Introduction to Mental Health
Mental health is a state of balance between the individual and the surrounding world.
Mental health is a state of harmony between oneself and others.
Mental health is a co-existence between the realities of the self and that of other people and that of the environment.
HEALTH is a state of well being of an individual, socially, physically, mentally, not merely the absence of a disease or infirmity. (WHO)
PSYCHIATRY is a branch of medicine which deals with assessment, diagnosis and treatment of mental disorders.
"Mental health is a state of well-being in which an individual realizes his or her own abilities, can cope with the normal stresses of life, can work productively and fruitfully, and is able to make a contribution to his or her community."
To break down:
- A State of Well-being: This means that mental health is about feeling good, having a sense of purpose, and experiencing overall life satisfaction. It's not static but dynamic, fluctuating as we navigate life's challenges.
- Realizes His or Her Own Abilities: A mentally healthy person has a realistic understanding of their strengths and weaknesses. They can recognize their potential and strive to achieve it, fostering self-esteem and self-efficacy.
- Cope with the Normal Stresses of Life: Life inevitably brings challenges, disappointments, and pressures. Mental health equips us with resilience – the ability to adapt, recover, and grow stronger in the face of adversity. This doesn't mean being stress-free, but rather having effective strategies to manage stress.
- Can Work Productively and Fruitfully: This refers to the ability to engage in meaningful activities, whether it's employment, education, caregiving, or creative pursuits. It encompasses concentration, motivation, problem-solving, and a sense of accomplishment.
- Is Able to Make a Contribution to His or Her Community: Mental health enables individuals to form meaningful relationships, participate in social life, and contribute positively to their families, friendships, and broader society. It's about a sense of belonging and connectedness.
Distinguishing Mental Health from Mental Illness
- Mental Health: As discussed, this is a state of optimal psychological and emotional well-being. Someone can have good mental health even if they experience occasional stress, sadness, or anxiety, as long as they can cope effectively and maintain overall functioning.
- Mental Illness (or Mental Disorder): This refers to a wide range of conditions that affect mood, thinking, and behavior. Mental illnesses are characterized by significant distress, impairment in daily functioning, and often require diagnosis and treatment. They are not merely temporary reactions to stress or personal weaknesses.
Key Differences:
| Feature |
Mental Health |
Mental Illness |
| State |
State of well-being, thriving |
Diagnosable condition affecting thinking, mood, or behavior |
| Coping |
Effective coping with life's stresses |
Difficulty coping, significant distress |
| Functioning |
Productive, fruitful, contributes to community |
Significant impairment in social, occupational, or other important areas of functioning |
| Presence of Symptoms |
May experience normal fluctuations in mood/stress |
Presence of persistent, often distressing symptoms (e.g., hallucinations, severe depression, extreme anxiety) |
| Duration |
Dynamic, but generally stable functioning |
Prolonged or recurrent, often requires professional intervention |

Characteristics of a Mentally Healthy Person
Building on the WHO definition, a mentally healthy individual typically exhibits several key characteristics:
- Positive Self-Concept: Possesses a realistic and generally positive view of themselves, including their strengths and limitations.
- Sense of Identity: Has a clear understanding of who they are, their values, and their purpose.
- Autonomy and Independence: Capable of making their own decisions and taking responsibility for their actions, while also recognizing the importance of interdependence.
- Resilience: Ability to bounce back from adversity, adapt to change, and learn from difficult experiences.
- Emotional Regulation: Can recognize, understand, and appropriately express emotions (both positive and negative) without being overwhelmed by them.
- Effective Coping Strategies: Has a repertoire of healthy ways to manage stress, problem-solve, and deal with challenges.
- Meaningful Relationships: Capable of forming and maintaining healthy, reciprocal relationships based on trust, empathy, and respect.
- Purpose and Direction: Finds meaning in life, sets goals, and works towards achieving them, contributing to a sense of fulfillment.
- Adaptability: Can adjust to new situations, unexpected events, and changing circumstances.
- Realistic Perception of Reality: Able to differentiate between reality and fantasy, and make sound judgments.

Stress in Mental Health
Stress is a natural and often unavoidable part of life. It's essentially your body's way of responding to any kind of demand or threat. When you perceive a threat – whether it's physical (like a near-miss car accident) or psychological (like a looming deadline or a difficult conversation) – your body initiates a "fight-or-flight" response.
- Stress: Is a stimulus or demand that generates disruption in homeostasis or produces a reaction.
- Stress: Is a state of disequilibrium that occurs when there is a disharmony between demands occurring within an individual’s internal and external environment and his or her ability to cope with those demands.
- Stressor: a demand from within an individual’s internal and external environment that elicits a physiological and or psychological response.
- Stressor: is a source of stress.
Stress can produce adaptive and maladaptive responses.
This physiological reaction involves:
- Release of hormones: Adrenaline and cortisol flood your system.
- Increased heart rate and blood pressure.
- Rapid breathing.
- Muscle tension.
- Sharpened senses.
Historically, this response was necessary for survival, enabling our ancestors to react quickly to danger. In modern life, however, many of our stressors are not physical threats but ongoing psychological pressures.
Eustress vs. Distress:
- Eustress (Good Stress): This is positive, short-term stress that can motivate us, enhance performance, and help us achieve goals. Examples include the excitement of a new job, the challenge of learning a new skill, or the anticipation of a big event. Eustress is invigorating and can lead to personal growth.
- Distress (Bad Stress): This is negative stress that can be overwhelming, prolonged, and detrimental to health. It occurs when demands exceed our perceived ability to cope. Examples include chronic work pressure, relationship problems, financial difficulties, or major life changes (e.g., loss of a loved one). Distress can lead to burnout, anxiety, depression, and various physical health problems.
Impact of Stress on Mental Well-being:
While short-term stress can be adaptive, chronic or overwhelming distress can significantly impair mental well-being. It can lead to:
- Emotional Symptoms: Irritability, mood swings, anxiety, depression, feelings of being overwhelmed, difficulty relaxing, low self-esteem.
- Cognitive Symptoms: Difficulty concentrating, memory problems, negative thinking, impaired judgment, excessive worry.
- Behavioral Symptoms: Social withdrawal, changes in eating habits (overeating or undereating), sleep disturbances (insomnia or hypersomnia), increased use of substances (alcohol, drugs), procrastination, fidgeting.
- Physical Symptoms (linked to mental impact): Headaches, muscle tension, digestive problems, fatigue, weakened immune system.
Responses to Stress
Individuals react to stress in a myriad of ways, influenced by their unique genetic makeup, past experiences, coping mechanisms, and the nature of the stressor. Responses can be categorized broadly:
- Physiological Responses:
- Fight-or-Flight: The immediate, automatic response involving the sympathetic nervous system (increased heart rate, blood pressure, muscle tension, rapid breathing).
- General Adaptation Syndrome (GAS) - Hans Selye: A three-stage model describing the body's long-term response to stress:
- Alarm Reaction: Initial shock, fight-or-flight response.
