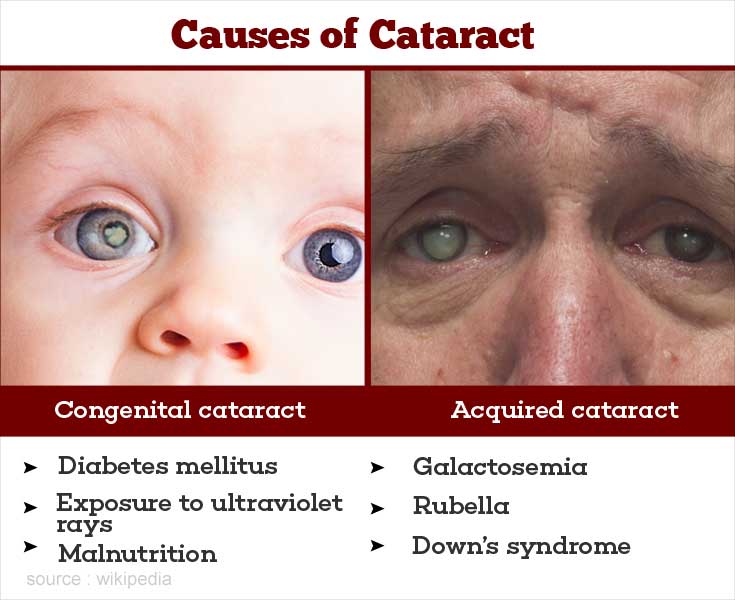
Assessing and Classification of a Sick Child and General Danger Signs.
Assessing and Classification of a Sick Child
Determine whether this is an initial visit or a return (follow up) visit.
If it is an initial visit, Ask, “what is the child’s problem”?
After knowing the problem;
- Check for general danger signs.
A general danger sign is an indicator that a child has a severe problem and therefore needs urgent referral.
General danger signs include:
- The child is unable to drink/feed.
- Vomiting everything.
- The child has convulsions.
- The child is lethargic or unconscious.
- Child is convulsing now.
Then ask about other main symptoms
GENERAL DANGER SIGNS
General danger signs in children are indicators of serious illness that require immediate action to save the child’s life.
These signs are seen in children aged two months up to five years and should not be ignored. The 4 general Danger signs according to World Health Organisation are;
- Unable to breastfeed or drink: If a child is unable to suck or swallow when offered a drink or breastmilk, it is considered a danger sign.
- Vomiting everything: If a child is not able to hold down any food, fluids, or oral medications and vomits everything, it is a cause for concern.
- Convulsions: Convulsions are characterized by the child’s arms and legs stiffening due to muscle contractions or repeated abnormal movements. The child may also lose consciousness or be unresponsive to spoken directions.
- Lethargic or unconscious: A lethargic child is drowsy, not alert, and shows no interest in their surroundings. They may not look at their mother or respond to stimuli. An unconscious child cannot be awakened and does not respond to touch, shaking, or spoken words.
Child with any General Danger sign needs URGENT attention: complete assessment, give any pre-referral treatment immediately and refer.
-
Not able to drink or breastfeed:
“Not able to drink or breast-feed” means that the child is not able to suck or swallow when offered a drink or breast milk.
NB: if not sure of the mother’s answer, offer the child clean water/breastmilk
-
Vomiting everything:
“Vomiting everything” means;
- Child is not able to hold anything down at all.
What goes down comes back up.
- NB: if not sure of the mother’s answer, offer the child clean water/breastmilk. Observe if the child vomits
-
Convulsions:
A convulsion is any involuntary movement in any part of the body.
- A child can have this danger sign if there is a history of convulsions or convulsing during the visit.
- A history of convulsions only counts as a danger sign if the convulsions happened during the present illness.
- Use words for convulsions that caregivers understand. eg fits or spasms.
-
Lethargic or unconscious:
“Lethargic or unconscious” means that:
- The child is not awake and alert when he should be.
- He is drowsy and does not show interest in what is happening around him.
- The child may stare blankly and appears not to notice what is going on around him.; or
- Unconscious child cannot be awakened. He does not respond when touched, shaken, or spoken to
CHECK FOR GENERAL DANGER SIGNS
|
ASK |
LOOK |
|
–Is the child able to drink or breastfeed? |
-See if the child is lethargic or unconscious. |
|
-Does the child vomit everything? |
-Is the child convulsing now? If yes, treat immediately. |
|
-Has the child had convulsions in this illness? |
A child with any general danger sign needs URGENT attention; complete the assessment and any pre-referral treatment immediately so referral is not delayed.
SIGNS | CLASSIFY AS | TREATMENT |
Any general danger sign | VERY SEVERE DISEASE | • Give diazepam if convulsing now. • Quickly complete the assessment. • Give any pre-referral treatment immediately. • Treat to prevent low blood sugar. • Keep the child warm. • Refer URGENTLY. • Screen for possible TB disease and check for HIV. |
THEN ASK ABOUT MAIN SYMPTOMS:
Four main symptoms
The health worker attending to a sick child MUST ask about all the four main symptoms below.
