POST ABORTION CARE
POST ABORTION CARE
Post abortion care (PAC) is an approach aimed at reducing injuries and death resulting from incomplete and unsafe abortions and their related complications. It is a critical component of comprehensive abortion care and includes five essential elements.
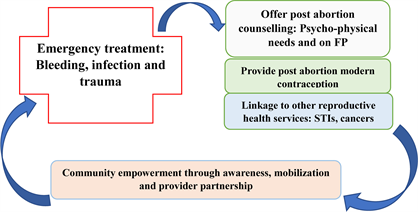
Components of PAC:
1. Emergency Treatment of Incomplete Abortion and Life-Threatening Complications:
- Immediate medical attention for incomplete abortions and associated complications.
- Evacuation of the uterus to prevent further health risks.
2. Post-Abortion Counseling:
- Psychological support to help mothers overcome trauma.
- Education on recognizing and responding to complications, such as fever, severe hemorrhage, and acute lower abdominal pain (LAP).
- Guidance on when to seek medical help if complications arise.
3. Initiation of Post-Abortal Family Planning Counseling and Services:
- Education on the rapid return to fertility post-abortion, typically around 10 days.
- Information on available family planning methods.
- Support and provision of chosen contraceptive methods to prevent future unwanted pregnancies.
4. Integration with the Reproductive Health Care System:
- Continuation of post-abortion emergency services within the broader reproductive health care framework.
- Access to comprehensive services including STI/HIV screening and cervical cancer prevention.
5. Community Participation in Complication Prevention:
- Education of community members on the dangers of abortion complications, such as:
- Bleeding
- Foul-smelling discharge
- Abdominal pain
- Fever
- Promotion of measures to prevent complications:
- Personal hygiene practices
- Treatment of STIs
- Use of post-abortion family planning methods
IMPORTANCE OF POST ABORTION CARE
- Life-saving services: PAC reduces the risk of maternal mortality and morbidity associated with unsafe abortions.
- Reduces fertility problems: Helps mitigate long-term reproductive health issues caused by unsafe abortions.
- Prevents unwanted pregnancies: Through effective family planning and contraceptive services.
- Accessible quality health services: Ensures women have access to necessary health services.
- Improves overall health: Enhances the physical, social, spiritual, and psychological well-being of women.
- Better referral management: Streamlines the process for accessing advanced medical care.
- Encourages proactive health-seeking behavior: Empowers women to seek timely medical assistance.
COMPREHENSIVE ABORTION CARE
Comprehensive abortion care involves the primary, secondary, and tertiary prevention of unsafe abortions and connects abortion care to other reproductive health services.
It aims to minimize and prevent the negative outcomes of an abortion.
Components of Comprehensive Abortion Care:
Prevention of unintended pregnancies:
- Sexuality education
- Safe sex practices
- Contraception and family planning
- Emergency contraception
- Community involvement
Provision of abortion services to the full extent of the law:
- Legal and safe abortion procedures
- Medical and surgical options
Post-abortion care, which includes five elements:
- Emergency treatment of abortion complications including evacuation of the uterus for incomplete abortion.
- Provision of post abortion and family planning counselling.
- Provision of family planning methods.
- Linkage between abortion care services and other RH services such as STI/HIV prevention and screening for cancer of the cervix
- Community involvement.