- Stage of Resistance: The body tries to cope with the stressor, maintaining elevated physiological responses but attempting to return to normal. Resources are gradually depleted.
- Stage of Exhaustion: If stress is prolonged, the body's resources are depleted, leading to weakened immunity, fatigue, and increased vulnerability to illness and disease (both physical and mental).
- Emotional Responses:
- Anxiety: Feelings of unease, worry, nervousness, apprehension.
- Anger/Irritability: Frustration, resentment, short temper.
- Sadness/Depression: Feelings of hopelessness, helplessness, loss of interest.
- Fear: Response to perceived danger or threat.
- Overwhelm: Feeling swamped, unable to cope.
- Cognitive Responses:
- Negative self-talk: "I can't do this," "I'm not good enough."
- Rumination: Repetitive thinking about a stressor.
- Catastrophizing: Blowing problems out of proportion.
- Difficulty concentrating or making decisions.
- Memory impairment.
- Behavioral Responses:
- Adaptive/Healthy: Exercise, seeking social support, engaging in hobbies, problem-solving, relaxation techniques (meditation, deep breathing), healthy diet, adequate sleep.
- Maladaptive/Unhealthy: Social withdrawal, aggression, substance abuse, excessive eating or undereating, procrastination, avoidance, excessive sleeping, lashing out at others.
Determinants of Response to Stress
Why do some people thrive under pressure while others crumble? The way an individual responds to stress is determined by a complex interplay of factors:
- Perception of the Stressor (Appraisal):
- Primary Appraisal: Is this event a threat, a challenge, or irrelevant?
- Secondary Appraisal: Do I have the resources to cope with this threat/challenge?
- If a situation is appraised as highly threatening and resources are perceived as insufficient, the stress response will be more intense and negative.
- Coping Mechanisms:
- Problem-focused coping: Directly addressing the source of stress (e.g., studying for an exam, creating a budget).
- Emotion-focused coping: Managing the emotional reaction to stress (e.g., meditation, talking to a friend, exercise).
- The effectiveness and healthiness of coping strategies significantly influence outcomes.
- Individual Differences:
- Genetics: Some individuals may be genetically predisposed to higher stress reactivity.
- Personality:
- Resilience: The ability to adapt and recover from adversity.
- Hardiness: Commitment, control, and challenge (seeing stressors as opportunities).
- Optimism vs. Pessimism.
- Self-efficacy: Belief in one's ability to succeed.
- Temperament: Innate behavioral and emotional patterns.
- Social Support:
- A strong network of family, friends, and community provides emotional, informational, and practical support, acting as a buffer against stress.
- Lack of social support can exacerbate the negative effects of stress.
- Past Experiences and Learning:
- Previous encounters with similar stressors, and how they were handled, shape current responses.
- Traumatic experiences can lead to heightened stress responses.
- Physical Health Status:
- Underlying chronic illnesses, poor nutrition, lack of sleep, or substance abuse can deplete energy reserves and reduce the body's ability to cope with stress.
- Environmental Factors:
- Socioeconomic status, living conditions, access to resources, cultural background, and exposure to chronic environmental stressors (e.g., noise, pollution, violence) can all impact stress levels and coping abilities.

MENTAL ILLNESS
Mental illness is the maladjustment in living. The inability to cope with stress and environment.
It produces a disharmony in the person’s ability to meet human needs comfortably or effectively and function with culture
Mentally ill person loses his ability to respond according to the expectations he has for himself and the demands that society has for him
In general an individual may be considered to be mentally ill if
- The personal behavior is causing distress to self and others
- The person’s behavior is causing disturbance in his day-to-day activities, job and interpersonal relationships
Key aspects of mental illness include:
- Significant Distress: The individual experiences profound emotional pain, discomfort, or suffering that is disproportionate to circumstances or is persistent over time. This distress can manifest as sadness, anxiety, anger, confusion, or other intense negative emotions.
- Impairment in Functioning: The condition significantly interferes with one or more major life activities. This could include:
- Social Functioning: Difficulty maintaining relationships, social withdrawal, inability to interact appropriately.
- Occupational/Academic Functioning: Inability to work, perform daily tasks, attend school, or maintain employment.
- Self-Care: Neglect of personal hygiene, eating, or other basic needs.
- Role Performance: Inability to fulfill roles as a parent, spouse, student, or employee.
- Deviation from Norms: The thoughts, feelings, or behaviors are significantly outside of what is culturally expected or considered typical. This deviation must be considered within a cultural context, as what is "normal" can vary.
- Duration and Persistence: Unlike transient mood changes or reactions to stress, the symptoms of mental illness are usually persistent over a certain period, not just a brief episode.
Common Signs and Symptoms of Mental Illness
It's important to remember that experiencing one or two of these symptoms does not necessarily mean a person has a mental illness.
- Changes in Mood:
- Persistent sadness or irritability: Lasting for weeks or months, not just a day or two.
- Loss of interest or pleasure: In activities once enjoyed (anhedonia).
- Extreme mood swings: Rapid shifts from extreme happiness to extreme sadness or anger.
- Feelings of hopelessness or helplessness.
- Elevated mood, euphoria, or grandiosity: Unusually high energy, racing thoughts, reduced need for sleep (can indicate mania).
- Changes in Thinking and Perception:
- Difficulty concentrating or focusing: Problems paying attention or easily distracted.
- Memory problems: Significant, unexplainable forgetfulness.
- Confused thinking: Disorganized thoughts, difficulty following conversations.
- Paranoia: Unreasonable suspicion or distrust of others.
- Delusions: False beliefs not based in reality (e.g., belief that one is being persecuted or has special powers).
- Hallucinations: Hearing, seeing, smelling, tasting, or feeling things that are not there (e.g., hearing voices).
- Obsessive thoughts: Repetitive, intrusive, unwanted thoughts.
- Changes in Behavior:
- Social withdrawal: Avoiding friends, family, or social activities.
- Changes in sleep patterns: Insomnia (difficulty sleeping), hypersomnia (sleeping too much), or disturbed sleep.
- Changes in appetite or weight: Significant weight loss or gain.
- Decreased energy or fatigue: Feeling constantly tired and lacking motivation.
- Increased agitation or restlessness: Inability to sit still, pacing.
- Neglect of personal hygiene: Not showering, grooming, or changing clothes.
- Impulsive or risky behavior: Excessive spending, reckless driving, substance abuse.
- Aggression or violence.
- Suicidal thoughts or self-harm behaviors.
- Physical Symptoms (without a clear medical cause):
- Unexplained aches and pains: Headaches, stomach aches, muscle tension.
- Digestive problems: Nausea, diarrhea, constipation.
- Fatigue.
PROBLEMS ASSOCIATED WITH MENTAL DISODERS
Profound Impairments in Daily Functioning:
- Self-care limitations: Individuals may struggle with basic hygiene, nutrition, and personal upkeep.
- Impaired functioning: This can manifest as difficulty maintaining employment, managing finances, or fulfilling household responsibilities.