- Cough or difficult breathing
- Diarrhoea
- Fever
- Ear problem
When a symptom is present, assess further on that symptom.
Cough or Difficult breathing
- Pneumonia is among the leading causes of death in children under five years.
- Most children with cough or difficulty breathing have only a cough or a cold.
- A few children with cough or difficulty breathing may also have pneumonia.
ASSESSING FOR COUGH OR DIFFICULT BREATHING – THE CHILD MUST BE CALM
Does the child have cough or difficult breathing?
ASK | LOOK, LISTEN, FEEL | |
IF YES, ASK For how long? | Count the breaths in one minute. Use respiratory rate timers where available • Look for chest in-drawing* • Look and listen for stridor** • Look and listen for wheeze*** • Check for central cyanosis • Check for oxygen saturation using pulse oximetry where available. • Check AVPU**** • If wheezing with either chest indrawing or fast breathing: Assess for possible Asthma. • If wheezing assesses for possible TB disease. | |
-Is the child convulsing now? If yes, treat immediately. | ||
If the child is: 2 months up to 12 months 12 months up to 5 years | Fast breathing is: 50 breaths per minute or more 40 breaths per minute or more | |
SIGNS | CLASSIFY AS | TREATMENT |
Any general danger sign OR • Oxygen saturation less than 90% • Chest indrawing or Stridor in calm child. • Central Cyanosis • AVPU = V, P or U | VERY SEVERE DISEASE | • Give diazepam if convulsing now. • Quickly complete the assessment. • Give any pre-referral treatment immediately. • Treat to prevent low blood sugar. • Keep the child warm. • Give fist oral dose of oral cotrimoxazole • Give Vit A • Refer URGENTLY. • Screen for possible TB disease and check for HIV. |
Note: *ANYTHING IN BOLD IS URGENT PRE-
REFERRAL TREATMENT
*Chest in-drawing is present if the lower
chest wall moves in during inspiration.
**Stridor – a harsh sound heard during
inspiration.
***Wheeze – a musical sound heard
during expiration.
****AVPU – Alert, responsive to Voice,
responsive to Pain, Unresponsive
Chest indrawing in calm child OR • Fast breathing AND • No signs of severe pneumonia | PNEUMONIA | • Give Amoxicillin Dispersible Tablet. • Give Vitamin A. • Treat wheeze if present. • If wheezing, follow-up in 2 days. • Soothe the throat and relieve the cough with a safe remedy. • Screen for possible TB disease and check for HIV. • If wheezing (or disappeared after rapidly acting bronchodilator) give an inhaled bronchodilator for 5 days • If coughing for more than 14 days or recurrent wheeze, refer for possible TB or asthma assessment. • Review in 2 days, if not possible, admit OR refer children with chest indrawing. • Advise mother when to return immediately. |
No signs of pneumonia or very severe disease. | NO PNEUMONIA: COUGH OR COLD | • Treat wheeze if present. • If wheezing, follow-up in 2 days. • Soothe the throat and relieve the cough with a safe remedy. • Follow-up in 5 days if not improving. • Screen for possible TB disease and check for HIV. • Advise mother when to return immediately. |
Diarrhoea
- Diarrhoea is the passage of three or more watery stools in 24 hours. It is common in children, especially those between 6 months and 2 years of age.
- It is the 2nd leading cause of mortality in under-fives.
- Most of these deaths are usually due to dehydration.
- If an episode of diarrhoea lasts less than 14 days, it is an acute diarrhoea and if 14 days or more, it is persistent diarrhoea.
- Diarrhoea with blood in the stool, with or without mucus, is called dysentery.
- Frequent passage of normal stool is not diarrhoea.
Does the child have diarrhoea?
