Treatment of HIV/AIDS in Children (ARV therapy)
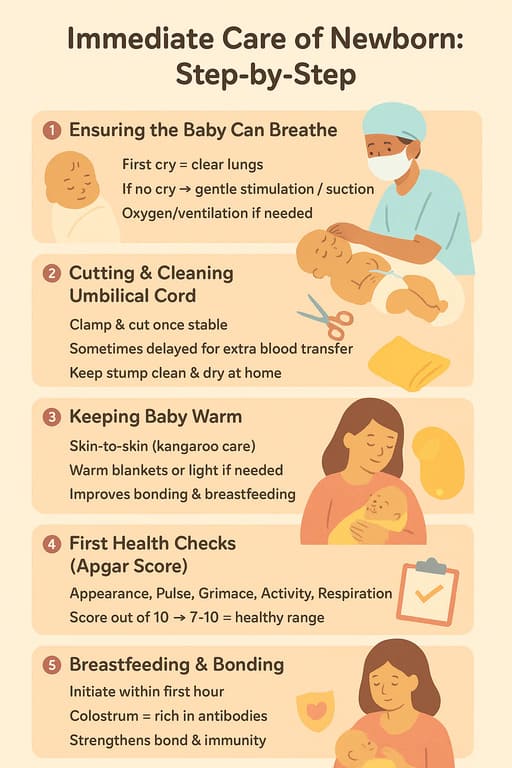
Managing HIV/AIDS in children is a complex, long-term endeavor that involves a combination of medical, nutritional, psychosocial, and developmental interventions. The primary goal is to suppress viral replication, restore immune function, prevent opportunistic infections, promote normal growth and development, and improve the child's quality of life and longevity.
Accurate and timely diagnosis is the critical first step before initiating Antiretroviral Therapy (ART). The diagnostic approach differs significantly for infants and children due to the presence of maternal antibodies in younger infants.
Diagnosis of HIV/AIDS in children relies on a combination of laboratory tests and clinical evaluation.
- Positive HIV Test Result: This is paramount. The type of test varies by age:
- For infants and children below 18 months of age: Virological tests (e.g., DNA PCR) are required to detect the virus itself, as maternal HIV antibodies can persist in the child's blood, making antibody tests unreliable for diagnosing infection in this age group.
- For children 18 months of age and above: Antibody tests can reliably confirm HIV infection, similar to adults.
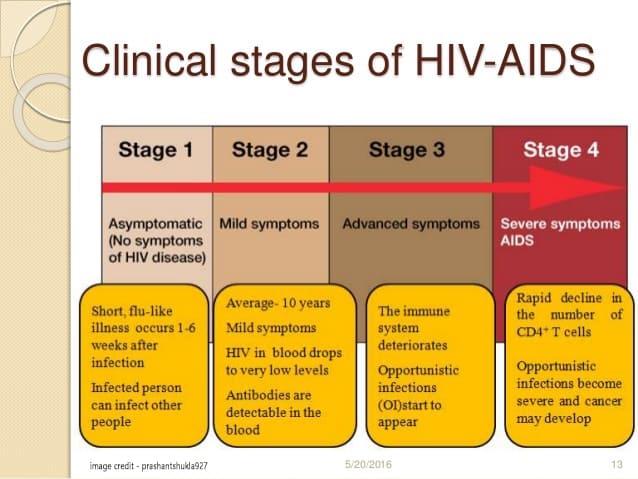
- Clinical Stage Criteria: The presence of HIV-related clinical signs and symptoms (as per WHO Clinical Staging) supports the diagnosis and indicates disease progression.
- Clinical Status, History, and Risk Factors: These should always be considered in conjunction with test results. A thorough history of exposure (e.g., maternal HIV status, breastfeeding history) and assessment of the child's health status are vital.
- Nucleic Acid Amplification Tests (NATs) / PCR Tests:
- Purpose: Detect the genetic material of HIV (DNA or RNA) directly, rather than antibodies or antigens.