- Significant deficits in biological, emotional, and cognitive functioning: These can include disruptions in sleep patterns, appetite, mood regulation, memory, attention, and problem-solving abilities.
Disability and Life-Process Changes:
- Mental disorders can lead to long-term disability, preventing individuals from engaging in typical life activities.
- They can alter major life trajectories, impacting educational attainment, career progression, and the formation of meaningful relationships.
Intense Emotional Distress and Dysregulation:
- Pervasive emotional problems: These include chronic anxiety, overwhelming sadness, debilitating anger, profound loneliness, and prolonged grief that can be disproportionate to life events.
- Emotional lability: Rapid and intense shifts in mood can make daily life unpredictable and challenging.
Co-occurring Physical Health Issues:
- Somatization: Mental distress can manifest as physical symptoms, such as chronic pain, fatigue, headaches, and digestive problems, often without clear medical explanation.
- Increased risk of physical illnesses: Individuals with mental disorders are at a higher risk for cardiovascular disease, diabetes, and other chronic conditions, partly due to lifestyle factors, medication side effects, and physiological stress responses.
Distortions in Perception, Thought, and Communication:
- Alterations in thinking: This can include delusional beliefs, disorganized thought processes, and difficulty with abstract reasoning.
- Distorted perception: Hallucinations (auditory, visual, etc.) can significantly impact an individual's reality.
- Communication difficulties: Disorganized speech, reduced verbal output, or an inability to express thoughts coherently can hinder social interaction.
- Impaired decision-making: Cognitive deficits can make it challenging to make sound judgments and plan for the future.
Challenges in Interpersonal Relationships:
- Difficulties relating to others: Mental illness can strain existing relationships and make it hard to form new ones due to social withdrawal, paranoia, irritability, or communication barriers.
- Social isolation and stigma: The misunderstanding and prejudice surrounding mental illness can lead to ostracization and loneliness.
Risk to Self and Others:
- Dangerous behaviors: In some cases, mental disorders can lead to self-harm, suicidal ideation, or, rarely, aggression towards others, particularly when psychosis or severe mood disturbances are present.
Widespread Adverse Effects:
- Individual well-being: Mental illness significantly diminishes an individual's quality of life, sense of purpose, and overall happiness.
- Family burden: Families often experience immense emotional, financial, and logistical strain as they try to support a loved one with a mental disorder.
- Community impact: Untreated mental illness can contribute to homelessness, crime, and a reduced workforce productivity, impacting societal well-being and economic stability.
Significant Life Domain Problems:
- Financial problems: Loss of employment, healthcare costs, and inability to manage finances can lead to severe financial hardship.
- Marital and family discord: Mental illness can be a major source of conflict, divorce, and family breakdown.
- Academic and occupational setbacks: Difficulty concentrating, maintaining attendance, and performing tasks can lead to school dropout and job loss.

Etiology of Mental Illness
Many factors are responsible for the causation of mental illness. These factors may predispose an individual to mental illness, precipitate or perpetuate the mental illness
- Predisposing Factors: These are long-term, underlying vulnerabilities that increase an individual's susceptibility to developing a mental illness. They set the stage, often present for extended periods or even from birth.
Examples:
- Genetic make-up: Inherited predispositions, not the illness itself, but a heightened vulnerability. Studies highlight the significant role of heredity in mental health conditions (e.g., three-fourths of mental defectives and one-third of psychotic individuals owing their condition mainly to unfavorable heredity).
- Physical damage to the central nervous system: Chronic or congenital neurological impairments.
- Adverse psychological influences: Early childhood trauma, developmental issues, or chronic maladaptive learned behaviors.
- Precipitating Factors: These are acute, immediate stressors or events that trigger the onset of a mental illness in a vulnerable individual. They often occur shortly before the symptoms emerge.
Examples:
- Physical stress: Acute illness, injury, or other physical demands on the body.
- Psychosocial stress: Significant life events such as bereavement, job loss, relationship breakdown, academic failure, or trauma.
- Perpetuating Factors: These are factors that maintain, aggravate, or prolong a mental illness once it has developed. They make it harder for the individual to recover or can lead to symptom exacerbation.
Examples:
- Psychological stress: Ongoing, unresolved stress can prevent recovery and worsen existing symptoms.
- Other examples could include lack of social support, financial difficulties, substance abuse, stigma, or inadequate treatment.
OTHER FACTORS;
A. Biological Factors
These involve genetic, neurochemical, structural, and physiological aspects of the body, particularly the brain.
- Heredity (Genetic Make-up):
- Mental illnesses are not typically inherited directly, but a predisposition or vulnerability can be passed down through genes. This means an individual might inherit a higher risk, but whether the illness develops often depends on the interaction with environmental and psychological factors.
- As you noted, studies indicate a significant genetic component in conditions like intellectual disability ("mental defectives") and psychoses.
- Biochemical Factors (Neurotransmitters):
- Disturbances in the balance or functioning of neurotransmitters (chemical messengers in the brain) are strongly implicated in various psychiatric disorders.
- Examples include:
- Dopamine: Linked to schizophrenia (excess) and Parkinson's disease (deficiency), also involved in reward pathways.
- Serotonin: Associated with depression and anxiety (deficiency).
- Norepinephrine (Noradrenaline): Involved in mood, arousal, and attention (imbalances linked to depression and anxiety).
- GABA: The primary inhibitory neurotransmitter (deficiency linked to anxiety disorders).
- Brain Damage / Structural and Functional Alterations:
- Any insult or damage to the brain can affect its structure and function, leading to mental health symptoms.
- Causes include:
- Infection: Neurosyphilis, encephalitis, HIV infection (can lead to neurocognitive disorders).
- Injury: Traumatic Brain Injury (TBI) from head injury, leading to cognitive, emotional, and behavioral changes.
- Intoxication: Damage from toxins like alcohol, barbiturates, lead, recreational drugs, or even certain medications.
- Vascular Issues: Poor blood supply (ischemia), bleeding (intracranial hemorrhage), or stroke, which can impair brain function.
- Alteration in Brain Function: Changes in blood chemistry (e.g., severe hypoglycemia, hypoxia/anoxia, electrolyte imbalances) that directly interfere with neuronal activity.
- Tumors: Brain tumors can cause a range of psychiatric symptoms depending on their location and size.
- Nutritional Deficiencies: In particular, B-complex vitamin deficiencies (e.g., B1, B3, B12) can lead to neurological and psychiatric symptoms (e.g., Wernicke-Korsakoff syndrome from thiamine deficiency).
- Degenerative Diseases: Conditions like Alzheimer's disease and other dementias involve progressive brain cell death, leading to cognitive and behavioral decline.
- Endocrine Disturbances:
- Hormonal imbalances can profoundly affect mood and cognition.
- Examples: Hypothyroidism (can mimic depression), hyperthyroidism (can cause anxiety, irritability), adrenal gland disorders.
- Physical Defects and Illnesses:
- Both acute and chronic physical illnesses can lead to mental health issues through various mechanisms:
- Direct physiological impact: The illness itself affecting brain function.
