Pain assessment
Pain assessment
We cannot assess pain without understanding what pain is, right? Let us first explore pain!
Pain
Pain is an unpleasant experience that involves both physical sensations and emotions, often connected to actual or possible harm to body tissues.
Pain is defined by the person experiencing it and exists as they perceive it. “Pain is what the patient says hurts.”
Pain is subjective, as individuals shape their own understanding of pain based on their personal experiences.
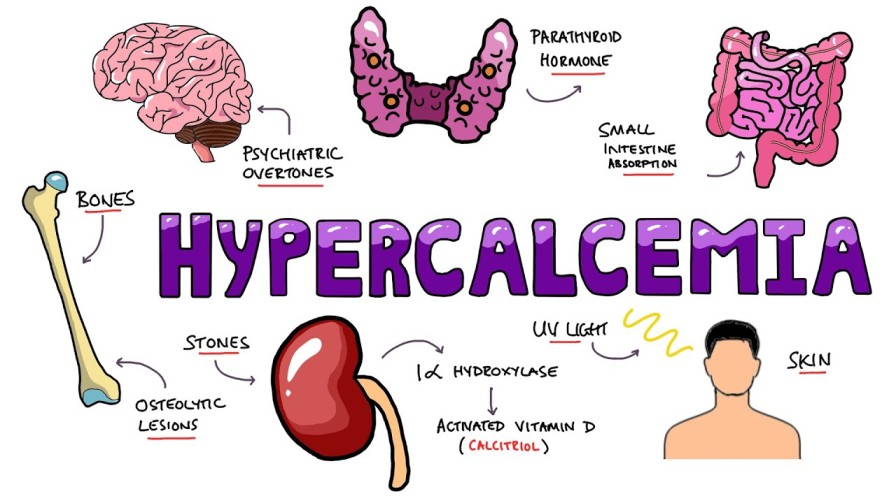
Pain is the most common and feared symptom among those nearing the end of life. It affects around 98% of cancer and HIV/AIDS patients.
Cancer pain is typically constant and worsens as the disease progresses.
In developing countries where less than 5% of cancer patients have access to chemotherapy or radiotherapy, pain tends to worsen gradually until death.
A study conducted in Africa involving stage IV AIDS patients identified the most common pains reported:
- Lower limb pain (66%) caused by peripheral neuropathy
- Mouth pain (50.5%)
- Headache (42.3%)
- Throat pain (39.8%)
- Chest pain (17.5%)
The Purpose of Acute Pain: Acute pain serves as a useful mechanism that alerts organisms to the presence of harmful or potentially harmful stimuli in their environment, such as extreme heat or cold.
Acute Pain in Cancer: Cancer patients may experience pain due to different factors, including:
- Direct effects of the disease, such as tumors infiltrating sensitive structures that cause pain
- Treatment-related effects, like radiotherapy that can harm visceral, musculoskeletal, and nervous tissues.
- Surgery, chemotherapy, and radiotherapy are all associated with potential side effects that may cause pain.
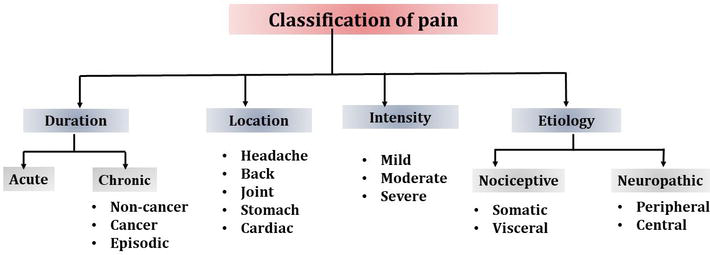
Classification of Pain
Pain comes in various forms and can be categorized based on its duration (acute or chronic) and its underlying physiological mechanism (nociceptive or neuropathic).
It is common for multiple types of pain to coexist. To effectively assess and treat pain, it is important to identify the specific type(s) of pain being experienced
| Type | Cause | Description | Treatment | |
|---|---|---|---|---|
| Nociceptive | Activation of intact nerve pathways | Normal response to a stimulus | Address underlying cause | |
| Tissue injury to skin, muscle, bone | Nagging, throbbing, aching sensation | Standard pain medication (according to WHO ladder) | |
| (e.g., toothache, burns) | ||||
| Pain originating from organs | Crampy, nagging sensation (e.g., crampy bowel pain) | Additional medication | |
| Neuropathic pain | Nerve pathway damage resulting in abnormal response to stimulus | Burning, shooting, pricking, electric shock, numb sensations (e.g., neuropathy, herpes zoster) | Antidepressants, anticonvulsants, +/- Opiates | |
| Damage to peripheral nerves | Pain originating from peripheral nerves (e.g., nerve compression, chemotherapy-induced damage) | Targeted nerve pain medications | |
| Damage to the central nervous system | Pain originating from the central nervous system (e.g., stroke, spinal cord injury) | Neuropathic pain medications | – |
| Duration of Pain | ||||
| Definite injury or illness | Characterized by definite onset, limited duration, and clinical signs of sympathetic overactivity (e.g., tachycardia, pallor) | Address underlying cause | |
| Chronic pathological process | Gradual or ill-defined onset, continues unabated(increase in intensity, and strength) may become severe No typical signs of sympathetic overactivity | Address underlying cause | |
| Pain according to Situation | ||||
| Transitory exacerbation of pain on a controlled pain background | Occurs on a background of otherwise controlled pain | Adjust pain management plan as needed | |
| Occurs in specific circumstances (e.g., after a particular movement) | Occurs only in certain circumstances (e.g., after a particular movement) | Address specific triggers or interventions | |
| Related to procedures or interventions | Appropriate pain management during procedures |
Factors that influence pain
- The patient’s mood
- The patient’s morale
- The meaning of the pain for the patient e.g. the meaning of pain in advanced cancer is
- “I ‘m incurable”: I ‘m going to die.
