NSAIDS & Prostanoids
NSAIDs & Prostanoids Pharmacology
This master guide covers the pharmacology of Non-Steroidal Anti-Inflammatory Drugs (NSAIDs) and Prostanoids. We will explore the Arachidonic Acid pathway, the profound differences between COX-1 and COX-2, specific drug classifications, and the synthetic prostanoids used to manipulate everything from childbirth to glaucoma. Enhanced with clinical scenarios and deep-dive explanations to guarantee exam success.
1. The Foundation: Prostanoids and the MOA of NSAIDs
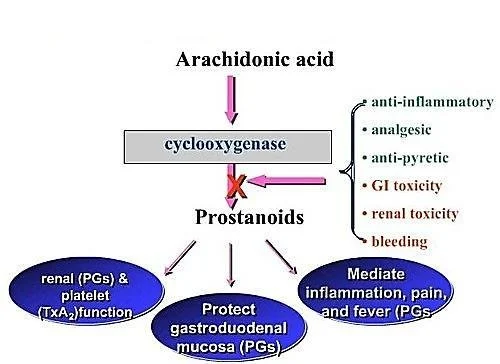
Before understanding the drugs, you must understand the assembly line that makes the molecules these drugs block. This is the Arachidonic Acid Pathway. Think of this pathway as a factory that takes raw materials from the cell wall and turns them into highly active chemical messengers.
MEMBRANE PHOSPHOLIPIDS
↓
Enzyme: Phospholipase A2 (BLOCKED by Corticosteroids)
↓
ARACHIDONIC ACID
↙
Lipoxygenase
↓
Leukotrienes
(Cause Bronchospasm/Asthma)
↘
Cyclooxygenase (COX)
(BLOCKED by NSAIDs)
↓
PGG2 → PGH2
(Endoperoxides)
↓
PROSTANOIDS:
Prostaglandins (PGE2, PGF2α, PGD2)
Thromboxane (TXA2)
Prostacyclin (PGI2)
Steroids vs. NSAIDs & The "Shunt" Phenomenon
Notice that Corticosteroids block the pathway at the very top (Phospholipase). Therefore, steroids stop BOTH Leukotrienes (which cause asthma) and Prostanoids. NSAIDs only block the COX enzyme lower down.
This means NSAIDs stop pain and fever (Prostanoids) but do nothing to stop Leukotrienes. In fact, in some asthma patients, giving an NSAID creates a "Leukotriene Shunt". Because the COX pathway is blocked, all the built-up Arachidonic acid is violently pushed down the Lipoxygenase pathway, causing a massive overproduction of Leukotrienes. This triggers a severe, life-threatening asthma attack (a condition clinically known as Aspirin-Exacerbated Respiratory Disease or AERD).
The Cyclooxygenase (COX) Isozymes: The "Housekeeper" vs. The "Fire Alarm"
The COX enzyme comes in different versions (isoforms). Knowing the difference is the absolute key to understanding NSAID side effects and why pharmaceutical companies spent billions inventing specific COX-2 inhibitors.
| COX-1 (The Housekeeper) | COX-2 (The Fire Alarm) | COX-3 (The Mystery) |
|---|---|---|
|
|
|
2. Classification of NSAIDs
NSAIDs are classified either by their chemical structure/efficacy or by how selectively they block the COX enzymes.
Classification by COX Selectivity (The Slide 4 Breakdown)
Note on exam preparation: Some drugs straddle the line of selectivity based on dose. For example, Aspirin is selective for COX-1 at low doses, but non-selective at high doses.
- Selective COX-1 Inhibitors (Usually low doses): Low dose Aspirin, Ketoprofen, Flurbiprofen, Indomethacin, and Ketorolac (sometimes spelled 'Ketoloid' on older slides).
- Non-Selective COX Inhibitors (Traditional NSAIDs): Piroxicam, Tenoxicam, Ibuprofen, Naproxen, Diclofenac. These hit both COX-1 and COX-2 equally, killing pain but ruining the stomach.