- Psychological distress: Coping with pain, disability, loss of function, or life-threatening diagnoses.
- Medication side effects.
- Physiological Changes at Critical Life Periods:
- Periods of significant hormonal flux and physiological change can increase vulnerability to mental illness due to their impact on neurochemistry and the added psychological demands.
- Examples: Puberty, menstruation (PMDD), pregnancy, delivery, puerperium (postpartum depression/psychosis), and climacteric (menopause).
B. Psychological Factors
These factors relate to an individual's thoughts, feelings, learning experiences, personality, and coping styles.
- Personality Types and Vulnerability:
- Certain personality traits or types may increase susceptibility to specific disorders under stress.
- Example: Individuals with schizoid personality traits (unsocial, reserved) may be more vulnerable to schizophrenia when facing significant adverse situations and psychosocial stress. Other examples include obsessive-compulsive traits leading to OCD, or anxious traits predisposing to anxiety disorders.
- Strained Interpersonal Relationships:
- Ongoing conflict and negativity in significant relationships can be a major source of psychological distress.
- Examples: Strained relationships at home, work, school, or college can erode self-esteem and lead to feelings of isolation and anxiety.
- Significant Life Events and Loss:
- Bereavement: The death of a loved one.
- Loss of prestige or social standing.
- Loss of employment/job: Can lead to financial stress, loss of identity, and purpose.
- Childhood Insecurities and Developmental Trauma:
- Early life experiences play a crucial role in shaping mental health.
- Examples:
- Parental psychopathology: Parents with their own mental health issues or maladaptive coping.
- Faulty parenting styles: Over-strictness, over-leniency, inconsistent discipline.
- Abnormal parent-child relationships: Over-protection (hinders independence), rejection (leads to feelings of worthlessness), unhealthy comparisons between siblings.
- Deprivation of essential needs: Lack of love, security, stimulation, or consistent care.
- Childhood abuse (physical, emotional, sexual) and neglect are profound risk factors for nearly all mental health disorders.
- Social and Recreational Deprivations:
- Lack of engaging activities, social connection, and opportunities for enjoyment can lead to boredom, isolation, loneliness, and feelings of alienation, contributing to depression and anxiety.
- Marriage Problems:
- Marital discord, forced relationships, disharmony due to incompatibility (physical, emotional, social, educational, financial), and issues like childlessness or having too many children can be significant stressors.
- Sexual Difficulties:
- Problems arising from improper sex education, unhealthy attitudes towards sexual functions, guilt feelings (e.g., about masturbation), pre- and extramarital sexual relations, and worries about sexual identity or "perversions" can lead to significant psychological distress and contribute to mental health issues.
- Stress, Frustration, and Environmental Variations:
- Chronic psychological stress and frustration deplete coping resources.
- Climatic conditions and seasonal variations: Conditions like Seasonal Affective Disorder (SAD) demonstrate how environmental factors can trigger mood disturbances.
C. Social Factors
These are broad societal and cultural influences that affect an individual's mental health.
- Socioeconomic Disadvantage:
- Poverty: Associated with chronic stress, lack of resources, poor nutrition, and limited access to healthcare.
- Unemployment: Leads to financial strain, loss of purpose, social isolation, and reduced self-esteem.
- Injustice and Inequality: Experiences of discrimination, systemic oppression, and lack of fairness.
- Environmental and Community Stressors:
- Insecurity: Living in unstable or unsafe environments (e.g., high crime areas).
- Migration: The stress of adapting to a new culture, language barriers, and loss of social networks.
- Urbanization: Can lead to overcrowding, social isolation despite proximity, and increased sensory stimulation.
- Social Disruptions and Deviance:
- Gambling, Alcoholism, Prostitution: These are often both symptoms of underlying distress and factors that perpetuate mental health problems.
- Broken homes, Divorce: Disruption of family structure, leading to instability and emotional distress, especially for children.
- Very big family: Can mean stretched resources, less individual attention, and increased stress for caregivers.
- Cultural and Political Influences:
- Religion and traditions: Can be sources of support or, in some cases, conflict and guilt.
- Political upheavals and other social crises: Wars, natural disasters, economic depressions create widespread trauma and stress.

CLASSIFICATION OF MENTAL ILLNESS
It’s important to classify mental illness because it serves as a guide to Diagnosis and prognosis (outcome). In psychiatry classification is based on clinical description of disease.
I. Classification Systems for Mental Disorders
To ensure consistent diagnosis, facilitate research, and guide treatment, mental health professionals rely on standardized classification systems. The two most widely used internationally are:
- Diagnostic and Statistical Manual of Mental Disorders (DSM):
- Published by the American Psychiatric Association (APA).
- Currently in its fifth edition, revised text (DSM-5-TR).
- Primarily used in the United States and heavily influences psychiatric practice globally.
- Provides explicit diagnostic criteria for hundreds of mental disorders, along with descriptive text, prevalence rates, and risk factors.
- It is atheoretical regarding etiology, meaning it describes disorders based on observable symptoms rather than endorsing a particular theory of causation.
Key Classifications in DSM-5:
- Neurodevelopmental Disorders: Autism spectrum disorder, ADHD, intellectual disabilities.
- Schizophrenia Spectrum & Other Psychotic Disorders: Delusional disorder, schizophrenia, brief psychotic disorder.
- Bipolar & Related Disorders: Bipolar I and II, cyclothymic disorder.
- Depressive Disorders: Major depressive disorder, disruptive mood dysregulation disorder, premenstrual dysphoric disorder.
- Anxiety Disorders: Generalized anxiety disorder, panic disorder, social anxiety.
- Obsessive-Compulsive & Related Disorders: OCD, hoarding disorder, body dysmorphic disorder.
- Trauma- & Stressor-Related Disorders: PTSD, acute stress disorder, adjustment disorders.
- Dissociative Disorders: Dissociative identity disorder, depersonalization/derealization.
- Somatic Symptom & Related Disorders: Somatic symptom disorder, illness anxiety disorder, conversion disorder.
- Feeding & Eating Disorders: Anorexia nervosa, bulimia nervosa, binge-eating.
- Disruptive, Impulse-Control, & Conduct Disorders: Oppositional defiant disorder, conduct disorder.
- Substance-Related & Addictive Disorders: Alcohol, cannabis, stimulant-related disorders.
- Personality Disorders: Antisocial, borderline, narcissistic personality disorders.
- International Classification of Diseases (ICD):
- Published by the World Health Organization (WHO).
- Currently in its 11th revision (ICD-11).
- Covers all health conditions, including mental and behavioral disorders.
- Used globally for health statistics, epidemiology, and clinical purposes, especially outside the U.S.
- While there are differences, the DSM and ICD systems are increasingly harmonized to allow for better international comparability of diagnostic data.