- Application: Essential for diagnosing HIV infection in infants and children below 18 months of age. The most common type is the DNA PCR test, often performed on Dried Blood Spot (DBS) specimens.
- Note: ELISA Ag/Ab tests (which detect HIV antigens and/or antibodies) are commonly used for screening in blood donations or in older individuals, but are NOT suitable for diagnosing infection in infants <18 months due to maternal antibodies.
- HIV Antibody Tests:
- Purpose: Detect antibodies produced by the body in response to HIV infection.
- Application:
- To determine HIV exposure: In infants born to mothers of unknown HIV status.
- To exclude infection: In an infant at 18 months of age if the child has ceased breastfeeding for at least 6 weeks and all previous virological tests were negative.
- To confirm HIV infection: In children 18 months of age and above.
The process of providing HIV testing should follow a standardized protocol to ensure ethical considerations, accurate results, and appropriate follow-up. This protocol typically involves four key steps:
- Content: Educate the client/patient (or caregiver) about HIV transmission, basic prevention methods, the benefits of testing, possible test results, available support services, and the principles of consent and confidentiality.
- Risk Assessment: Conduct an individual risk assessment.
- Documentation: Fill out the HTS card.
- Engagement: Allow ample opportunity for questions.
- Sample Collection: Blood samples are used.
- For children below 18 months: A DNA PCR test is performed.
- For children 18 months and above: An antibody test is performed.
- Algorithm Adherence: Always refer to and follow the national HIV testing algorithms specific to different age groups.
- Readiness Assessment: Ensure the client/patient (or caregiver) is ready to receive the results.
- Result Delivery: Communicate results clearly and simply.
- Support: Address concerns, discuss disclosure, partner testing, and risk reduction strategies.
- Information Provision: Provide essential information about basic HIV care and ART services.
- Documentation: Complete the HTS card and HTS register.
- Referral: Provide information and facilitate referral to appropriate HIV prevention, treatment, care, and support services.
- Documentation: Complete referral forms and update registers (e.g., pre-ART and ART registers upon enrollment and initiation of ART).
HTS delivery must be non-discriminatory and uphold a human rights approach, observing the "5 Cs":
- Confidentiality: All client information must be kept private and not disclosed without consent.
- Consent:
- Individuals 12 years and above can consent to HTS themselves.
- For children, consent is obtained from a parent, guardian, next of kin, or legally authorized person.
- Counseling: Quality pre- and post-test counseling is mandatory as per approved HTS protocols.
- Correct Test Result: HTS providers must strictly adhere to national testing algorithms and Standard Operating Procedures (SOPs) to ensure accurate results.
- Connection to Appropriate Services: Clients must be linked to necessary HIV prevention, treatment, care, and support services.
- Infants 1-4 months (<6 kg): Heels are generally best.
- Infants 5-10 months (<10 kg): Toes are often suitable.
- Larger infants and older children: Ring or middle finger.
- Virological testing (DNA/PCR) is recommended for determining HIV status.
- Sample Type: Usually Dried Blood Spot (DBS) specimens.
- First DNA/PCR Test: Should be performed at six weeks of age or at the earliest opportunity thereafter.
- POSITIVE DNA/PCR Result: The child is HIV-infected.
- Action: Initiate ART immediately.
- Confirmation: Collect another blood sample on the day of ART initiation to confirm the positive result.
- NEGATIVE 1st DNA/PCR Result: The child is currently not infected but could become infected if still breastfeeding.
- Action: Retest using DNA/PCR six weeks after cessation of breastfeeding.
- Final Confirmation: If the 2nd DNA/PCR is also negative, a final rapid antibody test should be performed at 18 months of age (after breastfeeding cessation).
- POSITIVE DNA/PCR Result: The child is HIV-infected.
- Warm the area (e.g., heel or finger) to increase blood flow.
- Position the baby with the foot down for gravity assistance (if heel prick).
- Sterilize the area thoroughly with alcohol and allow it to air dry completely.