MANAGEMENT OF ABORTION
(Find detailed management for each in Gynaecology by clicking here)
Triage for abortion patients:
- Monitor vital signs (e.g., BP, pulse)
- Assess for shock, excessive pain, level of consciousness, general condition, vaginal bleeding, and fever
- Resuscitate if necessary before taking history
THREATENED ABORTION:
- Admit to the maternity ward for monitoring
- Administer medications such as ferrous sulphate, nifedipine, and Nospa
- Advise on bed rest and avoid strenuous activities
- Follow up in antenatal clinic if bleeding stops; reassess if it persists
INEVITABLE ABORTION:
- Hospitalization and medical management
- Perform MVA for pregnancy <16 weeks; administer oxytocin or misoprostol for pregnancies >16 weeks
- Schedule follow-up and offer PAC
INCOMPLETE ABORTION:
- Hospitalization and surgical management
- Use forceps for minimal bleeding or MVA for profuse bleeding
- Administer oxytocin or misoprostol for pregnancies >16 weeks
- Perform PAC before discharge
COMPLETE ABORTION:
- Usually, no need for uterine evacuation
- Monitor for bleeding and provide PAC
- Administer antibiotics before discharge
COMPLICATIONS OF ABORTION
Acute Complications:
- Incomplete abortion
- Sepsis
- Hemorrhage
- Uterine perforation
- Bowel injury
Long-term Complications:
- Chronic pelvic pain
- Pelvic inflammatory disease
- Tubal blockage and secondary infertility
- Ectopic pregnancy
- Increased risk of spontaneous abortion or premature delivery in subsequent pregnancies
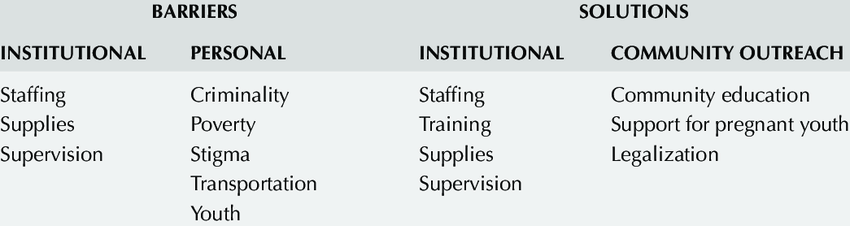
BARRIERS TO PAC
- Knowledge gap among health workers
- Inadequate infrastructure and facilities
- Insufficient supportive laws and policies
- Long distances to health facilities
- Lack of necessary equipment
- Mandatory waiting periods
- High costs of care
- Social stigma
- Health workers’ refusal based on personal beliefs.
PREVENTION OF ABORTION
- Primary Prevention: Avoiding unwanted pregnancies through education and family planning.
- Secondary Prevention: Preventing unsafe abortions through access to safe and legal abortion services.
- Tertiary Prevention: Managing post-abortion complications and preventing future unsafe abortions through comprehensive PAC.
Strategies for Prevention:
- Counseling and universal access to family planning.
- Increase availability of safe abortion services as per the law.
- Improve quality and accessibility of PAC.
- Educate communities about reproductive health and the dangers of unsafe abortion.
- Advocate for policy changes to protect women’s reproductive health.
- Promote gender equality and decision-making.
- Support education for girls and employment for women.
- Encourage attendance at antenatal service centers.
- Provide social protection for abandoned women.
- Offer non-judgmental counseling.
- Ensure access to emergency contraceptives.
Detailed Post-Abortion Care (PAC)
Post-abortion care involves several essential elements to ensure the health and well-being of women who have undergone an abortion.
1. Emergency Treatment of Abortion Complications:
Aspiration and Evacuation: For incomplete abortions, the uterus must be evacuated. The method of evacuation depends on the gestational period.
- For pregnancies below 12 weeks, Manual Vacuum Aspiration (MVA) is typically used.
- For pregnancies below 9 weeks, Misoprostol is used to terminate first-trimester intrauterine pregnancies. The standard dose is 800 micrograms administered orally, sublingually, or vaginally.
- Intravenous Fluids and Resuscitation: In cases of shock, administer normal saline (1 liter in 15-20 minutes) and use plasma expanders if available.
- Blood Transfusion: Monitor the amount of blood transfused and the patient’s response to treatment.