ASK | LOOK, LISTEN, FEEL | |
IF YES, ASK For how long? • Is there blood in the stool? | – Look at the child’s general conditions. Check:
Is the child:
– Look for sunken eyes – Offer the child fluid, is the child:
– Pinch the skin of the abdomen. Does it go back:
| |
Remember to classify all children with diarrhoea for dehydration. | ||
for DEHYDRATION
SIGNS | CLASSIFY AS | TREATMENT |
All four of: • Weak/absent pulse • AVPU* < A • Cold hands + Temp gradient • Capillary refill > 3 sec PLUS • sunken eyes and very slow/slow skin pinch. | HYPOVOLAEMIC SHOCK FROM DIARRHOEA/ DEHYDRATION | • TREAT FOR SHOCK. Give Ringer’s Lactate 20mls/kg. – A second bolus may be given if required before proceeding to step 2 of PLAN C. • Treat for and to prevent low blood sugar. • Assess for severe acute malnutrition. • Assess for severe anaemia. • NB: If HB<5g/dl transfuse urgently • Admit or refer urgently to hospital • Screen for possible TB disease and check for HIV • If a child is 2yrs or older & there’s cholera in your area, give oral cotrimoxazole for cholera. |
Two or more of the following signs: • Restless, irritable • Sunken eyes • Drinks eagerly, thirstily • Skin pinch goes back slowly | SOME DEHYDRATION | If child also has a severe classification: – Admit or refer URGENTLY to hospital with mother giving frequent sips of ORS on the way. Advise the mother to continue breastfeeding. OR If the child has no severe classification: – Give fluid and food for some dehydration (Plan B). – Give vitamin A. – Give ORS and Zinc Sulphate. • Follow-up in 2 days if not improving. • Screen for possible TB disease and check for HIV • Advise mother when to return immediately. |
Not enough signs to classify as some or severe dehydration. | NO DEHYDRATION | If child also has a severe classification: – Admit or refer URGENTLY to hospital with mother giving frequent sips of ORS on the way. Advise the mother to continue breastfeeding. OR • If the child has no severe classification: – Give fluid and food to treat diarrhoea at home (Plan A). – Give vitamin A. – Give ORS and Zinc Sulphate. • Follow-up in 5 days if not improving. • Screen for possible TB disease and check for HIV. • Advise mother when to return immediately. |
and if diarrhoea 14 days or more
Dehydration present: (hypovolaemic shock, severe dehydration, some dehydration). | SEVERE PERSISTENT DIARRHOEA | • Treat hypovolaemic shock and any other form of dehydration before referral unless the child has another severe classification. • Give Vitamin A. • Give ORS and Zinc Sulphate. • Give Multivitamin / Mineral supplements. • Admit or refer URGENTLY to hospital with mother giving frequent sips of ORS on the way. • Screen for possible TB disease and check for HIV. |
No dehydration | PERSISTENT DIARRHOEA | • Advise the mother on feeding a child who has PERSISTENT DIARRHOEA. • Give vitamin A. • Give ORS and Zinc Sulphate. • Give Multivitamin / Mineral supplements. • Check for HIV infection. • Follow-up in 5 days. • Screen for possible TB disease and check for HIV |
and if blood in stool
Blood in the stool. | DYSENTERY | • Treat with Ciproflaxacin. • Give Vitamin A. • Give ORS and Zinc Sulphate. • Follow-up in 2 days. • Screen for possible TB disease and check for HIV. |
Fever
Fever is present if :-
- There is history from parent/caregiver or
- Child feels hot or
- Child has temperature 37.5° c and above
A child with fever may have malaria, measles or another severe disease. Malaria currently causes 11% of under five deaths. Fever may also be due to a simple cough, cold or other viral infection.
To classify and treat fever, you must know malaria risk in your area.
- High Malaria risk; > 5% of fever cases in children are due to malaria.
- Low Malaria risk; 5% or less of fever cases in children are due to malaria.
Does the child have fever? (by history or feels hot or temperature 37.5°C* or above)
IF YES:
- Has the child travelled to a high risk (Malaria endemic, seasonal transmission or epidemic prone) area in the last 1 month?
Decide Malaria Risk: high or low risk.
ASK | LOOK & FEEL | |
THEN ASK: • For how long? • If more than 7 days, has fever been present every day? • Has the child had signs of measles within the last 3 months? | LOOK AND FEEL: • Look or feel for stiff neck. • Look for runny nose. Look for signs of MEASLES: • Generalized rash and one of these: cough, runny nose, or red eyes. • Look for any other cause of fever••• | |
High Malaria risk: Do a malaria test ○ Endemic Zone ○ Seasonal Transmission Zone ○ Epidemic prone areas Low malaria risk: ○ Do a malaria test if there is no obvious cause of fever. | TEST POSITIVE • P.falciparum PRESENT • P.vivax PRESENT
TEST NEGATIVE • P.falciparum or P.vivax absent NOTE: If you can’t test, don’t withhold treatment. | |