- Selective COX-2 Inhibitors (The "-coxibs" & friends): Celecoxib, Etoricoxib, Meloxicam (preferential), Nimesulide. Designed to kill pain without giving you a stomach ulcer.
Classification by Efficacy and Chemical Class (The Slide 5 Breakdown)
Why do we care about chemical classes? Because if a patient is highly allergic or fails to respond to an NSAID from the "Propionic Acid" class, a wise doctor will switch them to a completely different chemical class, like an "Oxicam".
Non-Selective COX Inhibitors (Traditional)
- Salicylic Acid Derivatives: Aspirin
- Propionic Acid Derivatives: Naproxen, Ibuprofen, Ketoprofen
- Pyrazolon Derivatives: Phenylbutazone
- Acetic Acid Derivatives: Diclofenac, Aceclofenac, Nebumetone, Sulindac
- Pyrrolo-pyrrole Derivatives: Ketorolac
- Indole Derivatives: Indomethacin
- Oxicams: Piroxicam, Tenoxicam
- Fenamates: Meclofenamic acid, Tolfenamic acid, Flufenamic acid
- Anthranilic acid: Mefenamic acid
3. Preferential & Selective COX-2
- Preferential COX-2 Inhibitors: Meloxicam, Nimesulide.
- Selective COX-2 Inhibitors: Celecoxib, Etoricoxib.
4. Analgesics with POOR/NO Anti-inflammatory
- Para-aminophenol Derivatives: Acetaminophen/Paracetamol.
3. Mechanism of Action (MOA) and General Adverse Effects
Primary MOA: NSAIDs inhibit the cyclooxygenase (COX) enzyme, resulting in the reduced biosynthesis of Prostanoids (Prostaglandins, Prostacyclin, and Thromboxane A2).
Why do Traditional NSAIDs cause side effects?
Aspirin and older, non-selective NSAIDs block BOTH COX-1 and COX-2. By blocking COX-2, they brilliantly stop inflammation, pain, and fever. BUT, by blocking COX-1, the release of PGs required for homeostatic (housekeeping) function is totally disrupted.
The Mechanisms of Toxicity
- The Stomach: PGE2 and PGI2 normally stimulate the production of thick, protective gastric mucus and bicarbonate. They also maintain rich blood flow to the stomach wall. NSAIDs stop this synthesis.
Result: The stomach acid literally burns through the unprotected stomach wall, causing Gastric and Duodenal Ulcers, and severe GI Bleeding. - The Kidneys: PGE2 and PGI2 are responsible for actively dilating the afferent renal arteriole (the blood vessel bringing blood INTO the kidney filter), which maintains the Glomerular Filtration Rate (GFR). If you block this (especially in elderly patients with already impaired kidneys or low blood volume), blood flow to the kidney drops sharply.
Result: Serious kidney damage, acute renal failure, and severe fluid retention.
General Adverse Reactions of NSAIDs (System by System)
- Gastrointestinal Tract (Most Common): Nausea, vomiting, diarrhea, constipation, epigastric pain, indigestion, abdominal distress, intestinal ulceration, stomatitis, jaundice, bloating, anorexia, and dry mouth.
- Central Nervous System (CNS): Dizziness, headache, drowsiness, insomnia.
- Cardiovascular: Decrease or increase in blood pressure (often increasing it due to fluid retention), and cardiac arrhythmias.
- Renal: Hematuria (blood in urine) and acute renal failure (in those with pre-existing impaired function).
- Special Senses: Visual disturbances, blurred or diminished vision.
- Hematologic: Anemia (often secondary to chronic microscopic GI bleeding over months of daily NSAID use).