- Press the lancet into the foot/finger and prick the skin with a quick, firm motion.
- Wipe away the first drop of blood with a clean gauze.
- Allow a large drop of blood to collect.
- Add approximately 50µl (about 2 drops) into one circle on the DBS card, filling it completely.
- Fill at least 3 circles on the DBS card.
- Clean the foot/finger but do not bandage the prick site.
- Dispose of all contaminated materials appropriately.

- Never use expired HIV test kits.
- Avoid any modification of established procedures.
- Do not use clotted blood.
- Avoid "dirty" blood (e.g., contaminated with skin flakes, powder, sweat).
- Avoid introducing air bubbles into devices when adding samples.
- Strictly adhere to manufacturer instructions regarding:
- Amount of blood.
- Amount of buffer.
- Not exchanging buffers between different kits.
- Avoiding buffer contamination.
- Incubation times.
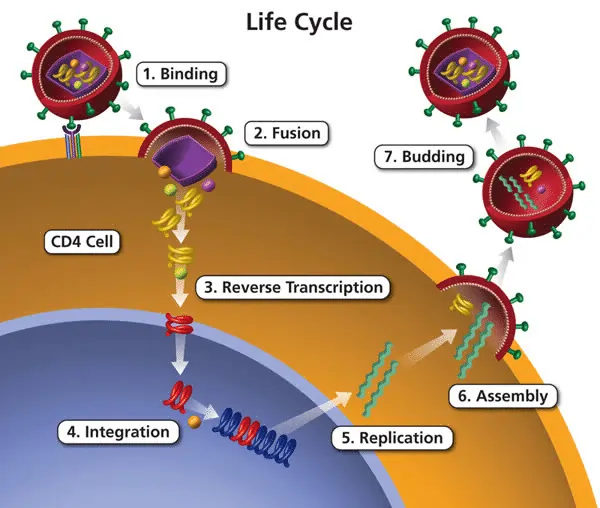
Antiretroviral therapy (ART) is the mainstay of HIV treatment. It involves the use of a combination of drugs that target different stages of the HIV life cycle, thereby suppressing viral replication. For children with HIV, ART is not just treatment; it is a life-saving intervention that has transformed HIV from a rapidly fatal illness into a manageable chronic condition.
- Viral Suppression: To reduce the HIV viral load to undetectable levels, thereby preventing further immune damage and reducing the risk of HIV transmission (though primarily a concern for adults, it has implications for future reproductive health of adolescents).
- Immune Reconstitution: To increase CD4+ T-lymphocyte counts and restore immune function, making the child less susceptible to opportunistic infections (OIs).
- Prevention of OIs: By restoring immune function, ART significantly reduces the incidence and severity of OIs.
- Promotion of Normal Growth and Development: By controlling the virus and preventing OIs, ART allows children to grow, gain weight, and achieve developmental milestones.
- Improved Quality of Life and Survival: Ultimately, ART aims to enable children with HIV to live long, healthy, and productive lives, comparable to their HIV-negative peers.