SIGNS | CLASSIFY AS | TREATMENT |
• Any general danger sign or • Stiff neck. AND • Confirm malaria with a test. |
VERY SEVERE FEBRILE DISEASE OR SEVERE MALARIA | • Give first dose of artesunate or quinine for severe malaria. • Give first dose of an appropriate antibiotic (Give first dose of Ceftriaxone) • Treat the child to prevent low blood sugar. • Give one dose of paracetamol in the clinic for high fever (≥38.5 °C). • Admit or Refer URGENTLY to hospital. • Screen for possible TB disease and check for HIV. |
• Malaria test POSITIVE** | UNCOMPLICATED MALARIA | • Give recommended first line oral antimalarial • Give Artemether + Lumefantrine (AL). • Give one dose of paracetamol in clinic for high fever (≥38.5 °C). • Give appropriate antibiotic treatment for an identified bacterial cause of fever. • Give Vitamin A. • Follow up in 3 days if fever persists. • If fever is present every day > 7 days assess further or refer • Screen for possible TB disease and check for HIV • Advise when to return immediately. |
• Malaria test NEGATIVE | FEVER: NO MALARIA | • Give one dose of paracetamol in clinic for high fever (≥38.5 °C) • Assess for other possible causes of fever • Follow up in 3 days if fever persists. • If fever is present every day for more than 7 days assess further or refer. • Screen for possible TB disease and check for HIV • Advise mother when to return immediately. |
CHECK FOR COMPLICATIONS OF MEASLES
Check for Complications of MEASLES. If the child has signs of measles now or within the last 3 months | • Look for mouth ulcers, are they deep or extensive? • Look for pus draining from the eye • Look for clouding of the cornea | |
SIGNS | CLASSIFY AS | TREATMENT |
Generalized rash of measles and • One of: cough, runny nose or red eyes | SUSPECTED MEASLES | • Give Vitamin A (See page 14) • Notify, take blood sample for confirmation. • Screen for possible TB disease and check for HIV. • Advise mother when to return immediately. |
Any general danger sign or • Clouding of cornea or • Deep or extensive mouth ulcers. | SEVERE COMPLICATIONS OF MEASLES | • Give Vitamin A. • Give first dose of Ceftriaxone Antibiotic. • If clouding of the cornea or pus draining from the eye, apply tetracycline eye ointment. • Notify, take blood sample for confirmation OR refer • Admit or refer URGENTLY to hospital • Screen for possible TB disease and check for HIV. |
• Pus draining from the eye or • Mouth ulcers. | EYE OR MOUTH COMPLICATIONS OF MEASLES | • Give Vitamin A • If pus draining from the eye, treat eye infection with tetracycline eye ointment. • If mouth ulcers, treat with nystatin • Follow-up in 2 days. • If child has no indication for referral, notify and draw blood sample for confirmation of measles • Screen for possible TB disease and check for HIV |
• No pus draining from the eye or mouth ulcers. | NO EYE OR MOUTH COMPLICATIONS OF MEASLES | • Give Vitamin A if not received in the last 1 month. • If child has no indication for referral, draw blood and send for confirmation. • Screen for possible TB disease immediately after the measles infection and check for HIV. |
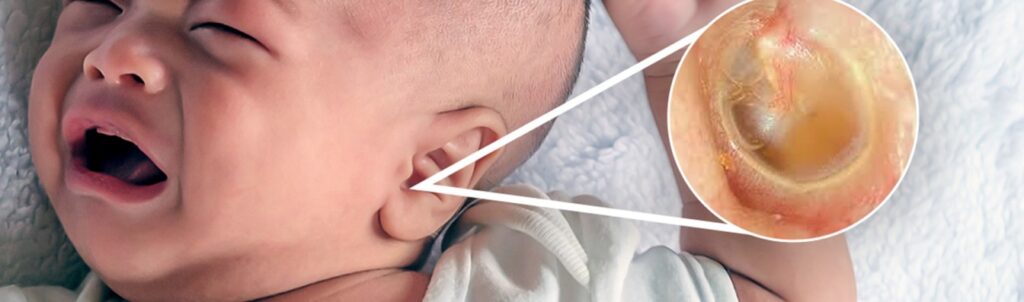
EAR PROBLEM
- A child with an ear problem may have an ear infection.
- Ear infection may cause pus to collect behind the eardrum causing pain and often fever.
- If not treated, the eardrum may burst, discharge pus and the child feels less pain.
- However, the child may suffer poor hearing or worse deafness.
- Ear infection may lead to Mastoiditis or Meningitis.
Does the child have an ear problem?
ASK | LOOK AND FEEL | |
IF YES, ASK • Is there ear pain? • Is there ear discharge? If yes, for how long? |
Pinch the skin of the abdomen. Does it go back:
| |
Classify EAR PROBLEM
SIGNS | CLASSIFY AS | TREATMENT |
• Tender swelling behind the ear. | MASTOIDITIS | • Give first dose of Ceftriaxone Antibiotic. • Give first dose of paracetamol for pain • Refer URGENTLY to hospital or admit • Check for HIV. |
• Pus is seen draining from the ear or • Discharge is reported for less than 14 days,or • Ear pain. | ACUTE EAR INFECTION | • Give Amoxicillin dispersible tablets for 5 days. • Give paracetamol for pain • Dry the ear by wicking • Check for HIV infection • Follow-up in 5 days |
• Pus is seen draining from the ear or discharge is reported for 14 days or more. | CHRONIC EAR INFECTION | • Dry the ear by wicking • Check for HIV infection • Follow-up in 5 days |
• No ear pain and No pus seen or reported draining from the ear. | NO EAR INFECTION |
|
THEN CHECK FOR ACUTE MALNUTRITION
Good nutrition results from the adequate intake of macronutrients, micronutrients and water to supply the metabolic (anabolic and catabolic) processes in the body.
There are two components of nutrition;
- Macronutrients
- Micronutrients
What is Malnutrition?