4. Deep Dive: Aspirin, Acetaminophen, and Selective COX-2s
A. ASPIRIN (Acetylsalicylic Acid)
Aspirin is completely unique among all NSAIDs. It irreversibly acetylates both isoforms of the COX enzyme. This means it covalently binds to the enzyme and kills it permanently. The cell must synthesize brand new enzymes from scratch to recover function. For a normal cell, this takes hours to days. But for platelets (which have no nucleus and cannot make new proteins!), the enzyme is dead for the entire 7-10 day lifespan of the platelet.
- As an Anti-inflammatory: Inhibits PG biosynthesis to modulate inflammation. Used in Rheumatoid Arthritis (RA), but note: it only helps the symptoms, it neither arrests nor cures the progress of the disease.
- As an Analgesic (Painkiller): Reduces production of PGE2. PGE2 normally sensitizes nerve endings to pain. By blocking it, Aspirin represses pain sensation. Used for toothache, dysmenorrhea (menstrual pain), and post-operative pain (often used alongside opioids to reduce the opioid dose). It also inhibits pain stimuli at subcortical sites (Thalamus & Hypothalamus).
- As an Antipyretic (Fever Reducer): Aspirin lowers raised body temperature by acting on the hypothalamus (resetting the brain's thermostat). It has no effect on normal body temperature.
- As an Antiplatelet (Blood Thinner): In low doses (e.g., 75mg - Ecorin-75), it permanently inhibits platelet aggregation because it stops the production of TXA2 (which normally promotes clotting). Used globally to prevent heart attacks and strokes.
Aspirin: Adverse Effects & Contraindications
Adverse Effects:
- GI disturbances (Can be prevented if given with Misoprostol or as enteric-coated tablets).
- Impaired hemostasis (prolonged bleeding—a small cut might bleed for a long time).
- Allergy / Hypersensitivity reactions.
- Hyperuricemia: At low doses, aspirin retains uric acid in the kidneys. (Clinical Trap: Giving low-dose aspirin to a patient with a history of Gout can trigger a massive gout attack!).
- Decreased renal function.
- Salicylism: A specific mild toxicity syndrome characterized by Vomiting, Tinnitus (severe ringing in ears), and Vertigo.
- Respiratory depression in toxic doses (due to CNS effects and acid-base disturbances).
- Reye's Syndrome: A fatal condition causing rapid brain and liver swelling in children recovering from viral illness (like chickenpox or the flu). Clinical Rule: Never give Aspirin to a child with a fever! Use Acetaminophen or Ibuprofen instead.
Contraindications:
- Peptic ulcer disease.
- Hemophilia or bleeding disorders.
- Hypersensitivity.
- Children with a viral illness.
- Chronic liver disease.
- Surgical Note: Aspirin must be stopped one week before elective surgery (because platelets take 7 days to regenerate).
- Avoid high doses in G-6-PD deficient patients.
- Pregnancy & Lactation: Avoid! Can cause rare but serious kidney problems in unborn babies and premature closure of the ductus arteriosus.
Note: There is NO specific chemical antidote for Aspirin overdose till date (treatment is supportive, largely involving alkalinizing the urine with sodium bicarbonate to trap the acid in the urine and force excretion).
B. ACETAMINOPHEN (Paracetamol)
MOA: Rapid absorption from GIT. Significant first-pass metabolism in gut wall and liver. It works mainly centrally (CNS) on COX-3.
Uses: Used for mild to moderate pain and fever.
Exam Trap: Acetaminophen has NO anti-inflammatory activity. It is NOT an NSAID. It will not reduce swelling in a sprained ankle or an arthritic knee.
Acetaminophen Toxicity & Overdose
At therapeutic doses, it is incredibly safe (may cause rare drug fever or mild increase in hepatic enzymes). However, in overdose (above 10-15g), the liver's normal metabolic pathways are totally overwhelmed. A minor pathway takes over, producing a highly toxic, tissue-destroying metabolite called NAPQI.
Overdose Symptoms: Hepatic necrosis (fatal liver failure), Renal tubular necrosis, Hypoglycemic coma.