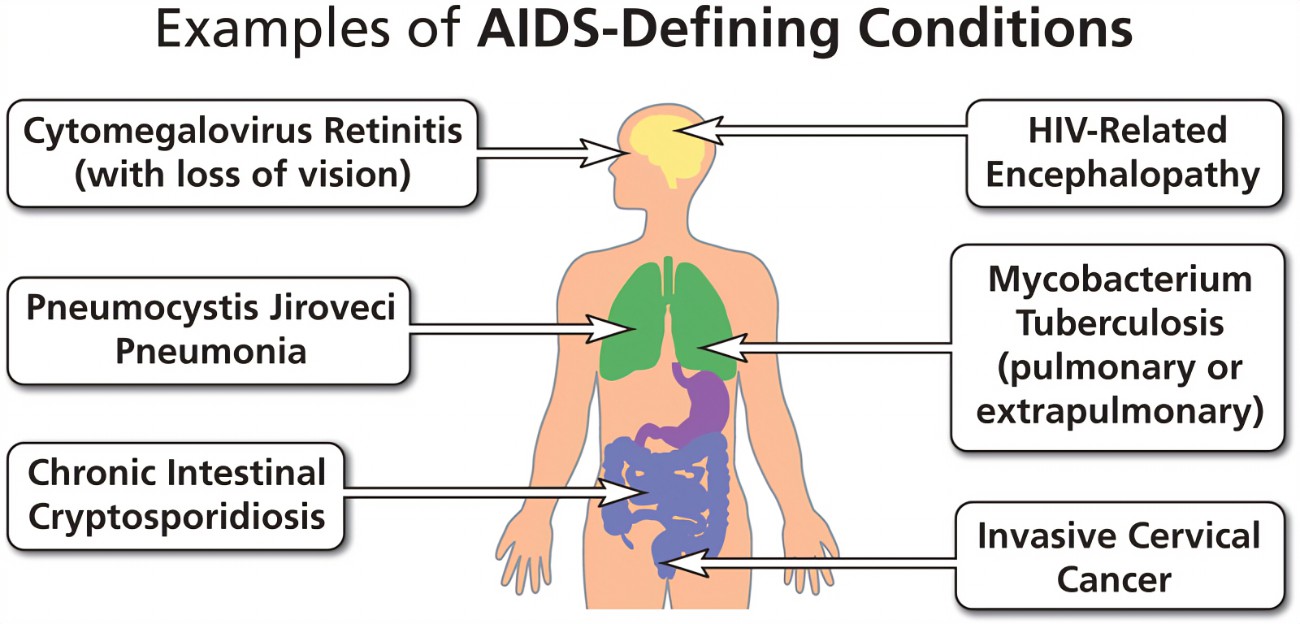
- Prevention of HIV-associated Morbidities: Such as HIV encephalopathy, cardiomyopathy, and nephropathy.
The current guidelines from the World Health Organization (WHO) and national bodies universally recommend "Treat All" – meaning all individuals diagnosed with HIV, regardless of clinical stage or CD4 count, should be offered ART. This is especially critical for children due to their rapidly progressing disease and immature immune systems.
Specifically for children, this translates to:
- All HIV-infected infants and children (0-19 years) should initiate ART regardless of clinical stage or CD4 count.
- Early initiation is crucial, especially in infants: Due to the rapid progression of HIV disease in young infants and higher rates of morbidity and mortality, ART should be started as soon as HIV infection is confirmed.
Rationale for "Treat All" in Children:
- Rapid Disease Progression: HIV progresses much faster in infants and young children than in adults.
- Higher Viral Loads: Infants often have higher viral loads, leading to more rapid immune destruction.
- Developmental Vulnerability: Their developing brains and bodies are particularly vulnerable to the damaging effects of uncontrolled HIV.
- Improved Outcomes: Numerous studies have shown that early ART initiation significantly reduces mortality and morbidity, improves neurodevelopmental outcomes, and normalizes growth in children.
An ART regimen typically consists of a combination of three antiretroviral drugs from at least two different classes. This combination approach is vital to achieve maximal viral suppression and prevent the development of drug resistance.
The main classes of antiretroviral drugs used in pediatric ART include:
- Nucleoside Reverse Transcriptase Inhibitors (NRTIs): These drugs block reverse transcriptase, an enzyme HIV uses to convert its RNA into DNA.
- Examples: Abacavir (ABC), Lamivudine (3TC), Zidovudine (AZT or ZDV), Tenofovir disoproxil fumarate (TDF), Emtricitabine (FTC).
- Non-Nucleoside Reverse Transcriptase Inhibitors (NNRTIs): These also block reverse transcriptase but in a different way than NRTIs.
- Examples: Efavirenz (EFV), Nevirapine (NVP), Rilpivirine (RPV).
- Protease Inhibitors (PIs): These drugs block protease, an enzyme HIV uses to cut long protein chains into smaller pieces needed for new virus particles.