- Malnutrition is defined as a state when the body does not have enough of the required nutrients (under-nutrition) or has excess of required nutrients (overnutrition).
- Inadequacies of macro or micro nutrients may result in failure to thrive, poor growth or wasting.
- These processes are often measured by their anthropometric consequences (weight for age, height for age or weight for height).
- Undernutrition is the most common form of malnutrition in developing countries.
ASSESS FOR MALNUTRITION
ASK | LOOK AND FEEL | |
Is there a history of TB contact? | • Look for oedema of both feet. • Determine the child’s weight for Height/length (WFHL*) and plot on the IMNCI Chart booklet to determine the z-score. • Determine the growth pattern; Is the growth faltering? (Weight curve flattening or dropping for at least 2 consecutive months?) • Measure MUAC**mm in a child 6 months or older • IF WFH/L less than -3 z-score or MUAC less than 115mm then: | |
Check for any medical complication present: • Any general danger signs. • Any severe classification. • Pneumonia with chest in-drawing. If no medical complication present: • Conduct an appetite test. • Child is 6 months or older, offer RUTF to eat Is the child > Not able to finish RUTF portion > Able to finish RUTF portion • Child is less than 6 months, assess breastfeeding > Does the child have breastfeeding problems? If child has acute Malnutrition and is receiving RUTF, DO NOT give iron because there is already adequate amount of IRON in RUTF | ||
Classify Malnutrition
SIGNS | CLASSIFY AS | TREATMENT |
• Oedema of both feet, OR • WFH/L less than -3 z scores OR • MUAC Less than 11.5 cm and ANY one of the following: – medical complication present or – not able to finish RUTF*** or – Breast feeding problem (<6 months) | SEVERE ACUTE MALNUTRITION WITH COMPLICATIONS FOR CHILDREN | • Treat the child to prevent low blood sugar. • Keep the child warm • Give first dose of Benzylpenicillin + Gentamicin • Give Vitamin A • Refer URGENTLY to hospital • Admit or refer urgently to hospital if child has any other complications (Danger signs: Diarrhoea, Pneumonia, Fever, No appetite, etc) • Immunize as per schedule. • Screen for possible TB disease and check for HIV. |
For all children: • With visible severe wasting • Oedema of both feet, • <-3 Z Score ( weight for age or weight for height/Length) For children 6 months up to 59 months: MUAC <11.5 cm | SEVERE ACUTE MALNUTRITION | As above |
• WFH/L less than -3 z scores OR • MUAC Less than 11.5 cm AND • Able to finish RUTF | SEVERE MALNUTRITION WITHOUT COMPLICATIONS | • Give oral Amoxicillin DT for 5 days • Give ready to use therapeutic food for child aged six months and above • Screen for possible TB disease and check for HIV • Follow up in 7 days. • Assess the child’s feeding and counsel the mother on the feeding recommendations. • Immunize as per schedule. • Advise mother when to return immediately |
For all age groups: • Static weight or losing weight • 3 to <-2 Z- Score If age 6 months up to 59 months MUAC 11.5 to 12.5 cm | MODERATE ACUTE MALNUTRITION | • If growth is faltering for 2 consecutive months, give Vitamin A, assess further or refer to a hospital. • Assess the child’s feeding and counsel the mother on the feeding recommendations. • Give Albendazole if the child is 1 year or older and has not had a dose in the previous 6 months. • If feeding problems, follow up in 14 days. • Screen for possible TB disease and check for HIV • Immunize as per schedule. • Advise mother when to return immediately |
For all age groups: • Static weight or losing weight • -2 to <-1 Z- Score If age 6 months up to 59 months MUAC 12.5 to 13.5cms | AT RISK OF ACUTE MALNUTRITION | • If child is less than 2 years old, and has growth faltering, assess the child’s feeding and counsel the mother on feeding according to the feeding recommendations • Give Albendazole if the child is 1 year or older and has not had a dose in the previous 6 months. • Follow up in 14 days • If feeding problem, follow-up in 5 days • Screen for possible TB disease and check for HIV • Immunize as per schedule • Advise mother when to return immediately |
WFH/L > -1 to +2 Z scores OR MUAC > 13.5 cm | NO ACUTE MALNUTRITION | • If the child is less than 2 years old, assess the child’s feeding and counsel the mother on feeding according to the feeding recommendations. • If feeding problem, follow-up in 5 days • Give Albendazole if child is 12 months and has not had a dose in the previous 6 months • Immunize as per schedule • Screen for possible TB disease and check for HIV |
CHECK FOR ANAEMIA
Anaemia
The condition of having a lower-than-normal number of red blood cells or quantity of hemoglobin. Anemia diminishes the capacity of the blood to carry oxygen.
Common Causes
- Iron deficiency; may result from a diet deficient in iron.
- Intestinal parasites.
- Repeated nosebleeds.
- Haemolysis, due to: Malaria and other Inherited blood disorders like sickle cell disease)
- Chronic illness, such as tuberculosis and AIDS.