The Antidote: N-acetyl Cysteine (NAC). Normally, the liver uses a substance called Glutathione to neutralize NAPQI. In overdose, glutathione runs out. NAC works by rapidly replenishing the liver's glutathione stores, neutralizing the toxic metabolite and saving the patient's liver.
C. SELECTIVE COX-2 INHIBITORS (The "Coxibs")
These drugs were engineered to be 10-20 times more selective for COX-2 and bind reversibly. The goal? Kill the pain/inflammation (by blocking COX-2) without hurting the stomach (by leaving COX-1 alone).
- Celecoxib: Chemically a sulphonamide (watch for sulfa allergies!). Half-life of 11 hours.
- Meloxicam: Related to Piroxicam. Preferentially selective.
- Etoricoxib: Long half-life (22 hours). Requires strict monitoring of hepatic functions.
- Nimesulide: Newer compound, less gastric irritation.
The "Coxib" Double-Edged Sword
The Advantages: Excellent analgesic, antipyretic, and anti-inflammatory effects. No inhibition of protective gastric PGs (No gastric irritation/ulcers). No inhibition of platelets (Does not prolong bleeding time).
The Disadvantages (The Fatal Flaw): High COX-2 selectivity ruins the delicate balance in the blood vessels. Normally, there is a "tug-of-war" between COX-2 (makes Prostacyclin, which dilates vessels and stops clots) and COX-1 (makes Thromboxane A2, which constricts vessels and makes platelets stick together).
By wiping out COX-2 completely, you leave COX-1 completely unopposed. The blood vessels clamp down and platelets clump together. Result: High risk of severe Cardiovascular thrombotic events (Myocardial Infarction / Strokes).
Historical Note: Drugs like Valdecoxib and Rofecoxib (Vioxx) were completely withdrawn from the market due to causing deadly heart attacks.
Other Adverse Effects: Renal toxicities (similar to non-selective NSAIDs) and Skin Rashes (specifically with Celecoxib due to its sulfa structure).
5. Master Clinical Uses Table (By Drug)
Memorize these specific associations based on your slides.
| Generic Name | Trade Name | Specific Clinical Uses | Specific Adverse Reactions |
|---|---|---|---|
| Celecoxib | Zycel | Rheumatoid arthritis (RA), Osteoarthritis (OA). | Ophthalmic changes, Skin rashes, CV risk. |
| Diclofenac Sodium | Voltaren, Olfen | RA, OA, Ankylosing spondylitis. | Gastric and duodenal ulcers formation, GI bleeding. |
| Fenoprofen | Nalfon | Long term management for mild to moderate pain. | Visual disturbances, Jaundice, Peptic ulcers. |
| Ibuprofen | Advil, Ibumex | Mild to moderate pain, Painful dysmenorrhea, RA. | GI Disturbances, Nausea, Dizziness, GI Bleeding. |
| Indomethacin | Indocin | RA, Ankylosing spondylitis, Acute gouty arthritis. | Hematologic changes, Nausea, Constipation, Duodenal Ulcers. |
| Meflofenamate | Meftal | Mild to moderate pain, Painful dysmenorrhea. | Rash, Bleeding, Headache, Dizziness, Nausea, Dyspepsia. |
| Naproxen | Aleve, Anaprox | Management of inflammatory disorders, Mild/mod pain, Dysmenorrhea. | Visual changes, Nausea, Vomiting, GI bleeding. |
| Rofecoxib | Vioxx | Signs/symptoms of OA, Acute pain, Primary dysmenorrhea. (Withdrawn) | Visual Disturbances, CV events. |
| Sulindac | Clinoril | Mild to moderate pain, RA, Ankylosing spondylitis, Gouty arthritis. | Nausea, Vomiting, Diarrhea, Constipation, GI bleeding, Ulcers. |
| Valdecoxib | Bextra | OA, RA. (Withdrawn) | Anemia, Headache, Dyspepsia, CV events. |
Choosing an NSAID (Advantages vs Disadvantages)
- Salicylates (Aspirin):
Advantage: Low cost, long history of safety.