II. General Classifications of Mental Illness
Historically, and sometimes still colloquially, mental illnesses have been broadly grouped. While modern classification systems offer more nuance, understanding these general categories can be helpful:
- Organic vs. Functional Mental Disorders:
- Organic Mental Disorders: These are conditions where a clear physical or physiological cause (e.g., brain injury, infection, substance intoxication, neurological disease) can be identified as directly causing the mental symptoms. Examples: Delirium, Dementia (e.g., Alzheimer's type), Substance-Induced Psychotic Disorder.
- Functional Mental Disorders: These are conditions where no clear organic or physical cause has been identified, and symptoms are believed to arise primarily from psychological, social, and genetic vulnerabilities. Most major psychiatric disorders (e.g., Schizophrenia, Major Depressive Disorder, Anxiety Disorders) traditionally fall into this category, though growing research often reveals subtle biological underpinnings.
- Neurosis vs. Psychosis: This is a historical distinction that is less used in formal diagnosis today but remains useful in understanding the severity and nature of impairment.
- Neurosis (Neurotic Disorders):
- Core Characteristics: Characterized by significant distress, anxiety, fear, and/or maladaptive behaviors, but the individual generally retains a grasp on reality. They understand that their thoughts or feelings are problematic, and their personality remains largely intact.
- Common Examples: Most anxiety disorders (e.g., Generalized Anxiety Disorder, Panic Disorder, Phobias), Obsessive-Compulsive Disorder (OCD), Post-Traumatic Stress Disorder (PTSD), and mild to moderate depressive disorders.
- Impact: Can cause significant impairment and suffering, but the individual usually maintains some level of social and occupational functioning, and there is no loss of contact with reality.
- Psychosis (Psychotic Disorders):
- Core Characteristics: Defined by a significant loss of contact with reality. Individuals experiencing psychosis have difficulty distinguishing between what is real and what is not. This often involves profound disturbances in thought, perception, emotion, and behavior.
- Key Symptoms:
- Delusions: Fixed, false beliefs not amenable to change in light of conflicting evidence (e.g., believing one is being persecuted, or that one has special powers).
- Hallucinations: Sensory experiences that occur in the absence of an external stimulus (e.g., hearing voices, seeing things that aren't there).
- Disorganized Thinking (Speech): Inferred from speech, which may be illogical, incoherent, or derail from topic to topic.
- Grossly Disorganized or Abnormal Motor Behavior: Catatonia (ranging from stupor to agitation) or other unusual movements.
- Negative Symptoms: Absence of normal mental functions (e.g., diminished emotional expression, avolition - decrease in motivated self-initiated purposeful activities).
- Common Examples: Schizophrenia, Bipolar Disorder (during manic or depressive episodes with psychotic features), Severe Depressive Disorder with Psychotic Features, Substance-Induced Psychotic Disorder.
- Impact: Can lead to severe functional impairment, often requiring hospitalization and significant support.

General Symptomatology of Mental Disorders
Mental disorders often manifest as exaggerated, distorted, or significantly atypical patterns of normal behavior and experience that cause distress or impair functioning. These deviations can occur across various domains, including mood, beliefs, perception, awareness, memory, and physical presentation.
Individuals experiencing mental disorders may sometimes present with non-specific physical complaints, such as persistent, unexplained headaches, or a general sense of malaise and poor health that lacks a clear medical explanation. There might also be a noticeable change in their typical engagement with work, school, or other gainful economic activities.
The signs and symptoms of mental disorders can be observed through various lenses, including a person's appearance, behavior, patterns of movement, and speech.
I. Observable Signs
- Appearance: A person's physical appearance can offer significant clues about their mental state. Individuals with certain mental disorders may exhibit:
- Poor grooming and hygiene: This can range from disheveled hair, unkempt clothing, and dirty nails to a complete neglect of personal care.
- Example: A person with severe depression might stop showering or changing clothes for days; someone experiencing psychosis might wear multiple layers of inappropriate clothing regardless of the weather.
- Unusual attire: Clothing that is mismatched, inappropriate for the weather, or bizarre in style.
- Behavior: Behavior refers to how an individual acts and reacts to their environment and social situations. Deviations from typical behavior can include:
- Social Withdrawal: Avoiding interaction with others, isolating oneself.
- Example: A person with social anxiety disorder might consistently decline invitations, or someone with depression might stay in bed all day.
- Hostility or Aggression: Verbal or physical aggression, irritability, or an argumentative demeanor.
- Example: A person experiencing a manic episode might become easily enraged or lash out at others with little provocation.
- Uncommunicativeness: Reluctance or inability to engage in conversation, providing minimal responses.
- Guardedness/Suspiciousness: Being overly cautious, distrustful of others, or secretive.
- Example: Someone with paranoid delusions might believe others are conspiring against them and refuse to share personal information.
- Disinhibition: Lack of impulse control, acting without considering consequences.
- Example: A person in a manic state might engage in reckless spending or inappropriate sexual behavior.
- Agitation: Restlessness, inability to sit still, increased motor activity.
- Disorders of Movement: These symptoms relate to the way individuals move their limbs and body, and can indicate underlying neurological or psychiatric conditions.
- Psychomotor Retardation: A noticeable slowing of movement and speech, appearing sluggish and lethargic.
- Example: Common in severe depression, where even simple tasks feel effortful.
- Akathisia (Restlessness): An inner sense of restlessness that compels continuous movement; the person cannot sit still. This is different from general restlessness in that it's a specific, often distressing, motor symptom.
- Example: A side effect of certain antipsychotic medications, where the person constantly shifts position, taps their feet, or paces.
- Echopraxia: Involuntarily imitating the movements or gestures of another person.
- Example: A symptom seen in some psychotic disorders, where the person mirrors the interviewer's actions.
- Stereotypies: Repetitive, seemingly purposeless movements (e.g., body rocking, head banging) that don't serve a goal.
- Pacing: Repeatedly walking back and forth in a confined space.
- Involuntary Movements:
- Tremors: Rhythmic, involuntary muscle contractions, causing shaking.
- Tics: Sudden, rapid, recurrent, non-rhythmic motor movements or vocalizations (e.g., eye blinking, head jerking, throat clearing).
- Bizarre Posturing/Mannerisms: Involuntarily maintaining an abnormal or exaggerated body position for an extended period, or performing idiosyncratic, stylized movements.
- Example: Catatonia, where a person might hold an unusual pose for hours; grimacing, or odd gestures that seem out of context.
II. Disturbances in Speech
Speech patterns are crucial indicators of mental state, reflecting thought processes, mood, and cognitive function. Disturbances can affect the speed, volume, appropriateness, and coherence of verbal communication.
- Speed of Speech:
- Pressured Speech (Extremely Rapid): Speaking excessively quickly, often loudly, and sometimes unintelligibly, as if words are being forced out. The person may interrupt frequently and be difficult to interrupt.
- Example: A classic sign of mania, where thoughts are racing.
- Slowed Speech (Bradyarthria/Slurred Speech): Speaking unusually slowly, sometimes with reduced articulation or volume.
- Example: Common in depression or in conditions affecting motor control like Parkinson's disease.
- Volume of Speech:
- Hypophonia (Low Volume/Whispered): Speech that is unusually quiet or whispered, even in normal conversational settings.