- Examples: Lopinavir/ritonavir (LPV/r), Darunavir (DRV), Atazanavir/ritonavir (ATV/r). PIs are often "boosted" with low-dose ritonavir to increase their levels in the blood.
- Integrase Strand Transfer Inhibitors (INSTIs): These drugs block integrase, an enzyme HIV uses to insert its viral DNA into the host cell's DNA.
- Examples: Dolutegravir (DTG), Raltegravir (RAL), Bictegravir (BIC). INSTIs are increasingly becoming preferred first-line agents due to their potency, good tolerability, and high barrier to resistance.
WHO guidelines are regularly updated, but broadly, current recommendations for first-line ART in children emphasize potent, well-tolerated, and affordable regimens.
- For most children (especially school-aged and adolescents): A regimen including an INSTI, such as Dolutegravir (DTG), combined with two NRTIs is preferred. A common combination is DTG + 2 NRTIs (e.g., ABC + 3TC or TDF + 3TC/FTC).
- DTG is highly effective, generally well-tolerated, and has a high barrier to resistance, making it an excellent choice.
- For infants and young children (under 3 years or specific weight bands): PI-based regimens (e.g., LPV/r + 2 NRTIs) were previously preferred due to concerns about DTG dosing and safety data in this very young age group, but DTG is increasingly being recommended across all age groups including very young infants based on newer data and formulations.
- Weight-band dosing is critical for pediatric ART.
- Fixed-Dose Combinations (FDCs): Wherever possible, ART should be administered as fixed-dose combinations (FDCs), where multiple drugs are combined into a single pill. This simplifies dosing, improves adherence, and reduces the pill burden. Pediatric-friendly formulations (e.g., palatable granules, dispersible tablets) are crucial.
- Crucial for Success: Strict adherence to the prescribed ART regimen is paramount for its effectiveness. Missing doses allows the virus to replicate, potentially leading to increased viral load, immune deterioration, and the development of drug resistance.
- Challenges in Children: Adherence can be particularly challenging in children due to:
- Unpalatable medicines.
- Multiple pills and complex dosing schedules.
- Caregiver burden and understanding.
- Stigma and disclosure issues (especially in older children/adolescents).
- Strategies to Improve Adherence:
- Caregiver education and support: Ensuring caregivers understand the importance of ART, correct dosing, and potential side effects.
- Patient education: Age-appropriate education for the child/adolescent as they grow.
- Simplified regimens and FDCs: Using once-daily, single-pill regimens when possible.
- Palatable formulations: Using child-friendly forms of medication.
- Adherence counseling: Regular and ongoing counseling.
- Peer support groups: For older children and adolescents.
- Disclosure of HIV status: Thoughtful and age-appropriate disclosure can empower the child to take ownership of their treatment.
Monitoring is a continuous and crucial component of HIV management in children on ART. It involves regular assessments to evaluate the effectiveness of the treatment, detect potential side effects, identify new opportunistic infections, and ensure overall well-being and adherence. Effective monitoring allows for timely adjustments to treatment plans, optimizing long-term outcomes.
Monitoring an HIV-infected child on ART typically involves assessing several key parameters:
- Growth and Development: Regular assessment of weight, height, head circumference (in infants), and plotting on growth charts. This is a crucial indicator of treatment success and overall health. Monitor developmental milestones.
- General Physical Examination: Look for new or persistent signs/symptoms, such as fever, rash, lymphadenopathy, organomegaly, and signs of OIs.
- Nutritional Status: Assess for malnutrition or wasting and provide appropriate nutritional support and counseling.
- ART Adherence: Regularly assess and reinforce adherence to medication. This involves direct questioning, pill counts (if feasible), and discussing any challenges.
- Side Effects of ART: Monitor for both acute and chronic drug-related toxicities (e.g., skin rashes, gastrointestinal upset, neurological symptoms, lipodystrophy).