Disadvantage: Upper GI disturbances are very common. - Indoleacetic acids (Indomethacin/Sulindac) & Oxicams (Piroxicam):
Advantage: Long half-life permits convenient daily or twice daily dosing.
Disadvantage: Very potent; should only be used after less toxic agents fail. CNS disturbances are common. - Propionic acids (Ibuprofen, Naproxen, Ketoprofen):
Advantage: Lower toxicity and better acceptance in some patients. Less GI irritation than Aspirin.
6. Crucial NSAID Contraindications & Drug Interactions
- Absolute Contraindications: Known hypersensitivity, Third trimester of pregnancy (causes premature closure of fetal heart vessels - the ductus arteriosus), and during lactation.
- Cross Sensitivity: If a patient is allergic to ONE NSAID, there is a high increased risk of an allergic reaction with ANY OTHER NSAID.
- Use Cautiously In: Patients with bleeding disorders, renal disease, cardiovascular disease, or hepatic impairment.
- The Elderly: Highly increased risk of severe Ulcers and fatal GI bleeds in patients age 65 and above.
Drug-Drug Interactions:
- Anticoagulants (Warfarin): NSAIDs prolong bleeding time and drastically increase the bleeding effects of anticoagulants. (Clinical Scenario: An elderly man on Warfarin for atrial fibrillation takes over-the-counter Ibuprofen for knee pain. A week later, he presents to the ER vomiting blood due to a massive, uncontrollable GI bleed).
- Diuretics & Antihypertensives: NSAIDs decrease the efficacy of blood pressure medications. (Clinical scenario: A patient on BP meds starts taking Ibuprofen daily for arthritis, and suddenly their blood pressure spikes out of control because the NSAID is retaining water and constricting renal vessels).
- The "Triple Whammy" (Kidney Death): A classic fatal interaction is a patient taking an ACE Inhibitor + a Diuretic + an NSAID simultaneously. The diuretic drops blood volume, the ACEi dilates the efferent arteriole, and the NSAID clamps the afferent arteriole. The kidney's filtration pressure drops to absolute zero, causing sudden Acute Renal Failure.
7. Therapeutic Uses of Prostanoids and Analogues
While NSAIDs block prostanoids, sometimes in medicine, we actually want to give the patient synthetic prostanoids to achieve a specific physiological effect.
PGE2 and PGF2α cause powerful uterine contractions.
- First Trimester Abortion: Misoprostol (PGE1) given orally alongside Mifepristone or Methotrexate in the first few weeks. It causes softening of the cervix and uterine contraction leading to expulsion of uterine contents.
- Second Trimester (Mid-Term) Abortion: Dinoprost (PGF2α) or Carboprost (given via intra-amniotic injection). Note: Carboprost is least used for this now due to severe side effects like anaphylactic shock and cardiovascular (CVS) collapse.
- Facilitation of Labour & Cervical Priming: Dinoprostone (PGE2) is used vaginally for ripening the cervix and inducing labor at full term. Gemeprost / Demeprost / Denoproste are used vaginally for cervical priming in early pregnancy.
- Postpartum Haemorrhage (PPH): Carboprost (IM) is powerfully effective at violently contracting the uterus to clamp down on bleeding vessels and control hemorrhage after birth.
Exam Trap: Oxytocin is the Drug of Choice (DOC) for labor induction. Prostaglandins are ONLY used when Oxytocin is contraindicated (e.g., renal failure, pre-eclampsia, eclampsia). The major advantage of PGs is that they do not cause Na+ and water retention (unlike oxytocin). Side effect of PGs here: prolonged bleeding.
