quick Quiz
HPV Quiz
Microbiology - mobile-friendly and focused practice.
Privacy: Your details are used only for quiz tracking and certificates.
HPV Quiz
Microbiology
Preparing questions...
Choose your answer and keep your streak alive.
Great effort.
Here is your quick performance summary.
HPV and Cervical Cancer Read More »