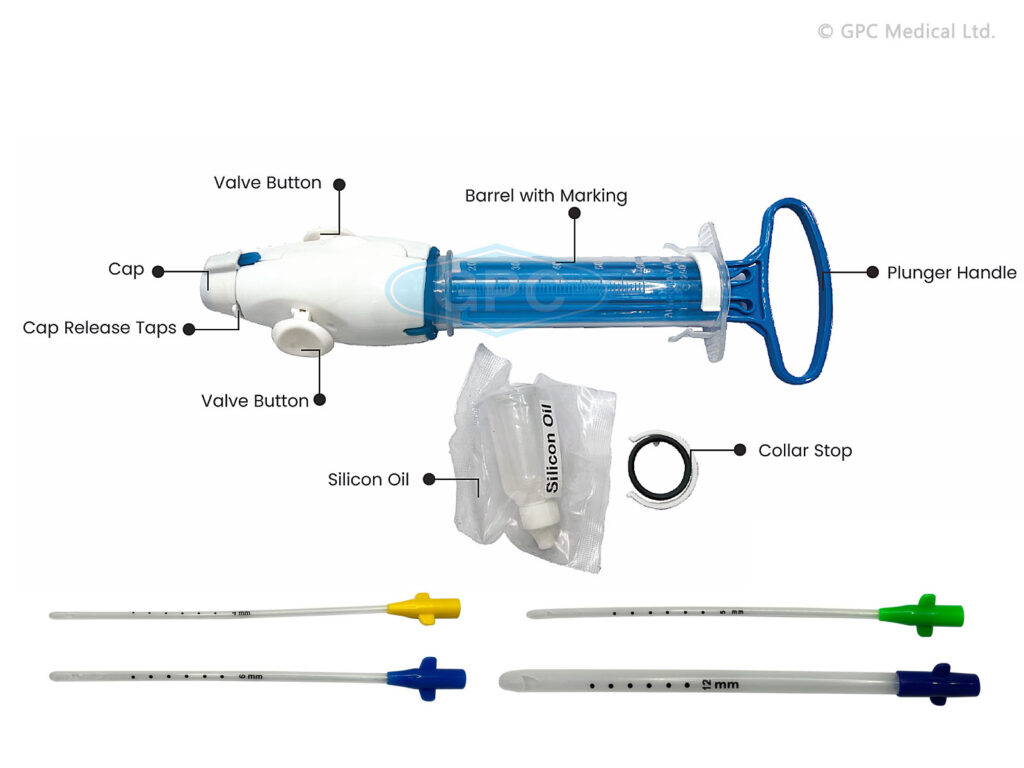
Manual Vacuum Aspiration (MVA):
Manual Vacuum Aspiration (MVA) is a method of termination of pregnancy where a healthcare provider uses a handheld device (an aspirator) to remove the contents from the uterus using suction undertaken with the patient awake. A narrow tube attached to a syringe is used to empty the womb using aspiration (gentle suction). Local anaesthetic is injected into the cervix (neck of the womb) to minimize discomfort.
Manual Vacuum Aspiration (MVA) is a preferred, appropriate, and cost-effective procedure for managing abortion in low-resource settings. It is particularly effective up to 12 weeks of pregnancy and has been proven highly efficacious in several randomized controlled trials. MVA has largely replaced dilation and curettage (D&C) in many industrialized and other countries.
Preparation:
- Prepare the patient, the room, equipment, supplies, and ensure the presence of an assistant.
- Select the appropriate size of the cannula based on the gestational age and uterine size.
Requirements for MVA Procedure:
Trolley (Top Shelf): | Trolley (Bottom Shelf): | Bedside Setup: |
Sterile MVA set Casco speculum Vulsellum uterine sound Receiver Bowl of cotton swabs Sponge holding forceps Cannula | Lidocaine or bupivacaine Syringe and needle KY jelly Antiseptic lotion Gumboots | Buckets Screens Apron |
Procedure for Manual Vacuum Aspiration:
1. Review Indications:
- Inevitable abortion before 16 weeks
- Incomplete abortion
- Molar pregnancy
- Delayed postpartum hemorrhage due to retained placental fragments
2. Provide Emotional Support and Encouragement: Ensure the woman feels supported and reassured throughout the procedure.
3. Offer Pain Relief: Administer paracetamol 30 minutes before the procedure or perform a para-cervical block.
4. Prepare the MVA Syringe:
- Assemble the syringe, close the pinch valve, and pull back on the plunger until the plunger arms lock.