- Example: Can be seen in severe depression or sometimes in schizophrenia.
- Inappropriately Loud Speech: Speaking at a volume that is much louder than warranted by the situation.
- Example: A person in a manic episode might shout or talk very loudly without realizing it.
- Absence of Speech:
- Mutism: Complete absence of speech, despite being physically capable of speaking.
- Example: Can occur in severe depression, catatonic states, or some anxiety disorders (selective mutism).
- Appropriateness of Speech:
- Irrelevant/Inappropriate Content: Speech that deviates significantly from the topic, or is logically disconnected from the conversation.
- Example: Responding to a question about their day with a detailed account of a conspiracy theory unrelated to the conversation.
- Specific Speech Disturbances:
- Echolalia: Involuntarily repeating words or phrases spoken by another person (like an echo).
- Example: A symptom seen in some individuals with autism spectrum disorder or psychotic disorders.
- Latency of Response: Taking an unusually long time to answer questions or respond to conversation.
- Example: Characteristic of slowed thinking in depression.
- Word Salad (Incoherence): A jumble of seemingly random words and phrases that have no logical connection, making the speech unintelligible.
- Example: "The sun is blue, and apples fly on the carousel, purple elephants sing." Often seen in disorganized schizophrenia.
- Neologisms: Inventing new words or phrases that have meaning only to the individual, and are not understandable by others.
- Example: A person might refer to their phone as a "thought-box" or a "mind-squeezer." Also common in psychotic disorders.
- Clang Associations: Speech driven by the sound of words rather than their meaning, often rhyming or alliterative.
- Example: "The train pain, it went in the rain, on the plain."
III. Mood and Affect
Mood refers to a person's sustained, pervasive internal emotional state, which colors their perception of the world and influences their behavior. It's often described by the individual themselves (e.g., "I feel sad" or "I feel joyful").
In mental disorders, mood can be significantly dysregulated:
- Elevated/Elated Mood: Characterized by extreme happiness, euphoria, or an exaggerated sense of well-being, often out of proportion to circumstances.
- Example: The persistent, elevated mood experienced during a manic episode in Bipolar Disorder.
- Depressed Mood: Characterized by extreme sadness, hopelessness, anhedonia (loss of pleasure), or a general feeling of misery.
- Example: The pervasive sadness and lack of interest in activities common in Major Depressive Disorder.
- Irritable Mood: Easily annoyed, frustrated, or prone to anger, often disproportionately so.
- Example: A person in a manic or hypomanic episode might become irritable when their plans are thwarted.
Affect is the external, observable expression of emotion. It's the way a person's mood appears to others. Clinicians assess affect based on its range, intensity, appropriateness, and stability.
Affective presentations in mental disorders can include:
- Normal (Euthymic) Affect: A wide range of emotional expression that is appropriate to the situation and content of speech.
- Elevated Affect: An expression of extreme cheerfulness or euphoria.
- Depressed Affect: An expression of sadness, gloom, or despondency.
- Labile Affect: Rapid, often abrupt, shifts in emotional expression, alternating quickly between extremes (e.g., crying one moment, laughing the next).
- Example: Seen in Borderline Personality Disorder or some neurological conditions.
- Inappropriate Affect: Emotional expression that is incongruent with the situation or the person's thoughts.
- Example: Laughing when describing a tragic event, often seen in psychotic disorders.
- Constricted/Restricted Affect: A mild reduction in the range and intensity of emotional expression.
- Blunted Affect: A significant reduction in the intensity of emotional expression; emotions are present but dulled.
- Flat Affect: A near or total absence of emotional expression, with a monotone voice and immobile facial features.
- Example: A common negative symptom of schizophrenia, where the person shows little to no emotional response.
IV. Perception
Perception is the process through which we interpret sensory information from our environment via our five senses (touch, taste, hearing, smell, sight). Mental disorders can distort these processes, leading to experiences that deviate from reality.
Key perceptual disturbances include:
- Illusions:
- A misinterpretation or distortion of an actual external sensory stimulus. The stimulus is real, but the interpretation is incorrect.
- Example: Mistaking a shadow for an intruder in a dimly lit room, or perceiving patterns in wallpaper as faces. Illusions can occur in normal individuals under certain conditions (e.g., fatigue, fear) but are more frequent and persistent in some mental disorders (e.g., delirium, psychosis). When associated with other symptoms, they can be indicative of a mental disorder.
- Hallucinations:
- A perception-like experience that occurs without an external stimulus. They are vivid and clear, with the full force and impact of normal perceptions, and are not under voluntary control.
- Hallucinations can occur in any sensory modality:
- Auditory Hallucinations: Hearing voices, sounds, or noises that no one else can hear. This is the most common type of hallucination in psychotic disorders.
- Example: Hearing critical, commanding, or conversing voices when no one is present.
- Visual Hallucinations: Seeing things (people, objects, patterns) that are not actually there.
- Example: Seeing deceased relatives, flashing lights, or distorted figures.
- Tactile Hallucinations: Feeling sensations on or under the skin without any physical cause.
- Example: Feeling insects crawling on the skin (formication) or a burning sensation.
- Olfactory Hallucinations: Smelling odors that are not present.
- Example: Smelling smoke, rotten food, or pleasant fragrances when there is no source.
- Gustatory Hallucinations: Experiencing a taste in the mouth without any food or drink.
- Example: A persistent bitter, metallic, or unpleasant taste.
V. Thinking
Thinking encompasses the mental processes involved in acquiring, processing, storing, and using information. Disturbances in thinking are central to many mental disorders and can affect the stream, form, and content of thoughts.
- Stream of Thought (Pace and Quantity): Refers to the amount and speed of thoughts an individual experiences and reports.
- Pressure of Thought: Thoughts come so rapidly and abundantly that the individual feels overwhelmed and unable to keep up or express them coherently.
- Example: Often accompanies pressured speech in mania.
- Flight of Ideas: Rapid, continuous flow of accelerated speech with abrupt changes from topic to topic, usually based on understandable associations, distracting stimuli, or plays on words. The connections are discernible, but the goal is not reached.
- Example: "I went to the store for milk. Milk is white. White clouds are in the sky. The sky is blue. Blue birds sing."
- Poverty of Thought: A reduction in the quantity of thoughts; the individual reports difficulty generating or sustaining thoughts.
- Example: A person with severe depression might feel their mind is empty, or struggle to elaborate on topics.
- Thought Blocking: A sudden interruption in the middle of a thought or sentence, leaving the individual unable to recall what they were saying. They may report that their thoughts have been "stolen" or "taken out of their head."
- Example: While talking, a person suddenly stops mid-sentence, appears blank, and then changes the topic or says they forgot what they were talking about. This is often associated with psychotic disorders.
- Form of Thought (Logic and Coherence): Refers to the logical connections between ideas and how thoughts are structured.
- Perseveration: Persistent, inappropriate repetition of the same words, ideas, or themes in response to different questions or topics.