- Psychological and Spiritual Factors: Pain can be influenced by psychological factors, such as mood, morale, and the meaning of pain for the individual. Spiritual beliefs and practices may also impact pain perception.
- Social Circumstances: Social factors, including support systems, relationships, and cultural influences, can affect the experience and management of pain.
- Emotional Component of Pain: Pain is not purely a physical sensation but also involves an emotional component. Individuals may describe their pain using terms like agonizing, cruel, or terrible.
- Integrated Multi-disciplinary Teams: Managing chronic pain often requires the involvement of various healthcare professionals working together as a team. This interdisciplinary approach ensures comprehensive care and improved outcomes.
- Holistic Support: Providing holistic support to patients with chronic pain can greatly impact their quality of life. This support focuses on addressing feelings of helplessness, building resilience, and addressing the physical, emotional, and social aspects of pain.
- Gender Differences: Biological, psychological, and social factors contribute to differences in how men and women experience pain. This includes variations in pain perception, response to treatment, and effectiveness of different pain management approaches.
| Factors Increasing Pain | Factors Decreasing Pain |
|---|---|
| Discomfort | Relief of other symptoms |
| Insomnia | Understanding |
| Fatigue | Companionship |
| Anxiety | Creative activity |
| Fear | Relaxation |
| Anger | Reduction in anxiety |
| Sadness | Elevation in mood |
| Depression | Analgesics |
| Boredom | Anxiolytics |
| Antidepressants |
Total pain
- The concept of total pain was developed by Cicely Saunders in 1960s.
She acknowledges that pain is not just a physical phenomenon. It encompasses physical, psychological, social and spiritual aspects of suffering.
- Physical: undesirable effects of treatment, insomnia, chronic fatigue
- Psychological: anger at delays in diagnosis, anger in treatment failure, disfigurement, fear of pain/death, feelings of helplessness, anger at friends who do not visit.
- Social: worry about family, Worry about finance, loss of job, loss of income, loss of social position.
- Spiritual: Why has this happened to me? Why does God allow me to suffer like this? Is there any meaning or purpose in life?
The impact of pain
- Severe pain in advanced cancer patients has negative physiological and psychological complications that may worsen an already bad situation.
- Interaction of pain with other symptoms (e.g. nausea, constipation, shortness of breath, depression, anxiety, insomnia) may worsen the patient’s condition.
- The patient’s functional status is further impaired.
- The patient’s autonomy is challenged.
- The patient’s dignity is challenged.
- The patient and family may interpret pain as impending death.
Barriers to pain management
- Inadequate pain assessment
- Inadequate knowledge about pain and its management
- Concerns about possible side effects of pain medications
- Patient and doctor’s attitudes, fears and misconceptions about pain and opioids.
- Poorly accessible or unavailable pain management services.
Principles for Assessing and Managing Pain
Comprehensive Approach: All aspects of total pain, including psychological, spiritual, social, cultural, and physical dimensions, should be addressed during pain assessment and management.
Multiple Causes of Pain: Pain can stem from various sources such as diseases (e.g., HIV), their consequences (e.g., opportunistic infections), treatments (e.g., chemotherapy), or concurrent disorders (e.g., arthritis).
Goal of Palliative Care: The aim of palliative care is to alleviate pain effectively, ensuring it does not significantly impact the patient’s quality of life.
Subjectivity of Pain: Pain is a subjective experience, relying on what the patient communicates and describes.
Guideline-based Management: Pain should be managed according to the guidelines provided by the World Health Organization (WHO) and the analgesic ladder.
Considerations in Pediatric Pain: Incident and procedural pain are particularly significant in children and should be addressed appropriately.
Clinical Presentation of Pain
Different types and causes of pain manifest with distinct clinical presentations:
Visceral Pain: This type of pain is not well localized and presents as a constant, aching sensation.
Bone Pain: Bone pain is well localized with local tenderness. It resembles a nagging toothache, worsens with movement and weight bearing, and is often caused by the release of prostaglandins.
Colic: Colic refers to gripping pain associated with spasms, typically occurring in the middle or upper abdomen (bowel) or related to micturition (bladder).
Raised Intracranial Pressure: Increased pressure within the skull results in a generalized headache that worsens in the mornings, when lying down, and during coughing. This type of pain may be accompanied by symptoms like nausea, projectile vomiting, and blurred vision.
Neuropathic Pain: Neuropathic pain is constant or aggravated by movement. It is described as burning, sharp, stabbing, shooting, or a nagging ache. It may be associated with altered sensation and can follow a dermatomal distribution.
Spiritual Pain: Spiritual pain is an emotional form of suffering, often expressed through dreams or nightmares.
Other Forms of Pain: Patients may exhibit refusal to take medication or engage in self-harming behaviors as active manifestations of pain.