- For molar pregnancy, have three syringes ready. For very early pregnancy, insert the cannula without prior cervical dilation.
5. Administer Oxytocin or Ergometrine: Give oxytocin 10 units IM or ergometrine 0.2 mg IM to firm the myometrium and reduce perforation risk.
6. Perform a Bimanual Pelvic Examination: Re-assess the size and position of the uterus and the conditions of the fornices.
7. Insert Sterile Speculum and Visualize the Cervix: Apply antiseptic solution to the vagina and cervix, especially around the os.
8. Check the Cervix for Tears or Protruding POC: Remove any products of conception (POC) with ring (or sponge) forceps.
9. Gently Grasp the Anterior Lip of the Cervix: Use ring forceps or a single-toothed tenaculum. If using a tenaculum, first inject 1 mL of 0.5% lignocaine solution into the cervix.
10. Dilate the Cervix if Needed: For missed abortion or prolonged retention of POC, use mechanical or osmotic dilators, or cervical priming with mifepristone or prostaglandin.
11. Insert the Cannula: While applying gentle traction to the cervix, insert the cannula through the cervix into the uterine cavity just past the internal os.
12. Attach the Prepared MVA Syringe: Hold the ring forceps or tenaculum and the end of the cannula in one hand and the syringe in the other. Release the pinch valve(s) to transfer the vacuum to the uterine cavity.
13. Evacuate Remaining Contents: Gently rotate the syringe from side to side and move the cannula back and forth within the uterine cavity without losing vacuum.
14. Check for Signs of Completion: Look for red or pink foam without tissue, a grating sensation, and the uterus contracting around the cannula.
15. Withdraw the Cannula: Detach the syringe, place the cannula in decontamination solution, and empty the syringe contents into a strainer.
16. Perform a Bimanual Examination: Check the size and firmness of the uterus post-procedure.
17. Inspect the Tissue Removed: Ensure complete evacuation, assess for molar pregnancy, and if necessary, strain and rinse the tissue for examination.
18. Address Absence of POC: If no POC are seen, consider complete abortion, breakthrough bleeding, or possible ectopic pregnancy.
19. Reinsert Speculum and Examine for Bleeding: If persistent bleeding or soft uterus, repeat evacuation.
Post-Procedure Care: Administer paracetamol 500 mg as needed, consider antibiotics, encourage the woman to eat, drink, and walk, and offer other health services.
Discharge Uncomplicated Cases: Discharge within 1-2 hours, advising on symptoms that require immediate attention.
Precautions for Performing MVA:
- Delay the procedure if conditions like shock, severe vaginal bleeding, or intra-abdominal injury are present, and stabilize the patient first.
- Stabilization involves oxygen, IV fluids, antibiotics for sepsis, and blood transfusion if needed.
Shock Management:
- Rapid, weak pulse, low blood pressure, pallor, sweatiness, rapid breathing, anxiousness, confusion, or unconsciousness.
- Treat with oxygen, IV fluids, antibiotics, and blood transfusion if necessary.
Severe Vaginal Bleeding:
- Heavy bright red bleeding, pallor, and blood-soaked materials.
- Assess all bleeding sources, stabilize, and evacuate POC.
Intra-Abdominal Injury:
- Distended abdomen, decreased bowel sounds, rigid abdomen, rebound tenderness, nausea, vomiting, pain, fever, or cramping.
- Immediate management with IV fluids, antibiotics, and potential surgery. Perform MVA after stabilization.
POST ABORTION CARE Read More »