- Example: If asked "How are you?" and then "What did you have for breakfast?", the person repeatedly answers with the first response, "I'm fine, I'm fine, I'm fine."
- Tangentiality: Digressing from the main topic, introducing irrelevant details, and never returning to the original point.
- Example: Asked "How was your day?", the person replies, "Well, the weather was nice, and the birds were singing, and I saw a squirrel, and my neighbor has a red car..." never answering about their day.
- Circumstantiality: Speech that is indirect and delayed in reaching its goal, due to the inclusion of excessive or irrelevant details. Unlike tangentiality, the person eventually returns to the original point.
- Loosening of Associations / Derailment: A disturbance in the logical progression of thoughts, where ideas shift from one subject to another in a way that is unrelated or only superficially connected.
- Example: "I like to eat at the restaurant. It has a nice window. Windows are made of glass. My friend broke a glass yesterday. He was very sad." The connections are increasingly difficult to follow.
- Abstract Thinking: The ability to understand concepts that are not concrete or directly observable, to generalize, and to interpret metaphors or proverbs. Impaired abstract thinking means thinking is excessively concrete.
- Example: When asked to interpret "People who live in glass houses shouldn't throw stones," a person with concrete thinking might say, "Because the glass would break," rather than understanding the metaphorical meaning about hypocrisy.
- Content of Thought (What one is thinking about): Refers to the themes, beliefs, and preoccupations that dominate an individual's thoughts.
- Delusions: Fixed, false beliefs that are firmly held despite clear evidence to the contrary, and are not consistent with the person's cultural or religious background.
- Types of Delusions:
- Grandiose Delusions: The belief that one is exceptionally important, famous, wealthy, powerful, or possesses special abilities or knowledge, often beyond what is realistic.
- Example: A patient believing they are a secret agent with a mission to save the world, or that they have invented a cure for all diseases.
- Delusions of Guilt or Worthlessness: Intense feelings of self-blame, remorse, or belief that one is deserving of punishment, has committed unforgivable sins, or is utterly worthless, even without any objective reason.
- Example: A patient believing they are responsible for all the suffering in the world or that they are a terrible person who doesn't deserve to live.
- Delusions of Jealousy (Morbid Jealousy or Othello Syndrome): The unfounded belief that one's spouse or partner is being unfaithful, despite a lack of evidence.
- Example: A person constantly accusing their partner of infidelity, checking their phone, or following them, without any basis for suspicion.
- Delusions of Persecution (Paranoid Delusions): The belief that one is being deliberately harmed, harassed, tormented, conspired against, spied upon, or otherwise ill-treated by others (individuals or agencies).
- Example: A patient believing the government is monitoring their thoughts, or that their neighbors are poisoning their food.
- Religious Delusions: Beliefs that are extreme or idiosyncratic interpretations of religious themes, outside the bounds of what is accepted by their religious community. These differ from culturally normative strong religious faith.
- Example: A patient believing they are a prophet chosen by God for a specific, often bizarre, mission, or that they are the reincarnation of a divine figure.
- Delusions of Control, Influence, or Passivity: The belief that one's thoughts, feelings, or actions are being controlled, imposed, or influenced by an external force or agency. This can manifest in several ways:
- Thought Insertion: The belief that alien thoughts are being placed into one's mind by an external source.
- Example: A patient stating, "These aren't my thoughts; the aliens are putting them in my head."
- Thought Withdrawal: The belief that thoughts are being removed or stolen from one's mind by an external force.
- Example: A patient explaining why they stopped mid-sentence: "My thoughts were just taken out of my head by the FBI."
- Thought Broadcasting: The belief that one's thoughts are being transmitted aloud or broadcasted to others, or that others can hear their thoughts.
- Example: A patient covering their head, saying, "Everyone can hear what I'm thinking, it's on the radio."
- Somatic Delusions: False beliefs about one's body, health, or bodily functions.
- Example: A patient believing their organs are rotting inside them, or that they are infested with parasites despite medical reassurance.
- Obsessions: Recurrent and persistent thoughts, urges, or images that are experienced as intrusive and unwanted, causing marked anxiety or distress. The individual attempts to ignore or suppress them, or to neutralize them with some other thought or action (compulsion).
- Example: Persistent intrusive thoughts about contamination, doubts about having locked the door, or aggressive impulses.
- Phobias: Persistent, irrational, and excessive fear of a specific object, situation, or activity, leading to avoidance or intense distress when exposed to the feared stimulus.
- Example: Arachnophobia (fear of spiders), Acrophobia (fear of heights), Social Phobia (fear of social situations).
- Suicidal or Homicidal Ideation: Thoughts about ending one's own life or harming others. These are serious symptoms requiring immediate assessment and intervention.
- Ideas of Reference: Belief that unrelated external events have a special, personal meaning for them (less fixed and bizarre than delusions of reference).
VI. Awareness and Cognitive Functions
These symptoms relate to an individual's fundamental mental capacities, which can be significantly impacted by mental disorders.
- Level of Consciousness: Refers to the state of alertness and wakefulness. Disturbances can range from mild alterations to complete unconsciousness.
- Clouding of Consciousness: A mild form of altered consciousness, characterized by reduced alertness, poor attention, and a lack of clear-mindedness in perception and comprehension. The person may appear dull or listless.
- Delirium: An acute state of mental confusion characterized by fluctuating awareness, disorientation, inattention, disorganized thinking, and often perceptual disturbances (e.g., hallucinations). The individual appears bewildered, restless, and confused.
- Stupor: A state of near-unconsciousness or profound unresponsiveness, characterized by a significant reduction in reaction to external stimuli. Despite appearing motionless, the person may still be aware of their surroundings. Can occur in severe depression, catatonia, or neurological conditions.
- Coma: A profound state of unconsciousness from which the person cannot be aroused, even with vigorous stimulation.
- Orientation: The ability to know one's current place in time, space, and person. A person is considered fully oriented if they can accurately identify:
- Time: Day, date, month, year, season.
- Place: Current location (hospital, home, city).
- Person: Who they are and who significant others around them are.
- Example: Disorientation is common in delirium, dementia, and states of acute confusion.
- Attention and Concentration:
- Attention: The ability to focus one's mental resources on a specific task or stimulus, selecting relevant information while ignoring distractions.
- Concentration: The ability to sustain that focus over a period.
- Assessment: Often assessed by tasks like serial sevens (subtracting 7 from 100 repeatedly), reciting months of the year backward, or spelling words backward.
- Impact: Impaired attention and concentration can significantly affect the ability to learn new information (poor registration) and immediate/short-term memory.
- Example: A person with ADHD struggles to maintain attention on schoolwork; someone in a manic state may have highly distractible attention.
- Memory: The ability to register, retain, and recall past and present events and general knowledge. Memory disturbances manifest as forgetfulness or an inability to remember important information.
- Immediate Memory: Ability to recall information just presented (e.g., repeating a short list of numbers).
- Short-Term Memory (Recent Memory): Ability to recall events from minutes to days ago.