- Healing of Peptic Ulcers (PGE2, PGI2): Misoprostol (Oral, 200μg QD) binds to PG receptors on the parietal cell, decreasing intracellular cAMP, which decreases the activity of the proton pump (↓ Acid secretion - anti-ulcerogenic). It also ↑ Mucous & bicarbonate production to protect stomach lining, and ↑ Mucosal blood flow.
- Enoprostil is specifically used for NSAID-induced ulcers and ulcers in chronic smokers. (Side effect of Misoprostol/Enoprostil: Severe GIT discomfort and profound diarrhoea due to increased gut motility).
- Chronic Constipation: Lubiprostone. It works by activating type 2 chloride channels in the intestinal epithelial cells. This promotes secretion of Cl-, followed by passive secretion of Na+ and water, increasing stomach content liquidity. It also stimulates smooth muscle contraction to facilitate stool passage.
- To Prevent Platelet Aggregation: Epoprostenol (PGI2) is used in renal dialysis machines to prevent blood from clotting in the tubes.
- Pulmonary Arterial Hypertension: Epoprostenol and Treprostinil (IV infusion). PGI2 lowers peripheral pulmonary and coronary resistance. They increase production of cAMP → decreases levels of intracellular Ca++ → causes vascular smooth muscle to relax (vessel dilation).
- Peripheral Vascular Disease: Beraprost (Oral PGI2 given thrice a day).
- Myocardial Infarction: Iloprost (IM) decreases infarct size when given after an MI.
- Patency of Ductus Arteriosus (PDA): In neonates born with a fatal congenital heart disease (like Transposition of the Great Arteries), the ductus arteriosus must be kept open until emergency surgery can be performed to allow blood to mix. Alprostadil (PGE1) or Epoprostenol (PGI2) IV infusion is used. Side effect: maintaining patency for a long time leads to ductus fragility and rupture.
- Treating Open Angle Glaucoma: Latanoprost (PGF2α analog), Bimatoprost, Travoprost, Unoprostone (Topical drops). They physically increase the outflow of aqueous fluid from the eye via the uveoscleral pathway, relieving intraocular pressure.
- Key Side Effect: Bimatoprost causes elongation of eye lashes (hypertrichosis - excessive hair growth anywhere on the body). (Clinical Fun Fact: This "side effect" is now sold commercially as the cosmetic drug Latisse to grow long eyelashes!).
- Male Impotence (Erectile Dysfunction): Alprostadil (PGE1) via intra-cavernosal injection. Increases cAMP → ↓Ca++ → relaxes the trabecular smooth muscle and dilates cavernosal arteries, allowing blood to rush in and improving erection.
- Bronchial Asthma: Prostanoids can cause bronchodilation, but they carry a prominent cough side effect, so they are rarely preferred over standard beta-agonists.
Summary: Side Effects of Prostanoids
Prostaglandins exhibit highly dose-related adverse effects because they are intense, natural inflammatory mediators. Giving them systemically effectively gives the patient full-body inflammation symptoms:
- General: Bronchoconstriction, Hypotension, Vomiting, Diarrhoea, Fever, Dizziness, and Flushing.
- Carboprost (Intra-amniotic): Can cause extreme anaphylactic shock and CVS collapse.
- Alprostadil: Ductus fragility and rupture (if used too long in neonates).
- Misoprostol / Enoprostil: Severe GIT discomfort and diarrhea.
- PGE (Acting on EP4 receptors): Stimulates osteoclast and osteoblast activity, breaking down bone and inducing hypercalciuria (excess calcium in urine).
Quick Quiz
NSAIDs & Prostanoids Quiz
Pharmacology - mobile-friendly and focused practice.
Privacy: Your details are used only for quiz tracking and certificates.
NSAIDs & Prostanoids Quiz
Pharmacology
Preparing questions...
Choose your answer and keep your streak alive.
Great effort.
Here is your quick performance summary.
NSAIDS & Prostanoids Read More »