- Severe malnutrition (due to lack of protein to produce haemoglobin)
ASSESS FOR ANAEMIA
LOOK AND FEEL | |
Look for palmar pallor. Is it: • Severe palmar pallor? • Some palmar pallor? • No palmar pallor? • Do haemoglobin level (HB) test. |
SIGNS | CLASSIFY AS | TREATMENT |
• Severe palmar pallor • If HB<5g/dL | SEVERE ANAEMIA | • Treat to prevent low blood sugar • Keep the child warm • Admit or refer URGENTLY to hospital • Screen for possible TB disease and check for HIV |
• Some palmar pallor | ANAEMIA | • Assess the child’s feeding and counsel the mother on feeding. • If growth is faltering for 2 consecutive months, assess further or refer to hospital • Give Iron and Folate. • Give vitamin A • Give Albendazole if child is 1 year or older and has not had a dose in the last 6 months • Screen for TB disease and check for HIV • Follow up in 14 days • Immunize as per schedule • Advise mother when to return immediately. |
• No Palmar pallor | NO ANAEMIA | • If child is less than 2 years old, assess the child’s feeding and counsel the mother according to the feeding recommendations • If feeding problems, follow up in 5 days • Give Albendazole if child is 1 year or older and has not had a dose in the last 6 months • Immunize as per schedule • Screen for possible TB disease and check for HIV |
CHECK FOR HIV EXPOSURE AND INFECTION
All children found to have pneumonia, persistent diarrhea, ear discharge or very low weight for age (any of these features) and have no urgent need or indication for referral, should be assessed for symptomatic HIV infection.
- Children may acquire HIV infection from an infected mother through vertical transmission in utero, during delivery or while breastfeeding.
- Without any intervention, 30 – 40% babies born to infected mothers will themselves be infected.
- Most children born with HIV die before they reach their fifth birthday, with most not surviving beyond two years.
- Good treatment can make a big difference to children with HIV and their families.
- The child’s status may also be the first indicator that their parents are infected too.
ASSESS FOR HIV EXPOSURE AND INFECTION
ASK | LOOK, FEEL AND DIAGNOSE | |
• Ask for mother’s HIV status to establish child’s HIV exposure* Is it: Positive, Negative or Unknown (to establish child’s HIV exposure) • Ask if child has had any TB Contact | Child <18 months • If mother is HIV positive**, conduct DNA PCR for the baby at 6 weeks or at first contact with the child • If mother’s HIV status is unknown, conduct an antibody test (rapid test) on mother to determine HIV exposure. PRESUMPTIVE SYMPTOMATIC DIAGNOSIS OF HIV INFECTION IN CHILDREN <18 MONTHS • Pneumonia *** • Oral Candidiasis /thrush • Severe sepsis • Other AIDS defining conditions** Child ≥18 months • If the mother’s antibody test is POSITIVE, the child is exposed. Conduct an antibody test on the child. Child whose mother is NOT available: • Child < 18 months Do an antibody test on the child. If positive, do a DNA PCR test. • Child ≥ 18 months Do an antibody test to determine the HIV status of the child | |
CLASSIFY HIV STATUS
SIGNS | CLASSIFY AS | TREATMENT |
• Child < 18 months and DNA PCR test POSITIVE • Child ≥ 18 months and Antibody test POSITIVE | CONFIRMED HIV INFECTION | • Initiate ART, counsel and follow up existing infections • Initiate or continue cotrimoxazole prophylaxis • Assess child’s feeding and provide appropriate counseling to the mother/caregiver • Offer routine follow up for growth, nutrition and development and HIV services • Educate caregivers on adherence and its importance • Screen for possible TB disease at every visit. • For those who do not have TB disease, start Isoniazid prophylactic therapy (IPT). Screen for possible TB throughout IPT • Immunize for measles at 6 months and 9 months and boost at 18 months • Follow up monthly as per the national ART guidelines and offer comprehensive management of HIV. Refer to appropriate national ART guidelines for comprehensive HIV care of the child. |
Child<18 months • If mother test is positive and child’s DNA PCR is negative OR • If mother is unavailable; child’s antibody test is positive and DNA PCR is negative | HIV EXPOSED | • Treat, counsel and follow up existing infections • Initiate or continue Cotrimoxazole prophylaxis • Give Zidovudine and Nevirapine prophylaxis as per the national PMTCT guidelines • Assess child’s feeding and provide appropriate counseling to the mother/caregiver • Offer routine follow up for growth, nutrition and development • Repeat DNA PCR test at 6 months. If negative, repeat DNA PCR test again at 12 months. If negative, repeat antibody test at 18 months • Continue with routine care for under 5 clinics • Screen for possible TB at every visit • Immunize for measles at 6 months and 9 months and boost at 18 months • Follow up monthly as per the national ART guidelines and offer comprehensive management of HIV. Refer to appropriate national ART guidelines for comprehensive care of the child. |
• No test results for child or mother • 2 or more of the following conditions: • Severe pneumonia • Oral candidiasis/thrush • Severe Sepsis OR • An AIDS defining condition | SUSPECTED SYMPTOMATIC HIV INFECTION | • Treat, counsel and follow-up existing infections • Give cotrimoxazole prophylaxis • Give vitamin A supplements from 6 months of age every 6 months • Assess the child’s feeding and provide appropriate counseling to the mother • Test to confirm HIV infection • Refer for further assessment including HIV care/ ART • Follow-up in 14 days, then monthly for 3 months and then every 3 months or as per immunization schedule |
Mother’s HIV status is NEGATIVE OR Mother’s HIV status is POSITIVE and child is ≥ 18 months with antibody test NEGATIVE 6 weeks after completion of breastfeeding | HIV NEGATIVE | • Manage presenting conditions according to IMNCI and other recommended national guidelines • Advise the mother about feeding and about her own health |
CHECK THE CHILD’S IMMUNIZATION, VITAMIN A & DEWORMING STATUS
- Immunization is one of the most cost effective health interventions for disease control.