- Example: Forgetting what one had for breakfast that morning, or misplacing keys frequently.
- Long-Term Memory (Remote Memory): Ability to recall events from months or years ago, or personal history.
- Example: Forgetting one's childhood home or significant life events.
- Amnesia: Partial or complete loss of memory.
- Confabulation: Fabricating imaginary experiences to fill in gaps in memory, often without conscious intent to deceive.
- Intellect: The overall ability to process, interpret, and use information, to learn from experience, and to adapt to new situations. It includes reasoning, problem-solving, and critical thinking.
- Assessment: While IQ tests are formal measures, clinical assessment involves observing the person's vocabulary, general knowledge, judgment, and ability to handle complex information.
- Example: Asking hypothetical questions like, "What would you do if you found a child playing with a sharp razor blade?" to assess judgment. Impaired intellect is characteristic of intellectual disability and neurocognitive disorders.
- Abstract Thought: The ability to understand concepts that are not concrete or directly observable, to generalize, and to interpret metaphors or proverbs. Impaired abstract thought (concrete thinking) means interpreting things literally.
- Example: If asked the meaning of "Don't cry over spilled milk," a person with concrete thinking might say, "Because it makes a mess," rather than the abstract meaning of not dwelling on past misfortunes.
VI. Sense of Self and Reality
These involve disruptions in the fundamental experience of one's own self and the reality of the external world.
- Depersonalization: A sense of detachment from one's own body, thoughts, feelings, or actions, as if observing oneself from outside or feeling unreal. The body may feel changed, distorted, or not truly one's own.
- Example: "I feel like I'm watching myself in a movie," or "My hand doesn't feel like it belongs to me." It is a change in the awareness of the self, often accompanied by emotional numbness.
- Derealization: A sense of detachment from one's surroundings, where the external world feels unreal, dreamlike, foggy, or distorted. Objects or people may appear strange, lifeless, or distant.
- Example: "The room looks flat, like a painting," or "People around me seem like robots." This can occur in anxiety, stress, fatigue, affective disorders, or hyperventilation.
VII. Insight and Judgment
- Insight: An individual's awareness and understanding of their own mental state, symptoms, and the nature of their illness, including the need for treatment.
- Degrees of Insight: Can range from complete denial of illness to full intellectual and emotional appreciation of the condition.
- Impact: Lack of insight is a significant barrier to treatment adherence, as individuals may not recognize the need for help.
- Example: A person with schizophrenia experiencing delusions may firmly believe they are not ill and refuse medication.
- Judgment: The ability to make sound decisions, understand the consequences of one's actions, and behave appropriately in social situations.
- Example: Poor judgment might be evident if a person in a manic episode makes impulsive financial decisions, or if someone with impaired reality testing walks into traffic without looking.
VII. Other Common Presenting Symptoms
- Relationship Problems: Mental disorders often impair an individual's ability to form and maintain healthy interpersonal relationships.
- Social Withdrawal: A pervasive avoidance of social interactions or activities, leading to isolation.
- Example: A person with depression or social anxiety might stop seeing friends and family, staying home all the time.
- Isolation: Keeping to oneself even when in a social environment; feeling disconnected from others.
- Poor Interpersonal Relations: Frequent conflicts, arguments, or difficulty empathizing and connecting with others.
- Example: Someone with Borderline Personality Disorder may experience intense, unstable relationships characterized by rapid shifts from idealization to devaluation.
- Appetite and Weight Disturbances: Significant changes in eating patterns and body weight are common symptoms across various mental disorders.
- Increased Appetite/Weight Gain:
- Example: Seen in atypical depression, or as a side effect of certain psychotropic medications.
- Decreased Appetite/Weight Loss:
- Example: A prominent symptom in major depressive disorder, anorexia nervosa, or anxiety.
- Specific Eating Disorder Symptoms: Such as refusing to eat, hiding food, excessive worry about weight and body image (as in anorexia nervosa or bulimia nervosa).
- Sleep Disturbances: Disrupted sleep patterns are nearly universal in mental disorders and can range from insomnia to hypersomnia.
- Altered Sleep-Wake Cycle: Disruption of the natural circadian rhythm, leading to being awake at night and drowsy during the day.
- Example: Common in bipolar disorder during manic or depressive episodes.
- Initial Insomnia: Difficulty falling asleep at the beginning of the night.
- Example: Often associated with anxiety disorders.
- Middle Insomnia: Waking up frequently during the night and having difficulty returning to sleep.
- Terminal Insomnia (Early Morning Awakening): Waking up much earlier than desired (e.g., 3 AM to dawn) and being unable to return to sleep.
- Example: A classic symptom of major depressive disorder.
- Hypersomnia: Excessive sleepiness, or prolonged sleep duration.
- Example: Can occur in atypical depression or some neurological conditions.
- Disturbed Sleep Quality: Sleeping for a sufficient duration but waking up feeling unrefreshed, often due to nightmares, night terrors, or fragmented sleep.
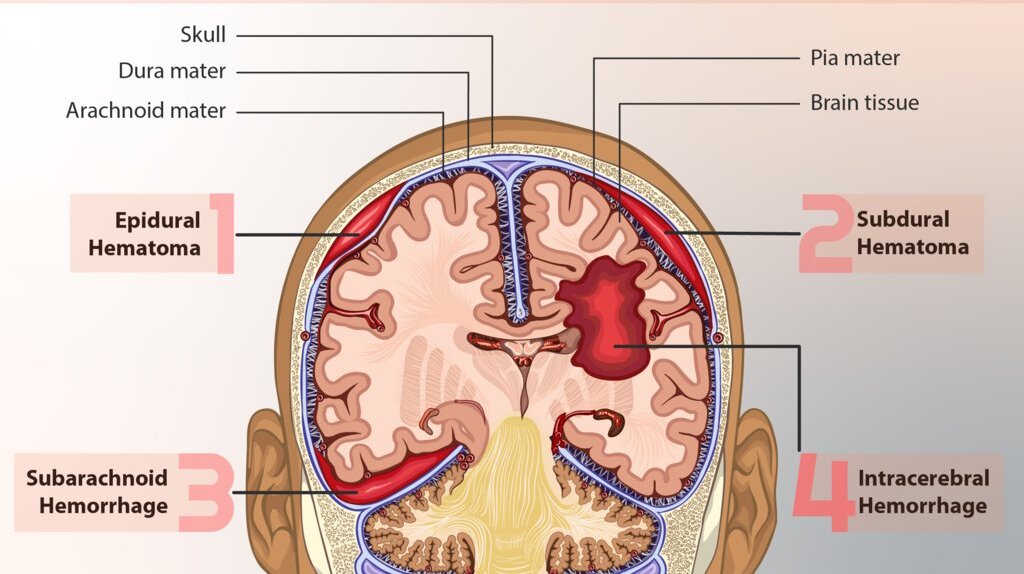
















 Image Placeholder - Stages in the development of blood cells diagram
Image Placeholder - Stages in the development of blood cells diagram