- Tuberculosis (TB) Screening: Regular screening for TB disease is vital given its high co-infection rate with HIV.
- Immunization Status: Ensure the child is up-to-date on all routine childhood immunizations.
- Purpose: CD4+ T-lymphocyte count (or percentage) measures the strength of the immune system. An increase in CD4 count indicates immune recovery.
- Frequency: Typically measured at baseline (before starting ART) and then every 3-6 months, or as clinically indicated.
- Interpretation: A rising CD4 count/percentage signifies a good response to ART. A falling CD4 count may indicate treatment failure or non-adherence.
- Purpose: Measures the amount of HIV RNA in the blood. It is the most sensitive indicator of ART effectiveness.
- Frequency: Baseline, and then typically 3-6 months after ART initiation, and every 6-12 months thereafter. More frequent monitoring may be needed if there are concerns about adherence or treatment failure.
- Interpretation:
- Viral Suppression: A viral load below the detectable limit (e.g., <20, <50, or <1000 copies/mL depending on the assay) indicates successful ART and good adherence. This is the primary goal of ART.
- Virological Failure: A persistently high or increasing viral load despite being on ART, or a confirmed viral load >1000 copies/mL (WHO definition), suggests treatment failure, often due to non-adherence or drug resistance.
- Purpose: To detect and manage potential side effects of antiretroviral drugs on various organ systems.
- Common Tests:
- Full Blood Count (FBC): To check for anemia (e.g., with AZT), neutropenia, or thrombocytopenia.
- Kidney Function Tests (Creatinine, eGFR): To monitor for nephrotoxicity, especially with tenofovir (TDF).
- Liver Function Tests (ALT, AST): To monitor for hepatotoxicity, which can occur with many ART drugs.
- Lipid Profile (Cholesterol, Triglycerides): To monitor for dyslipidemia, particularly with some PIs.
- Blood Glucose: To monitor for hyperglycemia.
- Frequency: Typically at baseline, 1-3 months after ART initiation, and then every 6-12 months, or as clinically indicated based on the specific ART regimen and child's health status.
Treatment failure can be clinical, immunological, or virological. Virological failure is the most sensitive and earliest indicator.
- Suspected Treatment Failure:
- Assess Adherence: The first step is always to thoroughly re-assess and address adherence issues. Most cases of virological failure are due to suboptimal adherence. Provide intensive adherence counseling.
- Confirm Virological Failure: Repeat viral load testing after a period of intensive adherence counseling (e.g., 3-6 months).
- Investigate Drug Resistance: If confirmed virological failure despite good adherence, consider performing a drug resistance test (genotyping). This guides the selection of a new regimen.
- Switch to Second-Line Regimen: Based on resistance test results (if available) or empirical guidelines, switch the child to a different ART regimen, often involving different drug classes or more potent drugs (e.g., a PI-based regimen if not already on one, or a new INSTI combination).
- Importance: Timely and age-appropriate disclosure is a critical part of monitoring and management. It empowers the child to understand their health, take ownership of their treatment, and better adhere to ART as they mature. It also helps them navigate social challenges.
- Process: It should be a planned, gradual, and ongoing process, not a single event.
- Early Childhood (0-6 years): Simple explanations and reassuring messages about taking medicine to stay healthy.
- Middle Childhood (7-12 years): More concrete explanations, answering questions honestly, introducing the term "HIV" if appropriate.
- Adolescence (13+ years): Full disclosure, detailed discussions about living with HIV, adherence, prevention, and future planning.
- Support: Involve caregivers, healthcare providers, and psychosocial support staff in the disclosure process.
Linkage refers to the critical process of connecting individuals newly diagnosed with HIV from the point of testing to subsequent prevention, care, and treatment services. Successful linkage means the patient actually receives the services they were referred to. This is especially crucial for children, where timely intervention is paramount for survival and well-being.
- Prompt Action: For all clients testing HIV-positive, linkage should ideally occur within seven days for referrals within the same facility and within 30 days for inter-facility or community-to-facility referrals.