- It targets children under the age of 5 years.
- It needs tremendous input and effort to make it happen.
- Immunization coverage can be enhanced through;
- Routine Immunization
- Supplemental Immunizations
- Surveillance of the target diseases
- Mopping up in high risk areas
A child’s body requires Vitamin A for; Growth and development, Protection against infections & reinforces the body’s immunity. Vit A deficiency may result from; Inadequate intake of vitamin A rich foods, Poor absorption of the vitamin A & rapid utilization of vitamin A stores due to illnesses
Vit A deficiency may lead to;
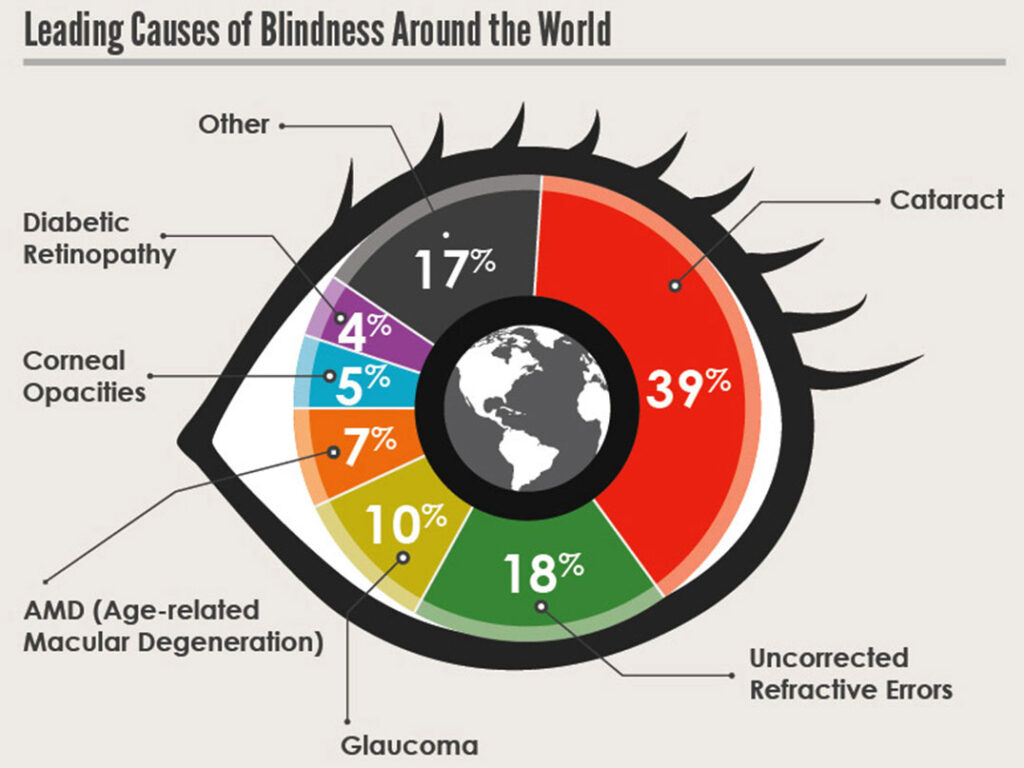
- Increased incidence of illness
- Delays recovery from infections
- Leads to eye damage and may even lead to blindness
- Increases the risk of death in sick children
Remember, Vit A supplementation can;
- Reduces measles mortality by 50%
- Reduces diarrhea mortality by 33%
- Reduces all causes of mortality by 23%
CHECK CHILD’S IMMUNIZATION
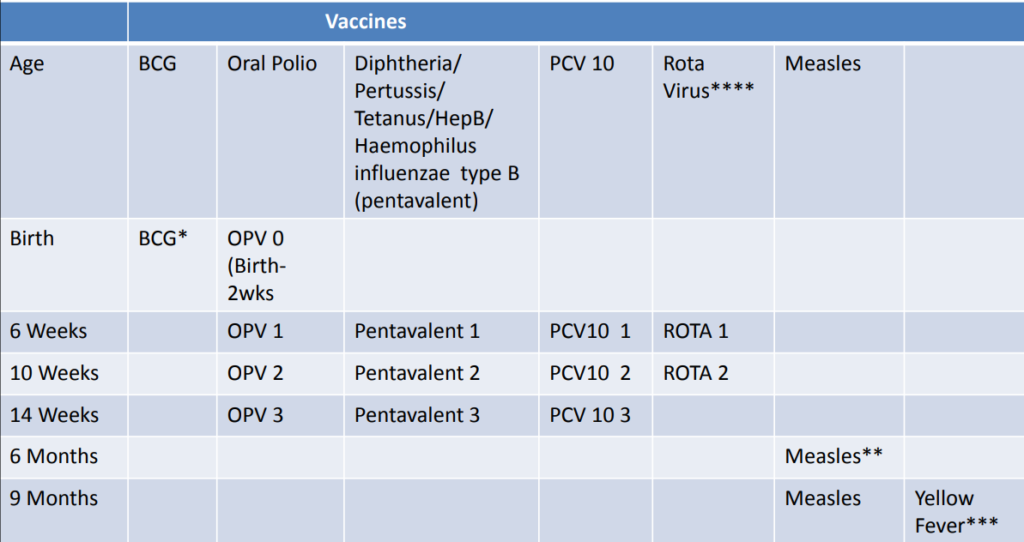
- *If BCG is not given at birth, it should not be given to children with symptomatic HIV /AIDS.
- **Measles vaccine at 6 months is for HIV exposed/ infected children
- ***Yellow fever vaccine should not be given to children with symptomatic HIV/AIDS.
- ****RotaVirus vaccine should not be given to children over 15 months
- Pentavalent not given if child had convulsion following previous dose or a child with recurrent convulsions or another active neurological disease
- PCV10 & Pentavalent not given to Infants with a moderate or severe illness (temperature ≥39°C) until their condition improves.
- PCV10 and Pentavalent contraindicated if severe allergic reactions or shock to a prior dose or any component of the vaccine.
- Do not delay referrals of children with severe classifications to administer immunizations.

ASSESS OTHER PROBLEMS THE CHILD MIGHT HAVE
It is important to remember that the already discussed IMCI case management process ;
- Does not cover all symptoms
- Is not reviewing all pediatric medicine
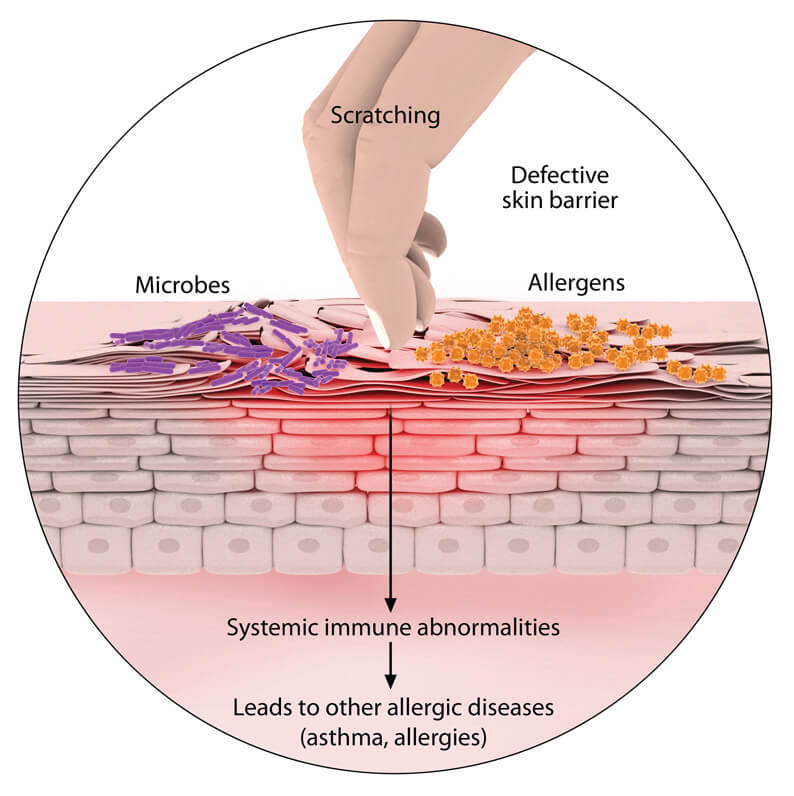
- Remember to address some complaints the caregiver may have raised, eg she may have said the child has an itchy skin, You may have observed another problem the mother didn’t say eg jiggers.
- Treat any other problems according to your training, experience and clinic policy.
- Refer the child for any other problem you cannot manage in the clinic.
Assessing and Classification of a Sick Child and General Danger Signs. Read More »