- Facilitators: The use of trained lay providers (e.g., community health workers, peer leaders, expert clients) as linkage facilitators is highly recommended to bridge gaps and support patients through the process.
- Internal Facility Linkage:
- Definition: Connecting a newly diagnosed patient from one department (e.g., HIV testing center, pediatric ward) to another department within the same health facility (e.g., the ART clinic or pediatric HIV clinic) for comprehensive HIV treatment, care, and support services.
- Inter-Facility Linkage:
- Definition: Connecting a newly diagnosed patient from one health facility to another different health facility for ongoing HIV treatment, care, and support services.
- Tracking: The referring facility has a responsibility to track all referred HIV-positive patients to ensure they are enrolled in care and initiated on ART within 30 days.
- Community-to-Facility Linkage:
- Definition: Connecting a client who tests HIV-positive in a community setting (e.g., mobile testing clinic, home-based testing) to a health facility for HIV treatment, care, and support services.
- Community Health Systems: HTS programs should establish robust community health systems (involving peer leaders, expert clients, community health volunteers) to mobilize individuals for testing and facilitate prompt linkage (within 30 days) for all who test positive.
This outlines a best-practice pathway for ensuring a smooth transition within a single facility:
- Post-Test Counseling:
- Provide accurate test results clearly and empathetically.
- Inform about available care services both within the facility and in the broader catchment area.
- Explain the immediate next steps for care and treatment.
- Emphasize the significant benefits of early ART initiation and the risks of delaying treatment.
- Identify and collaboratively address any potential barriers to linkage (e.g., transport, stigma, fear).
- Involve the parent/caregiver and child (age-appropriately) in decision-making regarding their care plan.
- Complete client cards and all necessary referral notes and forms (e.g., triplicate referral form).
- Introduce and hand over the patient to a dedicated linkage facilitator.
- If same-day linkage is not feasible, schedule an appointment for the client at the clinic and diligently follow up to ensure attendance.
- Escort to the HIV Clinic:
- The linkage facilitator physically escorts the client to the ART clinic, carrying all relevant linkage forms.
- The client is formally handed over to the responsible staff at the receiving clinic.
- Enrollment at the HIV Clinic:
- Register the patient in the pre-ART register.
- Create an individual HIV/ART card/file for the patient.
- Provide comprehensive ART preparatory counseling, covering adherence, side effects, and expectations.
- Conduct necessary baseline investigations (as outlined in the monitoring section).
- If the patient is ready (and all criteria met, especially the "Treat All" for children), initiate ART immediately.
- Continue with ongoing counseling support (e.g., disclosure, psychosocial support).
- Coordinate integrated care as needed (e.g., for TB/HIV co-infection, PMTCT follow-up for the mother).
- Schedule an appropriate follow-up appointment with the patient/caregiver.
This comprehensive framework outlines the essential components for holistic care of children living with HIV:
- Confirm HIV Status as Early as Possible: Early diagnosis is critical for prompt intervention.
- Monitor the Child’s Growth and Development: Regular assessment of physical growth and achievement of developmental milestones.
- Ensure Immunizations are Started & Completed as per Schedule: Protect against vaccine-preventable diseases.
- Provide Prophylaxis for Opportunistic Infections (OIs): Prevent common and severe infections.
- Actively Look for and Treat Infections Early: Prompt recognition and management of any infections.
- Counsel Mother & Family on:
- Optimal infant feeding practices.
- Good personal & food hygiene.
- Follow-up recommendations for the child.
- Conduct Disease Staging for the Infected Child: To assess disease progression and guide management.
- Offer ARV Treatment for the Infected Child: Initiate ART as per "Treat All" guidelines.
- Provide Psychosocial Support for the Infected Child and Mother/Family: Address emotional, mental, and social well-being.
- Refer the Infected Child to Higher Levels of Specialized Care if Necessary: For complex cases or specific complications.
Treatment of HIV/AIDS in Children (ARV therapy) Read More »